Heat Effects Chapter 4 Sensible Heat Effects Latent

Heat Effects Chapter 4 Sensible Heat Effects, Latent Heats of Pure Substances, Standard Heat of Reaction, Temperature Dependence of Ho , Heat Effects of Industrial Reactions
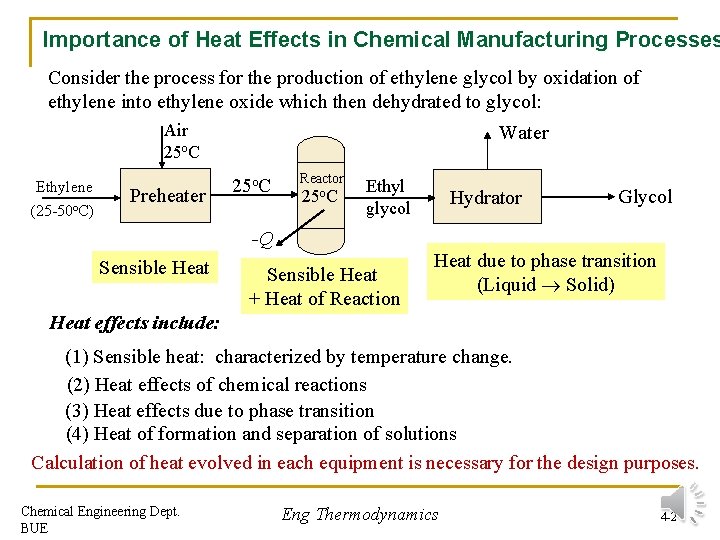
Importance of Heat Effects in Chemical Manufacturing Processes Consider the process for the production of ethylene glycol by oxidation of ethylene into ethylene oxide which then dehydrated to glycol: Air 25 o. C Ethylene (25 -50 o. C) Preheater Water 25 o. C Reactor 25 o. C Ethyl glycol -Q Sensible Heat + Heat of Reaction Hydrator Glycol Heat due to phase transition (Liquid Solid) Heat effects include: (1) Sensible heat: characterized by temperature change. (2) Heat effects of chemical reactions (3) Heat effects due to phase transition (4) Heat of formation and separation of solutions Calculation of heat evolved in each equipment is necessary for the design purposes. Chemical Engineering Dept. BUE Eng Thermodynamics 4 -2

Sensible Heat Effects It is the heat transfer to the system, or from the system, due to the temperature change when there is no chemical reaction and no change in the composition and no phase transition. Objective: Develop a relation between heat transfer and the temperature change v For pure homogeneous system, no change in composition: U = U(T, V) Remember that for mechanically reversible constant-V process: Q = U Chemical Engineering Dept. BUE Eng Thermodynamics 4 -3

Similarly, H = H(T, P) when: (1) d. P = 0 or (2) H H(P) Valid for: (a) Ideal gas (b) Low P gases Remember that for mechanically reversible constant-P non flow process: Q = H Evaluation of ∫CP d. T requires knowledge of temperature dependence of the heat capacity. In dimensionless: Chemical Engineering Dept. BUE Eng Thermodynamics 4 -4

Thermodynamic properties are often accomplished in two steps: Ø Calculate values for ideal-gas wherein ideal gas heat capacities are used, Ø Correction of the ideal-gas values to the real-gas values is added. Remember: a real gas becomes ideal gas as P 0 For ideal gas, heat capacities are designated as: Cig. P and Cig. V and can be expressed as: where • The parameters A, B, C and D are tabulated for number of materials • At low pressure, Cig. P Crg. P • Often CP for real gas is not too far from CP for ideal gas, thus it would be logical to assume that • For a gas mixture: Chemical Engineering Dept. BUE yi: mole fraction of each component Eng Thermodynamics 4 -5

Example: Calculate the heat required to raise the temperature of 1 mole of methane from 260 to 600 o. C in a flow process at a pressure sufficiently low ( ideal gas). For methane: A = 1. 702 B = 9. 08× 10 -3 C = -2. 16× 10 -6 D = 0 Chemical Engineering Dept. BUE Eng Thermodynamics 4 -6

Calculations involving sensible heat • If T & T are known Straightforward calculations 1 2 • If H is given and it is required to determine T 2 Trial & error calculations For solving the resulting equation, rearrange so that T 2 appears in the LHS , then solve for T 2 and make LHS as calculated while the RHS as guessed one: (See Example 4. 3, p. 132) Chemical Engineering Dept. BUE Eng Thermodynamics 4 -7

Matlab implementation The function to be solved for T 2: Define the function and then use the solver fsolve to solve for T 2 starting from an initial condition of T 2=550 K. In this case C=0. F='(37218/8. 314) - (3. 578*(x - 533. 15) + (3. 02 e-3/2)*(x^2 - 533. 15^2))'; x=fsolve (F, 550) Chemical Engineering Dept. BUE Eng Thermodynamics 4 -8

Latent Heat of Pure Substances Vaporization, or condensation, fusion or solidification, occur at constant temperature and pressure. However, theses processes require the transfer of certain amount of heat to or from the substance. Latent Heat Solid Liquid Latent heat of fusion (melting) Liquid vapor Latent heat of vaporization (melting) Ø Energy evolved upon separation or combining molecules together during the phase transition process Solid Fusion (melting) Chemical Engineering Dept. BUE Solidification Liquid Vaporization Condensation Eng Thermodynamics Vapor 4 -9
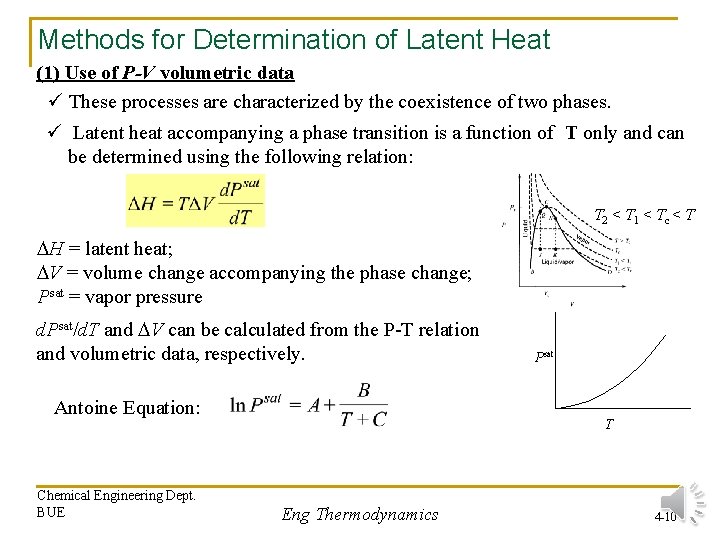
Methods for Determination of Latent Heat (1) Use of P-V volumetric data ü These processes are characterized by the coexistence of two phases. ü Latent heat accompanying a phase transition is a function of T only and can be determined using the following relation: T 2 < T 1 < Tc < T H = latent heat; V = volume change accompanying the phase change; Psat = vapor pressure d. Psat/d. T and V can be calculated from the P-T relation and volumetric data, respectively. Antoine Equation: Chemical Engineering Dept. BUE Psat T Eng Thermodynamics 4 -10

(2) Use of Calorimeteric data ü H can be determined experimentally (calorimetrically) ü Results are normally represented by certain correlations which are available in literature If point 1, the reference point (T 1, P 1) enthalpy assigned to be Ho = 0; At point 2, T = T 2, according to the first law: Q = H 2 Chemical Engineering Dept. BUE Eng. Thermodynamic 4 -11

(3) Approximate Methods Often, H is not available at the temperature of interest. Thus, approximate methods serve two purposes: a. Prediction of Hvap at normal boiling point Tn (i. e. at 1 atm) b. Estimation of Hvap at any temperature from the known value at certain temp. Riedel equation Valid for any substance Tn = normal boiling point; Hn = molar heat of vaporization at Tn Pc = critical pressure; Trn = reduced temperature at Tn (Trn = Tn/Tr) At any other temperature, T 2, latent heat of vaporization, H 2, can be determined based on the known latent heat, H 1, at temperature T 1: Watson equation Valid for any substance Chemical Engineering Dept. BUE Eng. Thermodynamic 4 -12

Standard Heat of Reaction ü So far heat was evolved due to physical changes only. ü Chemical reactions are accomplished either by the transfer of heat (exothermic or endothermic reactions) or by temperature changes during the course of reaction. ü In chemical reactions, heat is evolved due to the difference in the molecular structure between products and reactants, i. e. different levels of energy Example: Reactants in combustion reactions possess greater energy based on their structure than do the products: Reactants High energy Products Low energy +Q Ø To reduced the number of possible data for heat of reaction, reactions can be carried out at standard conditions. Ø For consistent treatment of reactions, heat effects can be done when both reactants and products are all at the same temperature. Chemical Engineering Dept. BUE Eng. Thermodynamic 4 -13

Standard Heat of Reaction For purpose of tabulation, consider the following reaction: a. A + b. B c. C + d. D Standard heat of reaction: The enthalpy change when a moles of A and b moles of B react to form c moles of C and d moles of D at their standard states at temperature T. Standard state: a particular state of a species at specification of T, P, composition and physical state Standard, designated as “o” • Gases: Pure substance in the ideal gas state at 1 bar. • Liquids & Solids: Actual pure liquid or solid at 1 bar. : Standard-state heat capacity listed in Table C-1 ü All conditions at standard state are fixed except T, i. e. standard state = f(T) Example: ½ N 2 + 3/2 H 2 NH 3 Doubling the stoichiometry H should be doubled N 2 + 3 H 2 2 NH 3 Petroleum and Chemical Engineering Dept. Sultan Qaboos University Thermodynamics I 4 -14

Standard Heat of Formation ü Tabulation of data for standard heat of all possible reactions is impossible ü Standard heat of formation of the component would be more practical Formation reaction: a reaction which forms a single compound from its constituent elements C + ½ O 2 CH 3 OH Formation reaction H 2 O + SO 4 H 2 SO 4 Not formation reaction • Formation reaction is based on 1 mole of compound, i. e. heat per 1 mole of compound • Heat of reaction at any temperature can be calculated if it is known at any other temperature Heat of formation is tabulated at 25 o. C, designated as (See Table C. 4, App. C) Can be added for each component in the reaction to give the standard heat of the desired reaction • Heat of formation for pure element is assigned as zero in order to determined Hof, 298 Petroleum and Chemical Engineering Dept. Sultan Qaboos University Thermodynamics I 4 -15

For a given reaction, heat of reaction can be calculated based on the heat of formation for each compound in the reaction using the following formula: Example: Calculate the heat of reaction at 25 o. C for the following reaction: CO 2 (g) + H 2 (g) CO (g) + H 2 O (g) Heat of formation are given for each compound: Reactants Products CO 2: CO: H 2 O: = (-241818 -110525)-(-393509) = 41166 J/mol Petroleum and Chemical Engineering Dept. Sultan Qaboos University Thermodynamics I 4 -16

Temperature Dependence of Ho Objective: Calculation of standard heats of reaction at a certain temperature from the knowledge of heat of reaction at a reference temperature ( ). Consider the following general chemical reaction: | i| +ve for products | i| -ve for reactants i itself is called the stoichiometric number | i| is the stoichiometric coefficient Example: N 2 = -1; H 2 = -3; NH 3 = 2 Thus, in terms of the stoichiometric number , the standard heat of reaction can be expressed as: Petroleum and Chemical Engineering Dept. Sultan Qaboos University Thermodynamics I 4 -17

Where Hio is the enthalpy of species i in its standard state which is equals to the standard heat of formation, that is: Example: From Table C. 4 at 298. 15 K H of Species i Petroleum and Chemical Engineering Dept. Sultan Qaboos University Thermodynamics I HCl (g) -92, 307 O 2 0. 0 H 2 O (g) -241, 818 Cl 2 0. 0 4 -18

For any species, i, enthalpy is function of T and P, but at constant P the change of enthalpy for species i due to change in T can be expressed as Thus, in the standard-state, since P is fixed, this can be written as: Multiplying by i Summing over all components where: Petroleum and Chemical Engineering Dept. Sultan Qaboos University Thermodynamics I 4 -19

Since LHS = RHS Integrating: Calculations involve the following steps: 1. Calculate heat of reaction at the reference temperature, T 0 H 0 o 2. Determine: 3. Perform the integration 4. Multiply the integration results by R and add the result to step 1. Petroleum and Chemical Engineering Dept. Sultan Qaboos University Thermodynamics I 4 -20

Example 4. 6 Calculate the standard heat of the methanol-synthesis reaction at 800 o. C: Table of data i o Hf A 103 B 106 C 10 -5 D CH 3 OH 1 -200, 660 2. 211 12. 216 -3. 450 0. 000 CO -1 -110, 525 3. 376 0. 557 0. 000 -0. 031 H 2 -2 0. 0 3. 249 0. 422 0. 000 0. 083 i i , 298 Calculation of the constants: Similarly: B = 10. 815 10 -3 C = -3. 45 10 -6 D = -0. 135 105 Perform the integration: = -1615. 5 K Ho = -90135+8. 314(-1615) = 103566 J Petroleum and Chemical Engineering Dept. Sultan Qaboos University Thermodynamics I 4 -21

Problem 4. 21(f) & 4. 23 Calculate the standard heat of reaction at 25 o. C and at 800 o. C for the reaction: Table of data (from C. 1) Calculation of the constants: Similarly: Perform the integration: i i H of H 2 O (g) 12 -241818 3. 47 0. 00145 N 2 (g) 7 0. 0 3. 28 0. 00593 NH 3(g) -8 -46110 3. 578 0. 00302 NO 2 (g) -6 33180 4. 982 0. 00119 i , 298 A B B = 0. 002761 = 24894 K Ho = -273201 + 8. 314(24894) = -2525. 0 k. J Petroleum and Chemical Engineering Dept. Sultan Qaboos University Thermodynamics I 4 -22

Industrial Reactions n Industrial reactions are carried out under standard-state conditions. Ø Ø Actual reactions may not present in stoichiometric proportions. Reactions may not go to completion. Products temperature might be different that reactant temperature. Inerts may present. Each reaction requires special treatment. Petroleum and Chemical Engineering Dept. Sultan Qaboos University Thermodynamics I 4 -23

Example What is the maximum temperature that can be reached by the combustion of methane with 20% excess air? Both the methane and the air enter the burner at 25 o. C. CO CH 4 2 T=25 o. C 20% excess O 2 H 2 O i i A B 10 -5 D ni, exit (mole) CH 4 -1 -74520 1. 702 0. 009081 0. 0 O 2 -2 0. 0 3. 639 0. 5060 -0. 227 0. 4 CO 2 1 -393509 5. 457 1. 045 -1. 157 1. 0 H 2 O (g) 2 -241818 3. 470 1. 450 0. 121 2. 0 0. 0 3. 280 0. 593 0. 040 9. 03 N 2 Petroleum and Chemical Engineering Dept. Sultan Qaboos University Thermodynamics I Tmax ? 4 -24

Assume combustion is complete and adiabatic: Q = 0. 0 Ho = 0. 0 Since enthalpy is a state function then assume the two paths: 1. Reaction occurs at 25 o. C to form the product 2. Products are heated to T Material balance: Assume basis 1 mole CH 4 Moles O 2 required = 2 moles Moles O 2 in excess = 2(0. 2)=0. 4 moles Moles of N 2 in = 2. 4(79/21)=9. 03 moles Reactant Product @ 1 bar &T . 0 0 o= H Const. T (25 o. C) 1 mol CO 4 2 mol H 2 O 0. 4 mol O 2 9. 03 mol N 2 Product @ 1 bar & 25 o. C 1 mol CH 4 2. 4 mol O 2 9. 03 mol N 2 where Petroleum and Chemical Engineering Dept. Sultan Qaboos University Thermodynamics I 4 -25

since Solve for T by trial and error procedure, to get: T = 2066 K Petroleum and Chemical Engineering Dept. Sultan Qaboos University Thermodynamics I 4 -26

Example 4. 8: Catalytic reforming of CH 4 with steam occurred according to the following reaction: (1) (2) § § 2 moles of steam entered the reactor for each mole of CH 4. Heat supplied to the reactor for a product temperature of 1300 K. CH 4 is completely converted. Product stream contains 17. 4 mol-% CO. If the reactants are at 600 o. C, calculate the heat requirement for the reactor. Petroleum and Chemical Engineering Dept. Sultan Qaboos University Thermodynamics I 4 -27

Solution (Ex. : 4. 8) -x -(1 -x) -x x -2(1 -x) 3 x 4(1 -x) Solution: Material balance to determine the constituents of the feed and the product streams Assume a basis of 1 mol CH 4 and 2 mold H 2 O fed to the reactor Let x be mol of CH 4 reacts by rxn 1 1 - x is mol of CH 4 reacts by rxn 2 CO: x Moles of product: H 2: 3 x + 4(1 -x) = 4 -x CO 2: 1 - x mole fraction of CO=0. 17=x/5 H 2 O: 2 -x-2(1 -x) = x x = 5(0. 17) = 0. 87 mol Total = 5 mol product Petroleum and Chemical Engineering Dept. Sultan Qaboos University Thermodynamics I 4 -28
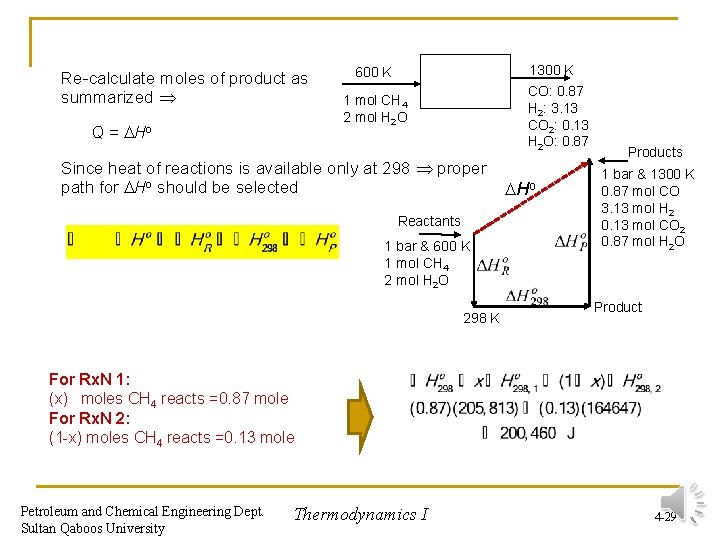
Re-calculate moles of product as summarized Q = Ho 1300 K 600 K CO: 0. 87 H 2: 3. 13 CO 2: 0. 13 H 2 O: 0. 87 1 mol CH 4 2 mol H 2 O Since heat of reactions is available only at 298 proper path for Ho should be selected Reactants 1 bar & 600 K 1 mol CH 4 2 mol H 2 O 298 K Ho Products 1 bar & 1300 K 0. 87 mol CO 3. 13 mol H 2 0. 13 mol CO 2 0. 87 mol H 2 O Product For Rx. N 1: (x) moles CH 4 reacts =0. 87 mole For Rx. N 2: (1 -x) moles CH 4 reacts =0. 13 mole Petroleum and Chemical Engineering Dept. Sultan Qaboos University Thermodynamics I 4 -29

i ni, R ni, P A B CH 4 1. 0 0. 0 1. 702 9. 081 H 2 O 2. 0 0. 87 3. 470 1. 450 CO 0. 0 0. 87 0. 337 0. 557 H 2 0. 0 3. 13 3. 250 0. 422 CO 2 0. 0 0. 13 5. 450 1. 045 Ho = Q = -34390 + 200460 + 161940 = 328 k. J Petroleum and Chemical Engineering Dept. Sultan Qaboos University Thermodynamics I 4 -30

Home work n 4. 9, 4. 11, 4. 17, 4. 19, 4. 21 (a, f), 4. 32 Petroleum and Chemical Engineering Dept. Sultan Qaboos University Thermodynamics I 4 -31
- Slides: 31