Heat Chapter 8 Heat Lesson 1 1 Understand























- Slides: 51

Heat Chapter 8 Heat

Lesson 1 1) Understand that heat is a form of energy and that it can be converted into other forms of energy. 2) Investigate and describe the expansion of solids, liquids and gases when heated, and contraction when cooled. 3) Demonstrate the expansion of water on freezing.

Heat is a form of energy • • • What is energy? Energy is the ability to do work Heat is a form of energy Heat has the ability to do work E. G. It helps flowers to grow

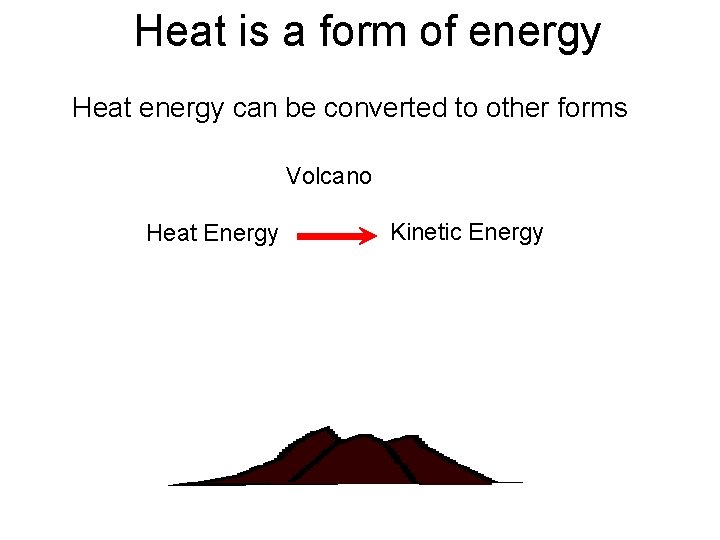
Heat is a form of energy • Heat can not be created or destroyed it can only be converted from one form to another • e. g. A runner: His kinetic (moving) energy is converted into heat energy as he gets hot and starts to perspire.

Heat is a form of energy Heat energy can be converted to other forms Volcano Heat Energy Kinetic Energy

Expansion Solids, Liquids and gases all expand when heated. On cooling they contract or get smaller

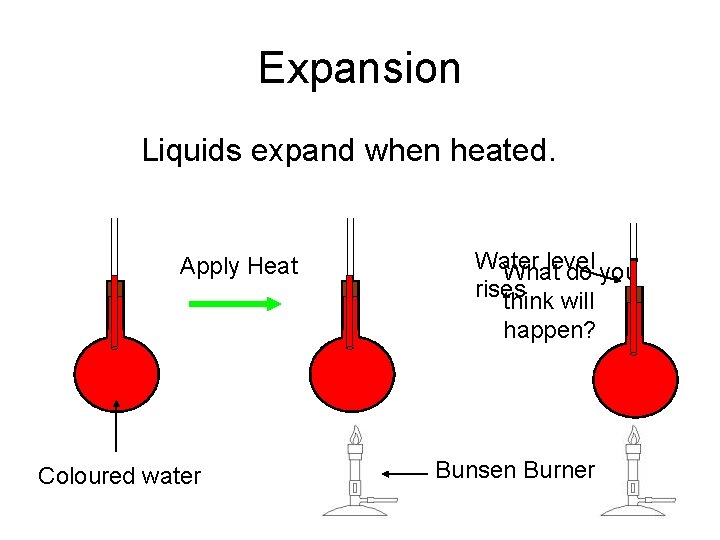
Expansion Liquids expand when heated. Apply Heat Water level What do you rises think will happen? Coloured water Bunsen Burner

Expansion of Liquids • A practical use of expansion of liquids is thermometers. • Examples include mercury and alcohol thermometers. • When placed in something hot the liquid rises up the Mercury tube. Note rise Hot Liquid

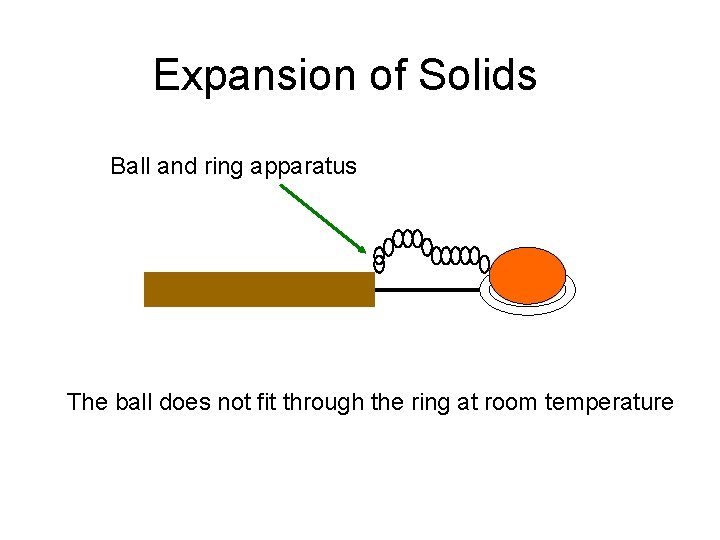
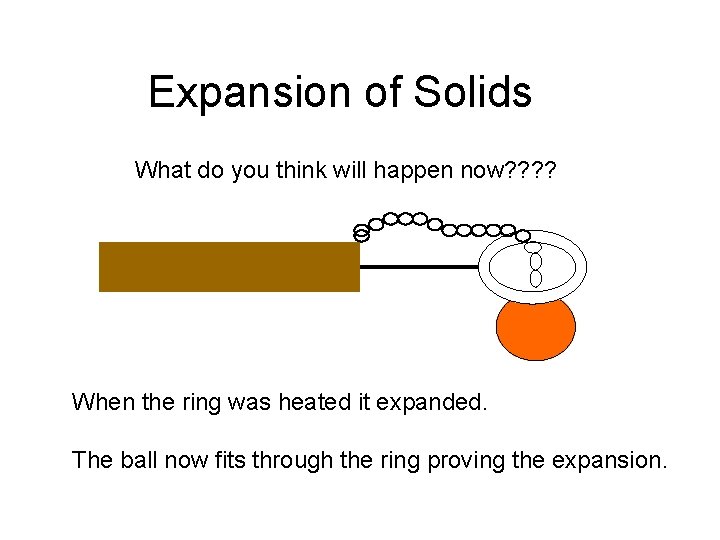
Expansion of Solids Ball and ring apparatus The ball does not fit through the ring at room temperature

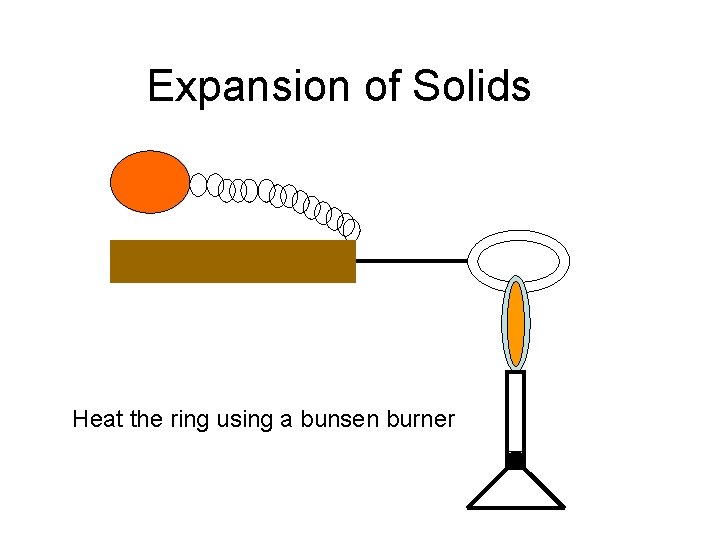
Expansion of Solids Heat the ring using a bunsen burner

Expansion of Solids What do you think will happen now? ? When the ring was heated it expanded. The ball now fits through the ring proving the expansion.

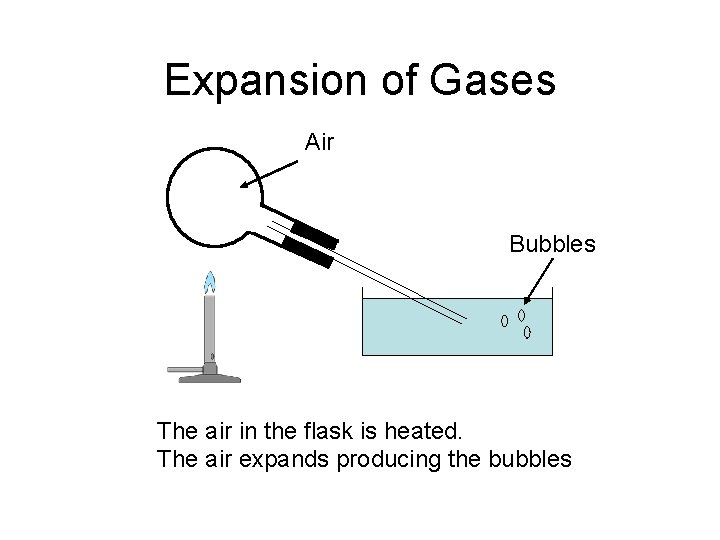
Expansion of Gases Air Bubbles The air in the flask is heated. The air expands producing the bubbles

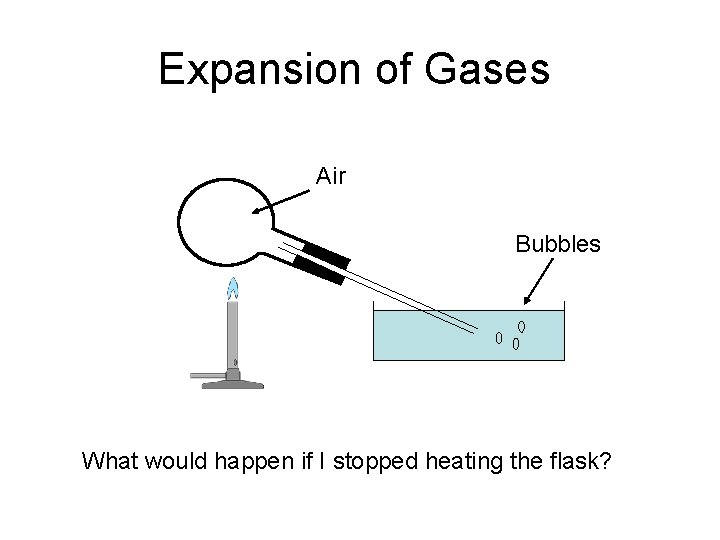
Expansion of Gases Air Bubbles What would happen if I stopped heating the flask?

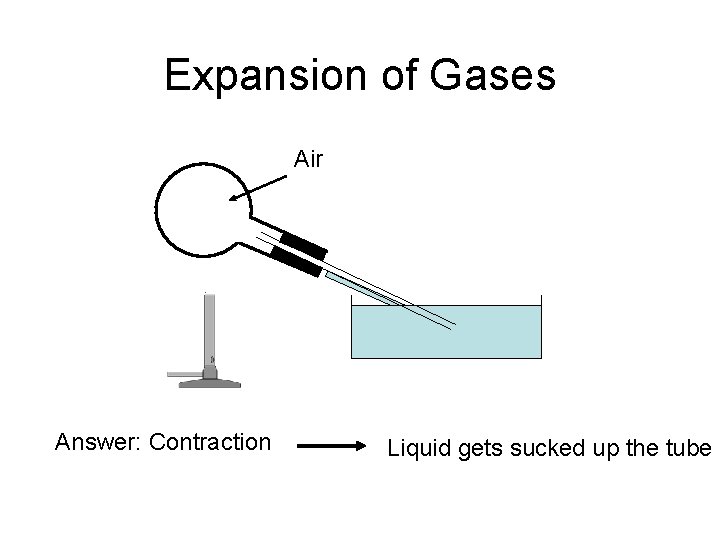
Expansion of Gases Air Answer: Contraction Liquid gets sucked up the tube

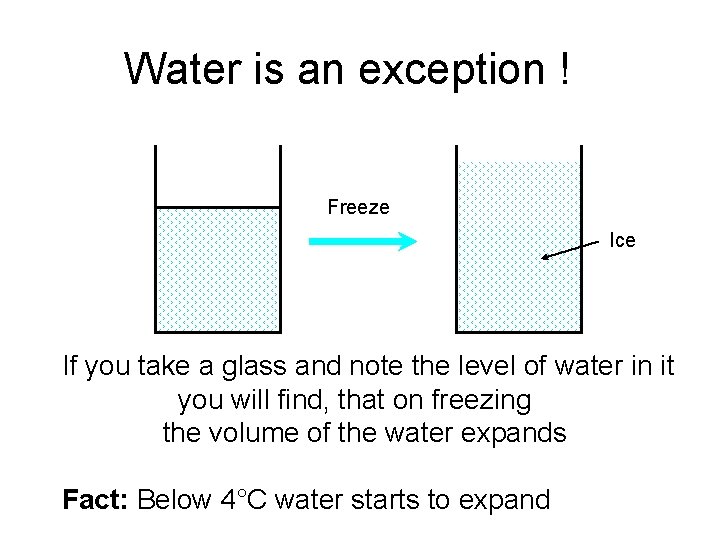
Water is an exception ! Freeze Ice If you take a glass and note the level of water in it you will find, that on freezing the volume of the water expands Fact: Below 4°C water starts to expand

Lesson 2 1) Explain the difference between heat and temperature. 2) Explain the 3 methods of heat transfer 3) Carry out simple experiments to show the transfer of heat energy by conduction, convection and radiation 4) Know that all hot bodies radiate heat.

Temperature and Heat Temperature == Heat is a form of energy and is measured in joules. Temperature is a measure of how hot something is. Thermometers are used to measure temperature. Normally thermometers contain alcohol or mercury. Liquid expands up the capillary tube when in contact with something hot

Heat Transfer There are 3 methods of heat transfer: 1) Conduction What are they? 2) Convection 3) Radiation

Conduction

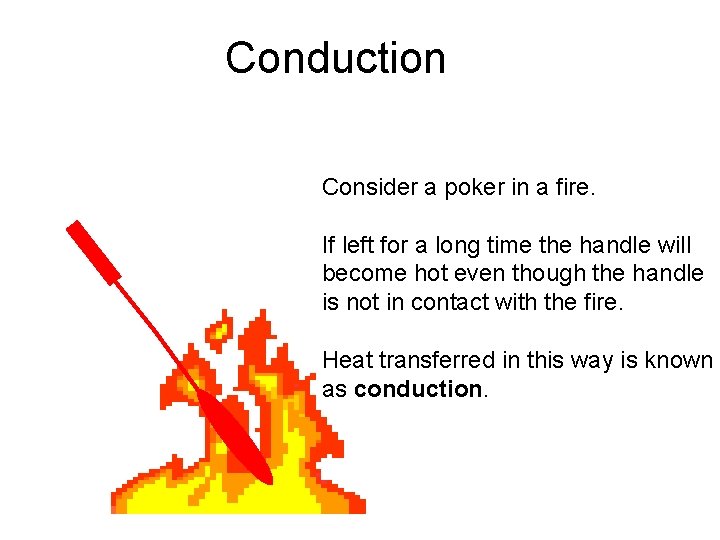
Conduction Consider a poker in a fire. If left for a long time the handle will become hot even though the handle is not in contact with the fire. Heat transferred in this way is known as conduction.

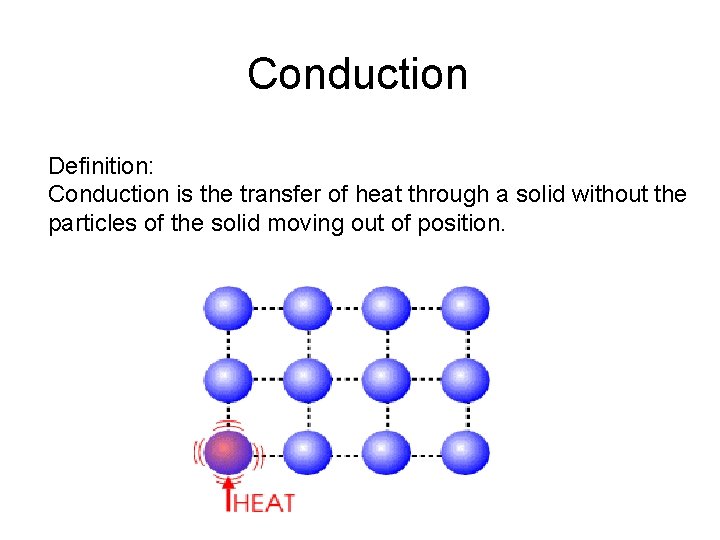
Conduction Definition: Conduction is the transfer of heat through a solid without the particles of the solid moving out of position.

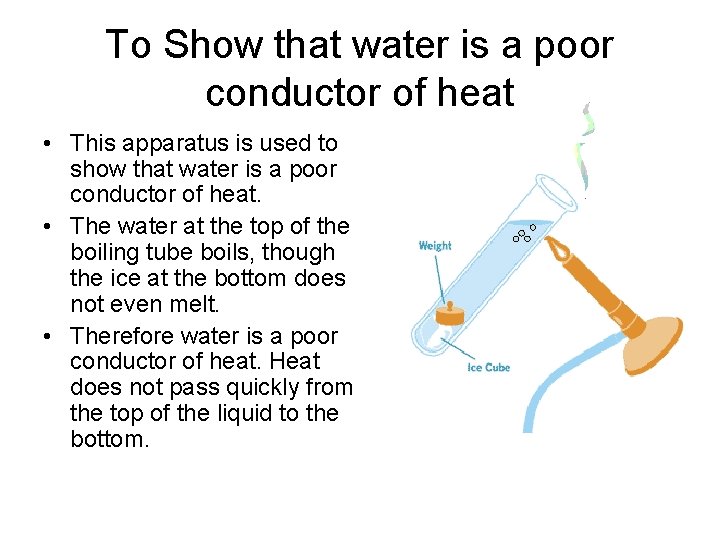
To Show that water is a poor conductor of heat • This apparatus is used to show that water is a poor conductor of heat. • The water at the top of the boiling tube boils, though the ice at the bottom does not even melt. • Therefore water is a poor conductor of heat. Heat does not pass quickly from the top of the liquid to the bottom.

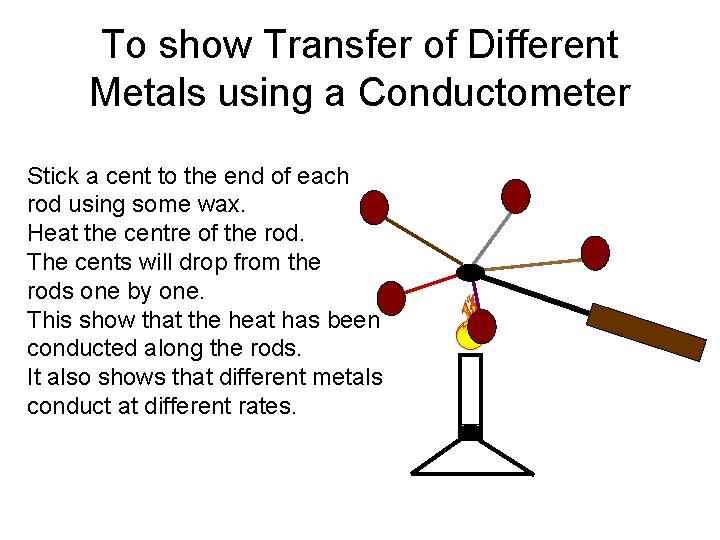
Conductometer • A conductometer is an instrument consisting of a heat proof handle • Together with 5 metal rods made of aluminium, brass, copper, nickel and steel. • The metal rods are of equal length and cross sectional area.

To show Transfer of Different Metals using a Conductometer Stick a cent to the end of each rod using some wax. Heat the centre of the rod. The cents will drop from the rods one by one. This show that the heat has been conducted along the rods. It also shows that different metals conduct at different rates.

Convection

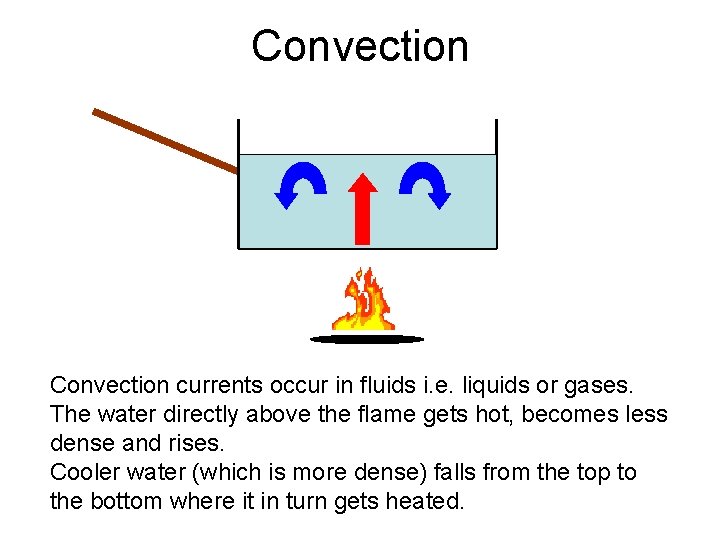
Convection currents occur in fluids i. e. liquids or gases. The water directly above the flame gets hot, becomes less dense and rises. Cooler water (which is more dense) falls from the top to the bottom where it in turn gets heated.

Convection Definition Convection is the transfer of heat through a liquid or gas when the particles of the liquid or gas move and carry the heat with them. A kettle of water boils using convection currents.

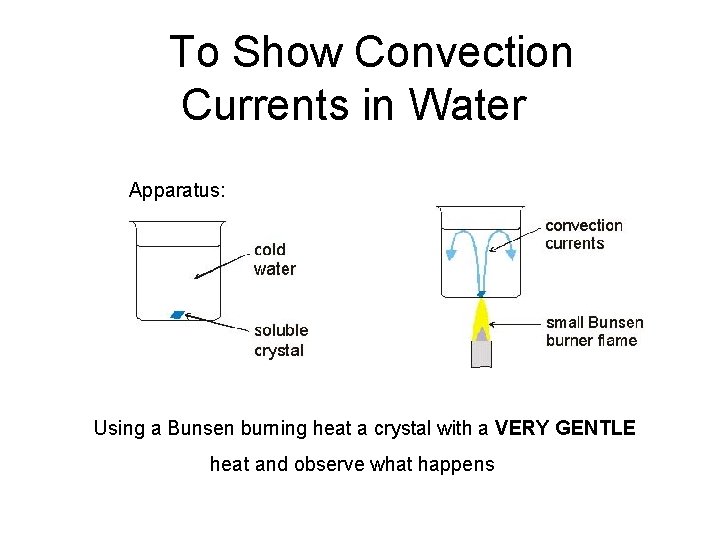
To Show Convection Currents in Water Apparatus: Using a Bunsen burning heat a crystal with a VERY GENTLE heat and observe what happens

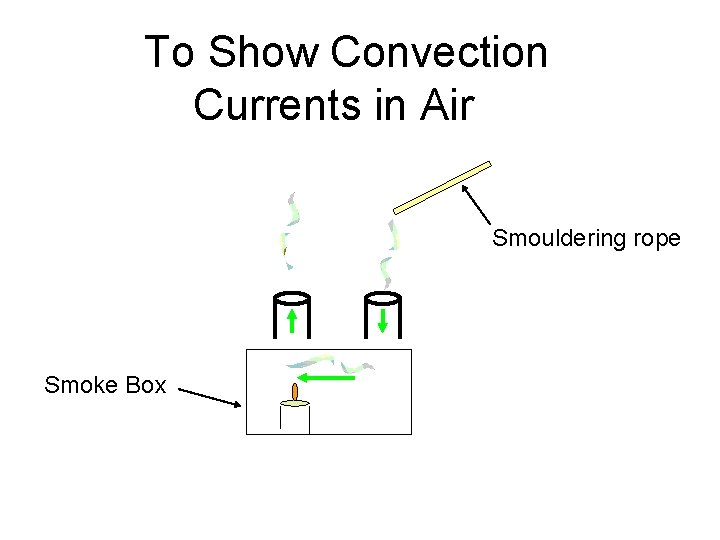
To Show Convection Currents in Air Smouldering rope Smoke Box

Radiation

Radiation Consider a grill cooking sausages. Air is a very poor conductor. In convection, heated particles rise upwards not downwards. So how do the sausages receive heat? Radiation!!!

Radiation Definition Radiation is the transfer of heat from a hot object without the need for a medium. The earth is heated by radiation rays from the sun. Radiation

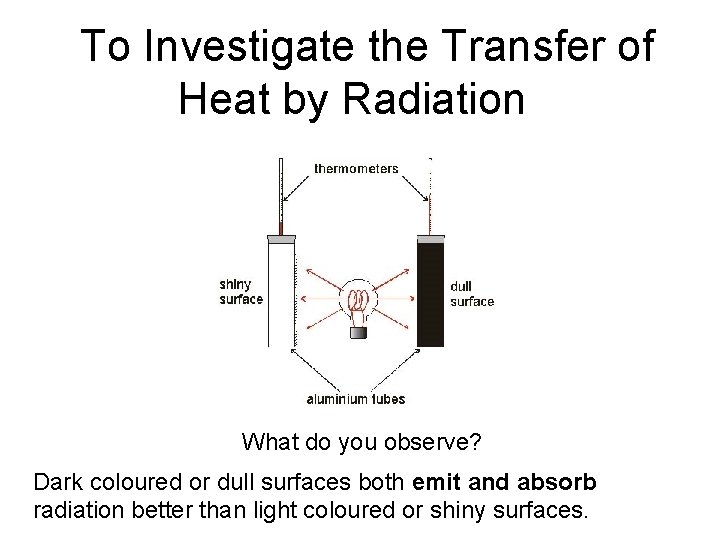
To Investigate the Transfer of Heat by Radiation What do you observe? Dark coloured or dull surfaces both emit and absorb radiation better than light coloured or shiny surfaces.

Radiation All hot bodies radiate heat.

Lesson 3 1) Identify good and bad conductors/insulators of heat. 2) Understand the changes of state 3) Explain latent heat. 4) Plot a heat curve and explain the shape of the curve in terms of latent heat 5) Investigate the effect of pressure on the boiling point of water

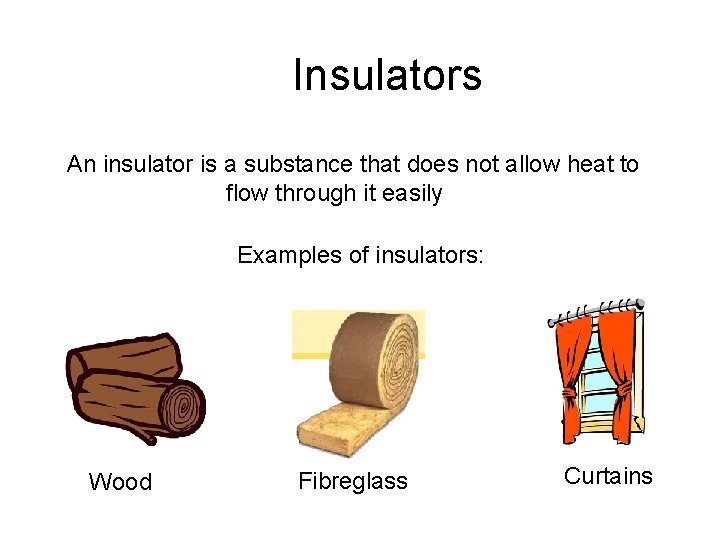
Insulators An insulator is a substance that does not allow heat to flow through it easily Examples of insulators: Wood Fibreglass Curtains

Conductors A conductor is a substance that allows heat to flow through it easily. Examples include: Gold Silver

Changes of State There are 3 states of matter: Solids Liquids Gas Solid Liquid Gas

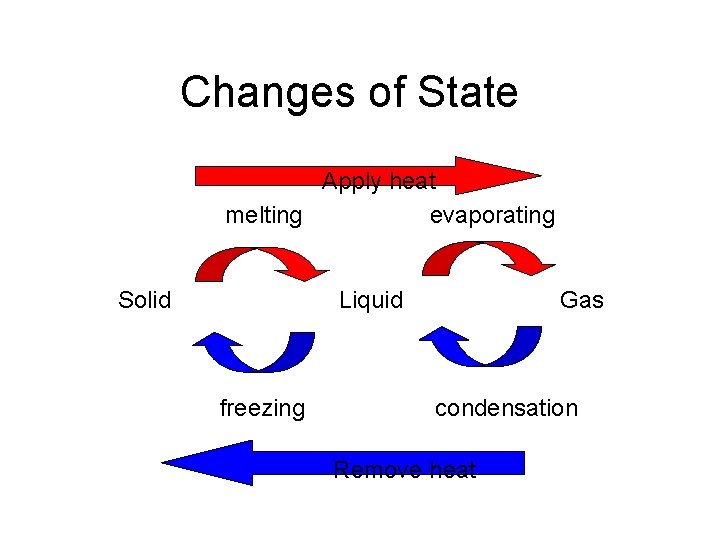
Changes of State Apply heat melting evaporating Solid Liquid freezing Gas condensation Remove heat

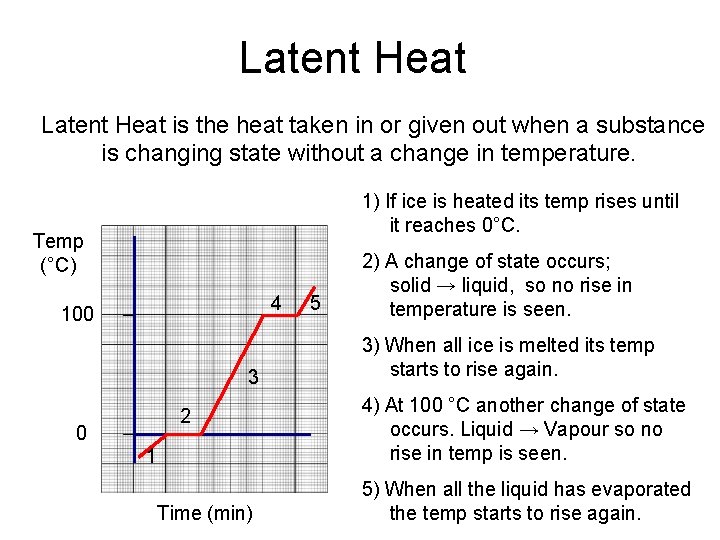
Latent Heat is the heat taken in or given out when a substance is changing state without a change in temperature. 1) If ice is heated its temp rises until it reaches 0°C. Temp (°C) 4 100 3 2 0 1 Time (min) 5 2) A change of state occurs; solid → liquid, so no rise in temperature is seen. 3) When all ice is melted its temp starts to rise again. 4) At 100 °C another change of state occurs. Liquid → Vapour so no rise in temp is seen. 5) When all the liquid has evaporated the temp starts to rise again.

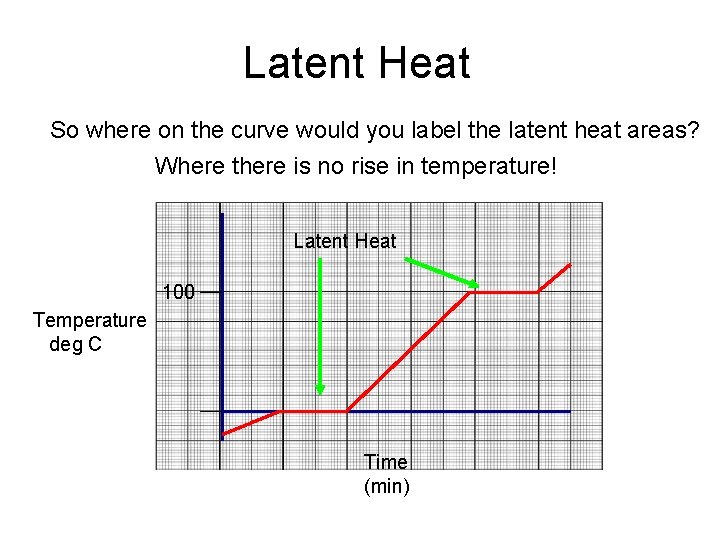
Latent Heat So where on the curve would you label the latent heat areas? Where there is no rise in temperature! Latent Heat 100 Temperature deg C Time (min)

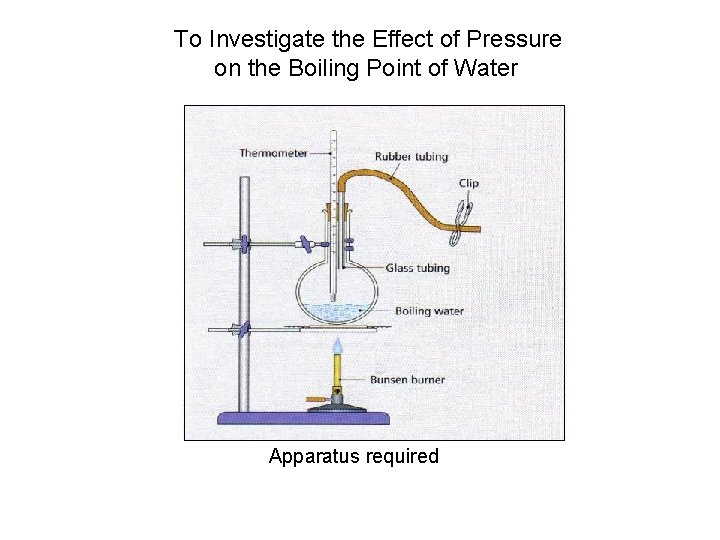
To Investigate the Effect of Pressure on the Boiling Point of Water Apparatus required

Assessment Section A Q 1 Heat is a form of _______. Q 2 The units of heat are ____. Q 3 Solids, liquids and gases _______ when heated and ____ when cooled. Q 4 Water is an exception to this rule as it starts to ______ below 4 °C. Q 5 Define temperature. What are its units? Q 6 On what principle are thermometers based?

Assessment Q 7 Conduction is the transfer of heat through a _______ without movement of the _____. Q 8 Convection is the transfer of _______ through a _____ Q 9 or a ____. What is Radiation? Q 10 A dull black surface _______ heat better than a bright shiny surface. Q 11 All hot bodies _____ heat. Q 12 What is an insulator? Name 2 examples.

Assessment Q 13 What is a conductor? Name 2 examples. Q 14 List the three states of matter. Q 15 Latent heat is the heat ______ in or given out when a substance is changing ______. Q 16 When latent heat is taking place there is _______change in temperature. Q 17 Increasing the pressure on water _____ its boiling point.

Assessment Section B Q 1 What is the following instrument and what is it used for?

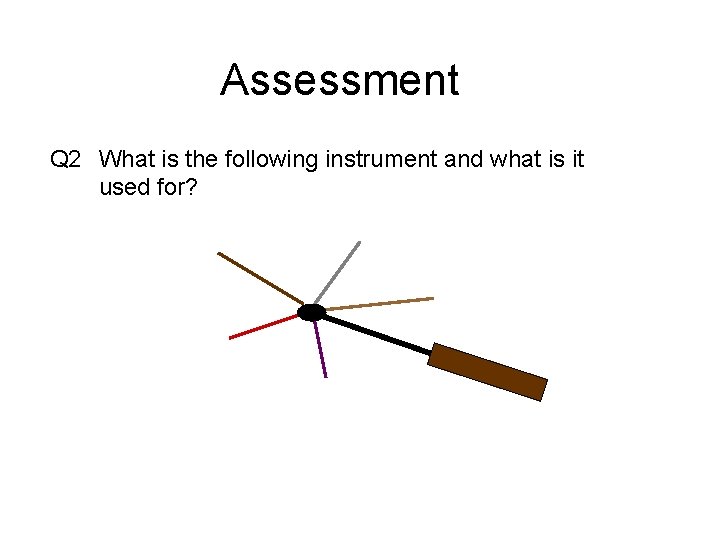
Assessment Q 2 What is the following instrument and what is it used for?

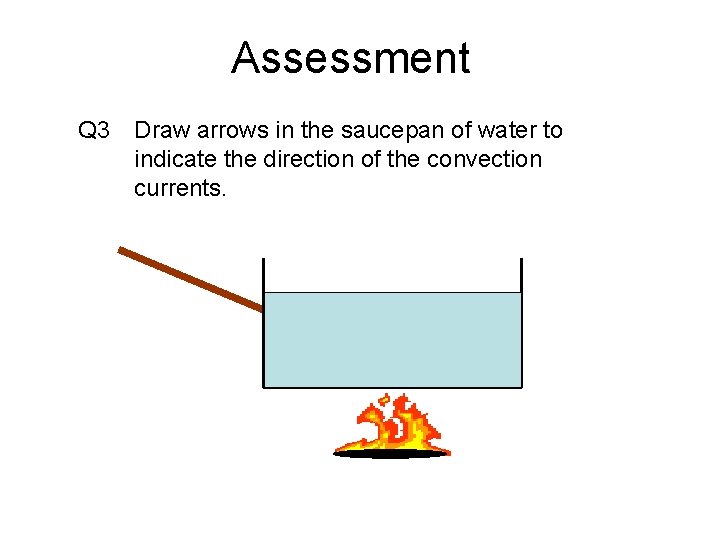
Assessment Q 3 Draw arrows in the saucepan of water to indicate the direction of the convection currents.

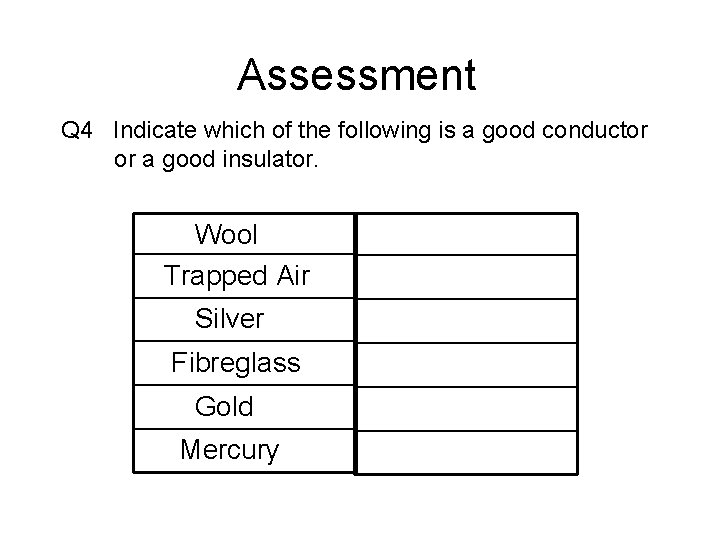
Assessment Q 4 Indicate which of the following is a good conductor or a good insulator. Wool Trapped Air Silver Fibreglass Gold Mercury

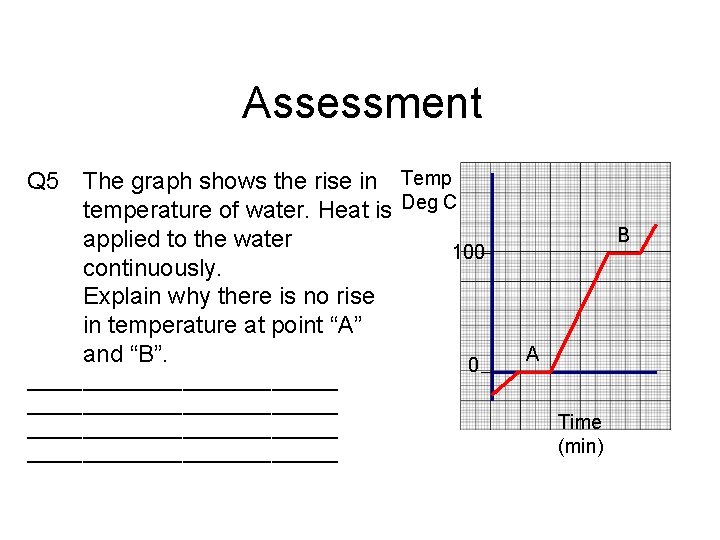
Assessment Q 5 The graph shows the rise in Temp temperature of water. Heat is Deg C applied to the water 100 continuously. Explain why there is no rise in temperature at point “A” and “B”. 0 ____________________________ B A Time (min)

Bibliography • http: //www. scienceatstmarys. net/VCl%20 KS 4%20 work /Energy/257 • http: //www. saburchill. com/physics/chapters/0107. html • www. juniorcertscience. ie • www. skool. ie • www. gcsescience. com • A voyage of discovery (Dorgan, Kennedy and Walsh)