Heat and Heat Transfer Scales of Measurement Celsius















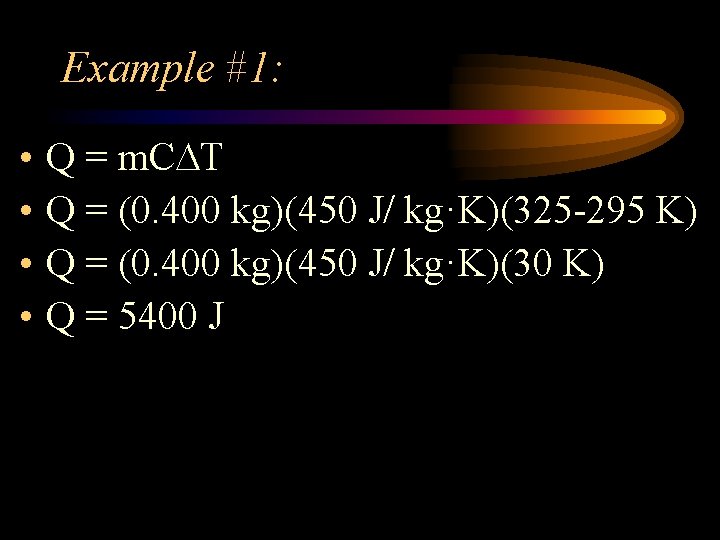


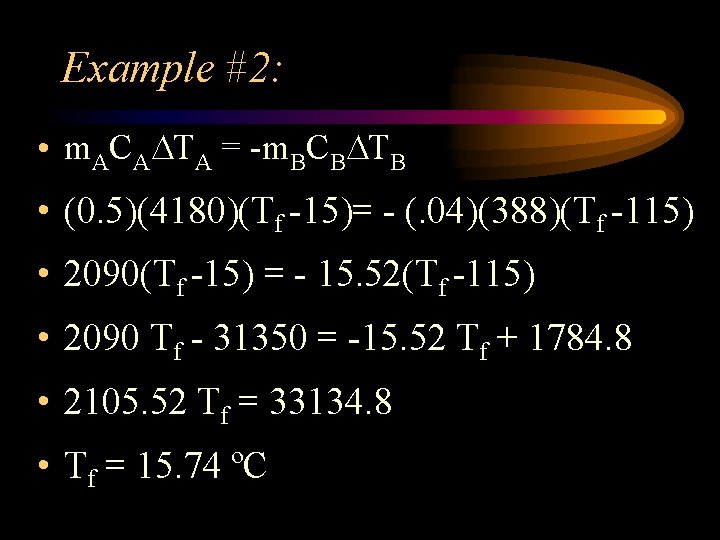
- Slides: 19

Heat and Heat Transfer

Scales of Measurement • Celsius scale – based on where water freezes (0 C) and where water boils (100 C) • Kelvin scale – based on the movement of particles

Absolute Zero • At 0 K, all particle movement has ceased • It is impossible to have a temperature lower than 0 K • 0 K = -273 C

• TC=5/9 (TF-32º) • TF= TC 9/5+32º • TK= TC+273. 15 0ºC 10ºC 20ºC 30ºC 40ºC 50ºC 273 K 283 K 293 K 303 K 313 K 323 K

Kinetic-Molecular Theory • The faster particles move, the greater the kinetic energy or thermal energy.

Definitions • Temperature –average kinetic energy of the particles in a substance – Thermometers measure temperature • Heat – amount of energy transferred

Thermal equilibrium • Energy will always travel from an area of higher energy to an area of lower energy.

Thermal Equilibrium, cont. • Two substances with different energies transfer energy (higher lower) until their energies are equal. • This point is “thermal equilibrium”.

Heat transfer • Conduction: molecular agitation; no motion as a whole • Convection: mass motion of a fluid • Radiation: emission of EM waves, no medium needed

Conduction • As materials are heated, electrons gain thermal energy which means they move faster. • As the electrons in a substance collide, the energy is transferred to surrounding electrons. • The actual molecules do not change places.

Convection • Heating occurs due to the motion of a fluid. • When a fluid is heated, it becomes less dense and rises. The cooler air is more dense and circulates to the bottom where it is heated and begins the process again.

Radiation • Radiation does not require a medium to transmit energy. This type of energy is called radiant energy and it travels in electromagnetic waves. • High temperatures emit short wavelengths whereas low temperatures emit long wavelengths.

Specific Heat • Amount of energy that must be added to the material to raise the temperature of a unit mass one temperature unit. • The units of specific heat are J/kg·K or J/kg·°C

Specific Heat Formula • Q = m. C T • Q = m. C (Tfinal – Tinitial) • • Q = Heat (J) m = mass (kg) C = Specific heat (J/kg·K or J/kg·°C) T = change in temperature (K or °C)

Example #1: • A 0. 400 kg block of iron is heated from 295 K to 325 K. How much heat had to be transferred to the iron if the specific heat of iron is 450 J/ kg·K?
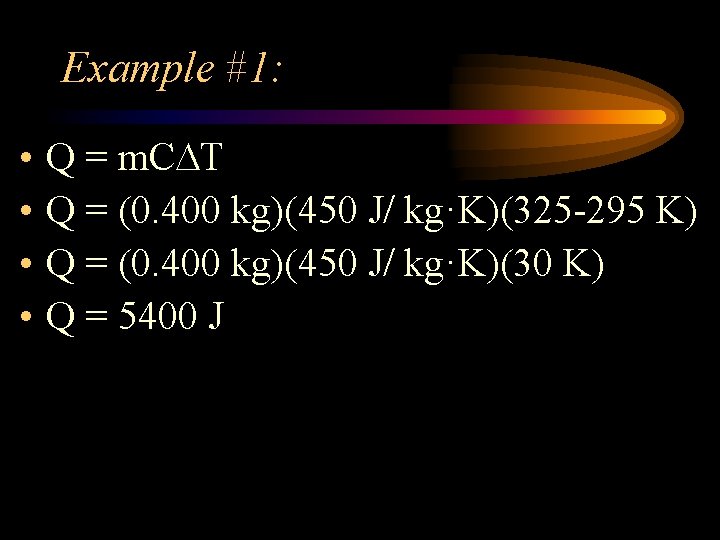
Example #1: • • Q = m. C T Q = (0. 400 kg)(450 J/ kg·K)(325 -295 K) Q = (0. 400 kg)(450 J/ kg·K)(30 K) Q = 5400 J

Law of Conservation of Energy • Energy lost by one object must be equal to the amount gained by another object. • Energy lost = - Energy gained • m. ACA TA = -m. BCB TB

Example #2: • A container has 0. 50 kg of water at 15 C. A 0. 040 kg block of zinc at 115 C is placed in the water. What is the final temperature of the system? (Czinc = 388 J/kg· C and Cwater = 4180 J/kg· C)
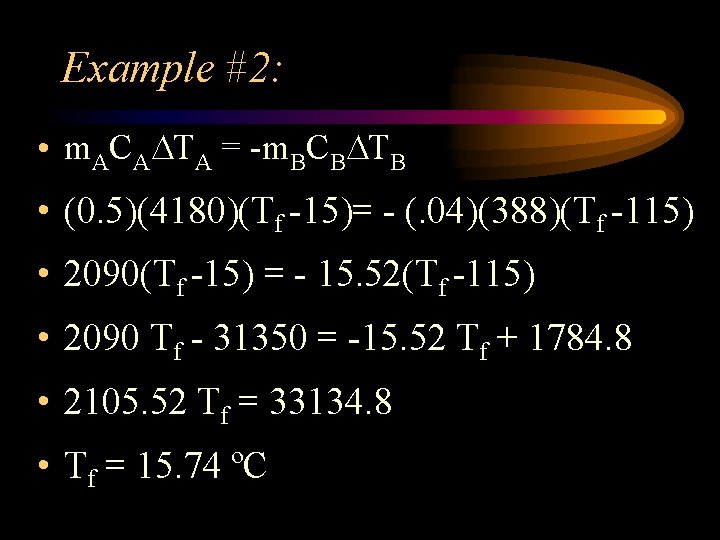
Example #2: • m. ACA TA = -m. BCB TB • (0. 5)(4180)(Tf -15)= - (. 04)(388)(Tf -115) • 2090(Tf -15) = - 15. 52(Tf -115) • 2090 Tf - 31350 = -15. 52 Tf + 1784. 8 • 2105. 52 Tf = 33134. 8 • Tf = 15. 74 ºC