Heart Failure Chronic and Decompensated Beatrice Wong Pharm

Heart Failure: Chronic and Decompensated Beatrice Wong, Pharm. D, BCPS November 15, 2011

Objectives �Review the epidemiology. �Describe the pathophysiology of heart failure. �Define the signs/symptoms of heart failure. �Compare the treatment of chronic vs decompensated states.

Definition �Complex clinical syndrome that can result from any structural or functional cardiac disorder that impairs the ability of the ventricle to fill with or eject blood.

Epidemiology �Approximately 5. 7 million patients have heart failure (HF) �(3. 1 million males, 2. 6 million females) � 670, 000 new cases annually �Hospital discharges: 990, 000 (2007) �Annual mortality: 22, 914 (men), 33, 651 (women) �Five-year mortality rate approximately 50%
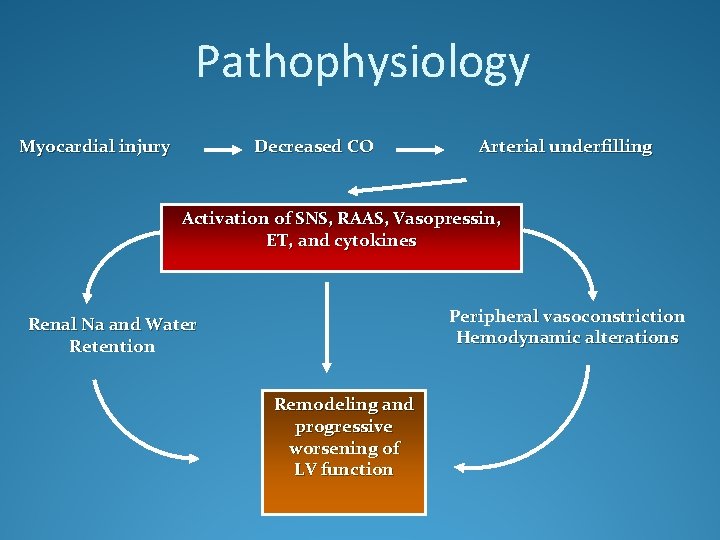
Pathophysiology Myocardial injury Decreased CO Arterial underfilling Activation of SNS, RAAS, Vasopressin, ET, and cytokines Peripheral vasoconstriction Hemodynamic alterations Renal Na and Water Retention Remodeling and progressive worsening of LV function
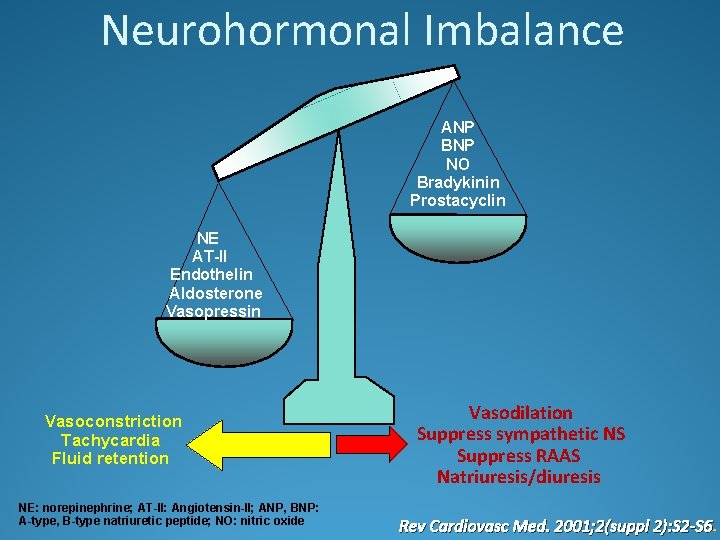
Neurohormonal Imbalance ANP BNP NO Bradykinin Prostacyclin NE AT-II Endothelin Aldosterone Vasopressin Vasoconstriction Tachycardia Fluid retention NE: norepinephrine; AT-II: Angiotensin-II; ANP, BNP: A-type, B-type natriuretic peptide; NO: nitric oxide Vasodilation Suppress sympathetic NS Suppress RAAS Natriuresis/diuresis Rev Cardiovasc Med. 2001; 2(suppl 2): S 2 -S 6

Causes �Hypertension �Coronary artery disease �Valvular heart disease �Cardiomyopathy �Alcohol �Drugs �Infectious diseases �Dilated vs Hypertrophic �Constrictive Pericarditis vs Restrictive
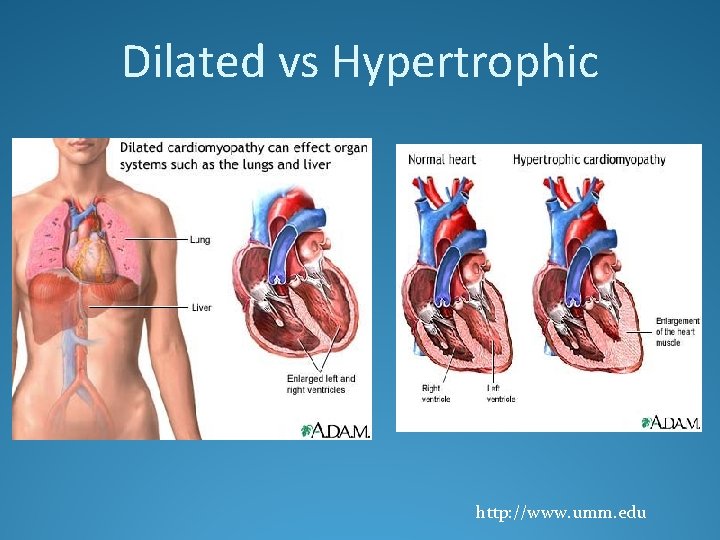
Dilated vs Hypertrophic http: //www. umm. edu

Systolic Dysfunction �Caused by a decrease in the normal contractile function of the ventricle �Characterized by a decrease in ejection fraction �Ejection fraction: the percentage of left ventricular end diastolic volume expelled during systole (normal 6070%) �The left ventricular cavity is often dilated
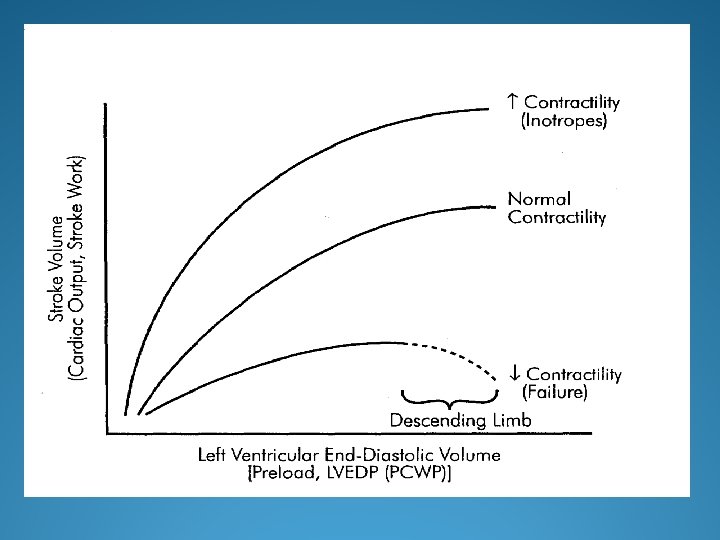

Diastolic Dysfunction �Caused by a failure of diastolic filling of the ventricle �Contractile function and ejection fraction are usually normal �The ventricular cavity can be thickened (hypertrophied)

Signs/Symptoms Left-sided �Pulmonary edema �Decreased perfusion to vital organs �Kidneys �GI tract �Brain Right-sided �Peripheral edema �Hepatic congestion

Functional Classification �New York Heart Association (NYHA) �Class I: Cardiac disease without resulting limitations of physical activity �Class II: Slight limitation of physical activitycomfortable at rest, but ordinary physical activity results in fatigue, palpitation, dyspnea, or anginal pain �Class III: Marked limitation in physical activitycomfortable at rest, but less than ordinary physical activity causes fatigue, palpitation, dyspnea, or anginal pain �Class IV: Inability to carry on any physical activity without discomfort or symptoms at rest

Grading of Heart Failure �ACC/AHA Classification �Class A: Risk for heart failure, no structural heart disease �Class B: Structural heart disease, no symptoms �Class C: Structural heart disease with symptoms �Class D: Refractory heart failure requiring special interventions

Monitoring �Echocardiography �Determine the ejection fraction �Evaluate for some causes of HF � Valvular � Tamponade �Angiography �Determine if coronary artery disease is present
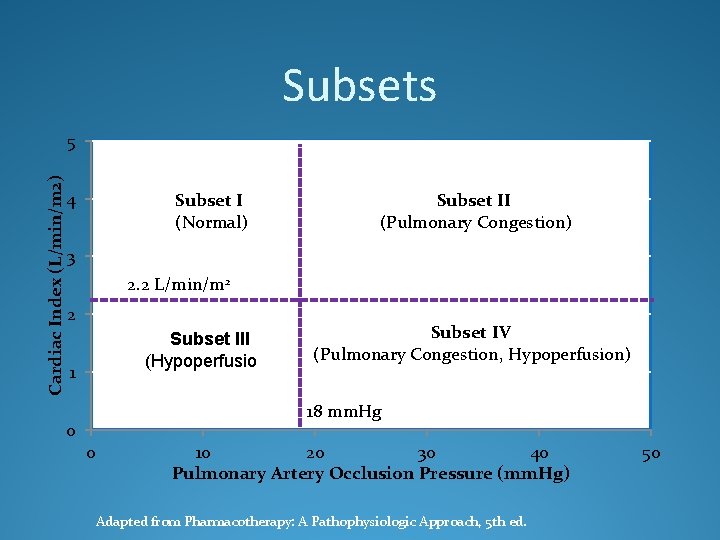
Subsets Cardiac Index (L/min/m 2) 5 4 Subset I (Normal) Subset II (Pulmonary Congestion) 3 2. 2 L/min/m 2 2 Subset III (Hypoperfusion) 1 0 Subset IV (Pulmonary Congestion, Hypoperfusion) 18 mm. Hg 0 10 20 30 40 Pulmonary Artery Occlusion Pressure (mm. Hg) Adapted from Pharmacotherapy: A Pathophysiologic Approach, 5 th ed. 50

Hemodynamic Variables Variable Equation Normal value Cardiac Output/Index CO = HR x SV Directly measured CO: 4 -7 L/min CI: 2. 5 -4. 2 L/min Pulmonary capillary wedge pressure (PCWP) Directly measured 5 -12 mm Hg Pulmonary artery pressure (PAS/PAD) Directly measured 20 -30/8 -12 mm Hg Systemic vascular resistance (SVR) SVR = [(MAP-CVP) / CO] * 80 800 -1440 dynes*sec*cm-5 Central venous pressure (CVP) Directly measured 2 -6 mm Hg
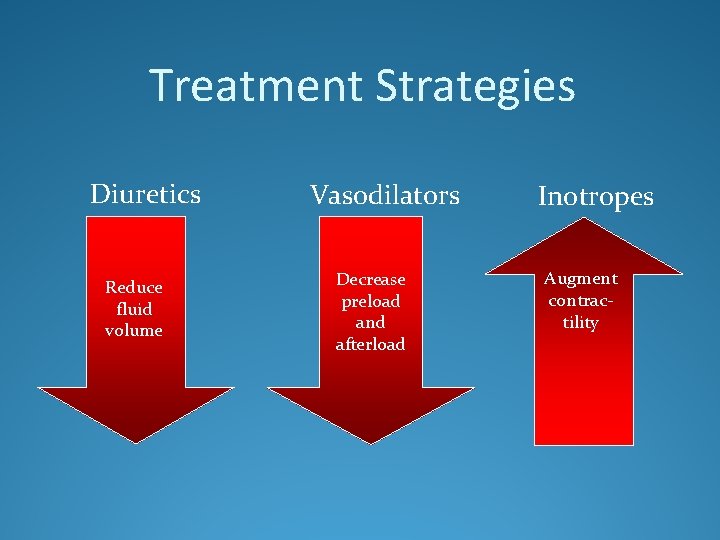
Treatment Strategies Diuretics Reduce fluid volume Vasodilators Decrease preload and afterload Inotropes Augment contractility

Acutely Decompensated Heart Failure
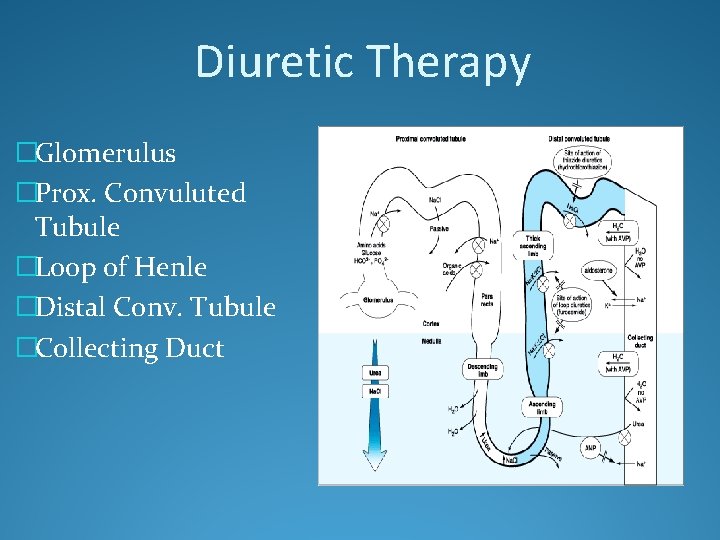
Diuretic Therapy �Glomerulus �Prox. Convuluted Tubule �Loop of Henle �Distal Conv. Tubule �Collecting Duct

Diuretics �Diuretics are frequently utilized to treat volume overload �Furosemide and other loop diuretics are first line in HF �Loop diuretics inhibit the reabsorption of Na, K, and Cl in the ascending loop of Henle �Lead to a reduction in intravascular volume and venous return to the heart �Reduce pulmonary congestion and peripheral edema
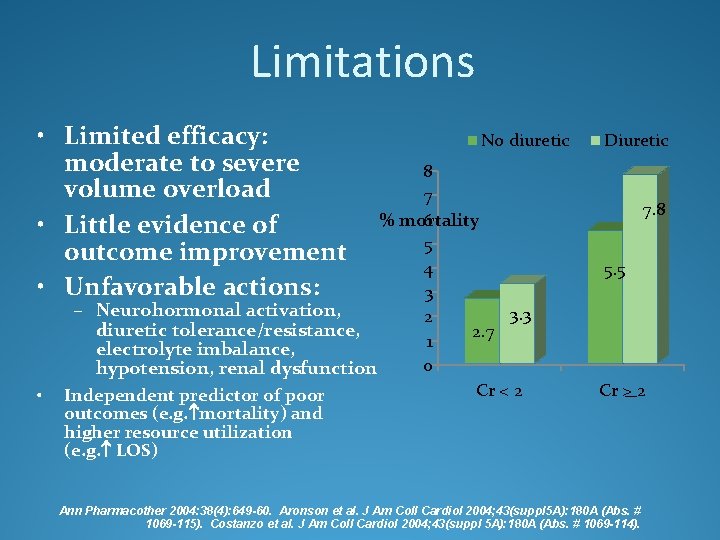
Limitations • Limited efficacy: moderate to severe volume overload • Little evidence of outcome improvement • Unfavorable actions: • No diuretic 8 7 % mortality 6 5 4 3 – Neurohormonal activation, 3. 3 2 diuretic tolerance/resistance, 2. 7 1 electrolyte imbalance, 0 hypotension, renal dysfunction Cr < 2 Independent predictor of poor outcomes (e. g. mortality) and higher resource utilization (e. g. LOS) Diuretic 7. 8 5. 5 Cr > 2 Ann Pharmacother 2004: 38(4): 649 -60. Aronson et al. J Am Coll Cardiol 2004; 43(suppl 5 A): 180 A (Abs. # 1069 -115). Costanzo et al. J Am Coll Cardiol 2004; 43(suppl 5 A): 180 A (Abs. # 1069 -114).

Potency of Diuretics �Furosemide (Lasix) � 40 -100 mg IV q 6 -24 h � Duration: 6 -8 hours �Torsemide (Demadex) � Better oral bioavailabilty � 20 mg torsemide = 40 mg lasix � Duration: 12 -16 hours �Bumetanide (Bumex) � 1 mg bumex = 40 mg lasix � Duration: 4 -6 hours

Adverse Effects • Electrolyte imbalances – Hypokalemia – Hypomagnesemia – Hypocalcemia • • Dehydration Glucose intolerance Precipitate gout Ototoxicity

Diuretic Resistance �Diuretic resistance is a complicating factor in HF that leads to increased admission rates and length of hospital stay �Pharmacokinetic and pharmacodynamic causes of diuretic resistance include: �Reduced active secretion into the tubular lumen (site of action) �Delayed/ reduced peak concentrations �Post diuretic sodium retention �Reduced natriuresis with chronic administration

Diuretic Resistance • Therapeutic strategies to overcome diuretic resistance: • Increase diuretic dose or switch drugs • Utilize intravenous administration • Salt Restricted Diet • Continuous infusion or multiple bolus dosing • Combination therapy with a thiazide diuretic • Chlorothiazide 250 -500 mg IV (30 min prior to loop) • Metolazone 2. 5 - 10 mg PO Ultrafiltration •

Vasodilators: Nitroglycerin �Reduce preload, slight effect on afterload �Dosing: � 0. 25 -2 mcg/kg/min �Adverse effects: �Hypotension �Headache �Tolerance

Vasodilators: Nitroprusside �Direct active arterial vasodilator �Decreases afterload �Dosing: � 0. 25 -5 mcg/kg/min (max 10 mcg/kg/min) �Half life 3 -5 minutes �Main uses: �Hypertension �CHF w/ high BP

Vasodilators: Nitroprusside �Considerations �Adverse effects: � Hypotension � Cyanide toxicity � Thiocyanate accumulation with long term, high doses, renal dysfunction � Pulmonary shunting � Reflex increase in HR

Inotrope: Milrinone �Phosphodiesterase inhibitor �Increases c. AMP levels, smooth muscle dilation, increased inotropy �Dosing � 0. 375 -0. 75 mcg/kg/min �Considerations �Long half-life (2 -4 hrs) �Renal elimination

Milrinone �More vasodilation than dobutamine �Doesn’t increase m. VO 2 �Can lower PA pressures �Thrombocytopenia less common than with amrinone �Arrhythmias, hypotension most common adverse effects

Milrinone: Best Use �Decompensated heart failure �Low cardiac index �High SVR �High PA pressures �Caution: long half life, renal elimination: longer hypotension if it occurs
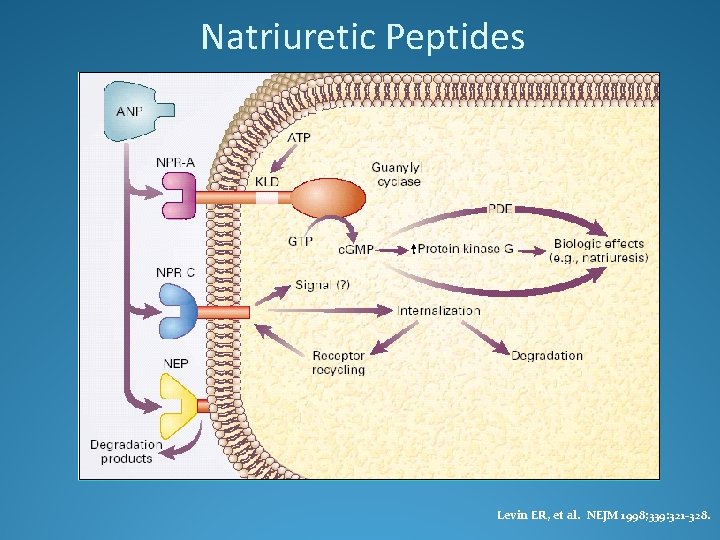
Natriuretic Peptides Levin ER, et al. NEJM 1998; 339: 321 -328.

Physiologic Effects of BNP �Hemodynamic: �Vasodilation of arteries, veins, and coronaries � Preload and Cardiac Output � Myocardial relaxation (lusitropy) �Neuroendocrine: �Counter-regulatory effect on RAAS, Aldosterone � SNS activity, Endothelin �Renal: � GFR, Diuresis and Natriuresis Levin ER et al, NEJM 1998; Venugopal J, Journal Clinical Pharmacy and Therapeutics 2001

Nesiritide �Nesiritide (recombinant BNP) is identical to endogenous BNP � 32 amino acid protein available as IV drug �Half-life is short – 18 minutes �Nesiritide does not have direct inotropic activity �Intravenous infusion of nesiritide studied in >1700 patients with acute decompensated CHF

Nesiritide �Patients with acute decompensated heart failure without cardiogenic shock (don’t use if SBP<90) �Use nesiritide with IV diuretics �Administer with an initial bolus dose (2 µg/kg) followed by a fixed-dose infusion (0. 01 µg/kg/min) �For greater hemodynamic effect, increase the infusion rate of nesiritide in increments of 0. 005 µg/kg/min every 3 hours up to a maximum of 0. 03 µg/kg/min �Do not need invasive hemodynamic monitoring

Nesiritide Controversy �Sold as a drug that “should be” initiated in all ADHF patients upon admission to the ED �Touted as “improving” renal function �“Suggestion” of a mortality benefit in ADHF �Outpatient infusions “encouraged” �“Natrecor Reimbursement and Billing Guide”

Panel Recommendations �Final Recommendations regarding nesiritide �Strictly limited to ADHF with dyspnea at rest, and physicians should assess the risks vs. benefit compared to alternative treatments �Nesiritide should not be used to replace diuretics and should not be used in the following manner � Intermittent outpatient infusion, scheduled repetitive use, to improve renal function, to enhance diuresis �Scios should undertake a pro-active educational program to inform physicians regarding appropriate use of nesiritide
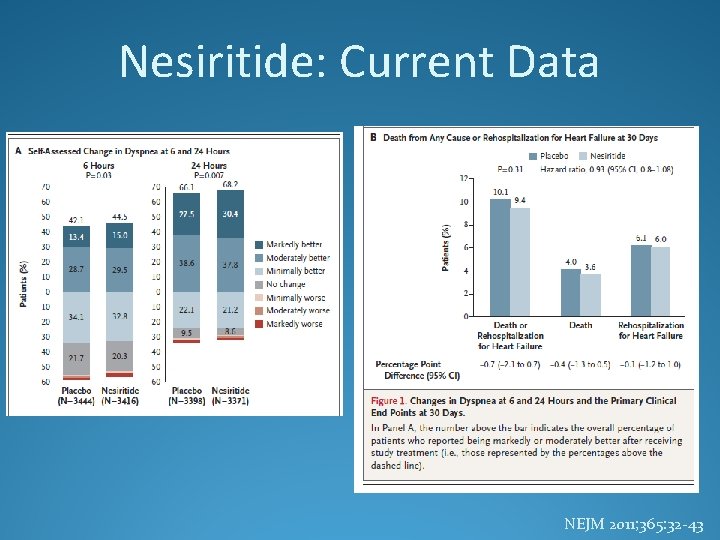
Nesiritide: Current Data NEJM 2011; 365: 32 -43

Catecholamines �Adrenergic Receptors �Dopaminergic receptors: renal/mesenteric vasodilation �Alpha-1: vasoconstriction �Beta-1: chronotropic, inotropic effects �Beta-2: bronchodilation, vasodilation

Dobutamine �Use: acute HF �Selective beta receptor agonist �Dosing � 3 -20 mcg/kg/min �Effects: increase CO, HR, +/- decrease in BP �Adverse effects �Tachyarrhythmias �Increased m. VO 2

Dobutamine: Best Use �Decompensated heart failure �Low cardiac index, normal or low SVR �Initiation should be in intensive care, titration with Swan-Ganz monitoring or Sv. O 2 �Telemetry for arrhythmia monitoring �Patients on home infusions need an AICD

Dopamine �Dosing � 0. 5 -5 mcg/kg/min: dopaminergic receptors � 5 -10 mcg/kg/min: beta effects: increase HR, CO �>10 mcg/kg/min: alpha effects: increase BP �Considerations �Adverse effects �“Renal” dopamine

Epinephrine �Use: cardiac failure with hypotension �Dosing � 0. 02 -0. 2 mcg/kg/min �Effects �Beta effects (HR, CO) at lower dose range �Alpha effects (BP) at higher dosages �Considerations �Useful mix of effects, ? less tachycardia than dopamine, metabolic effects

Norepinephrine �Use: cardiac failure with severe hypotension �Dosing � 0. 05 -0. 2 mcg/kg/min �Effects �Less potent than epinephrine at beta receptors; alpha effects predominate �Considerations �Splanchnic perfusion �Adverse effects
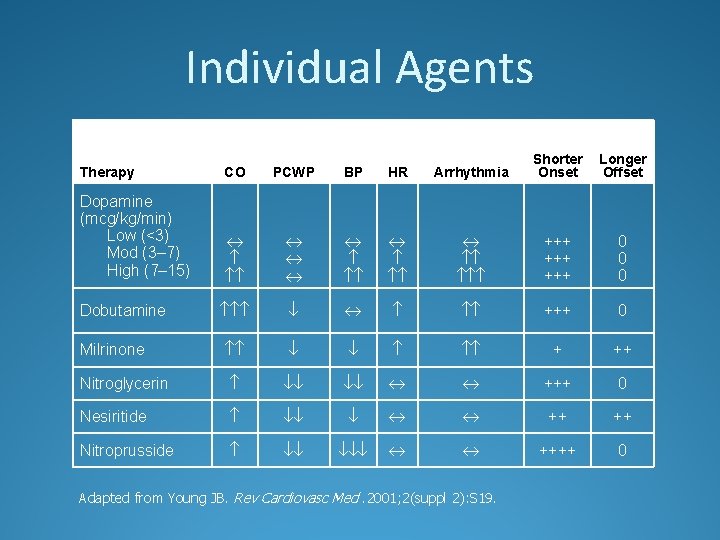
Individual Agents Therapy CO PCWP BP HR Arrhythmia Shorter Onset Dopamine (mcg/kg/min) Low (<3) Mod (3– 7) High (7– 15) +++ +++ 0 0 0 +++ 0 + ++ Nitroglycerin +++ 0 Nesiritide ++ ++ Nitroprusside ++++ 0 Dobutamine Milrinone Adapted from Young JB. Rev Cardiovasc Med. 2001; 2(suppl 2): S 19. Longer Offset

Mechanical Devices �Intra-aortic balloon pump (IABP) �Ventricular Assist Devices (VAD)

Chronic Management
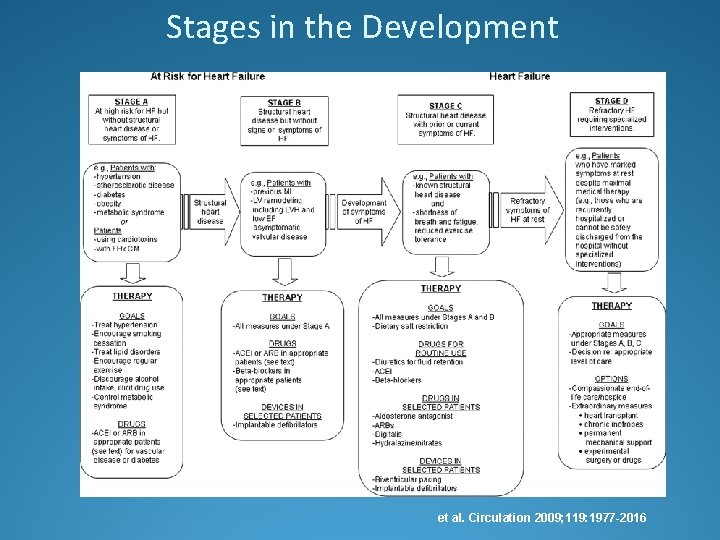
Stages in the Development et al. Circulation 2009; 119: 1977 -2016

Vasodilators �Angiotensin Converting Enzyme Inhibitors (ACE inhibitors) �Angiotensin II Receptor Blockers (ARBs)
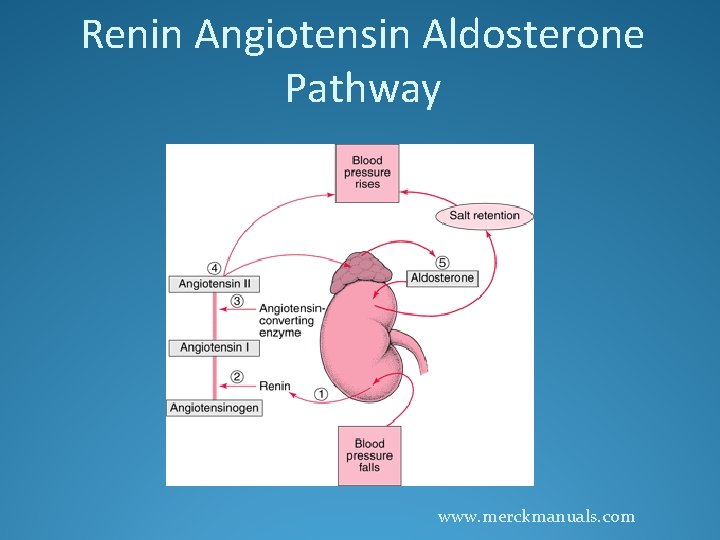
Renin Angiotensin Aldosterone Pathway www. merckmanuals. com
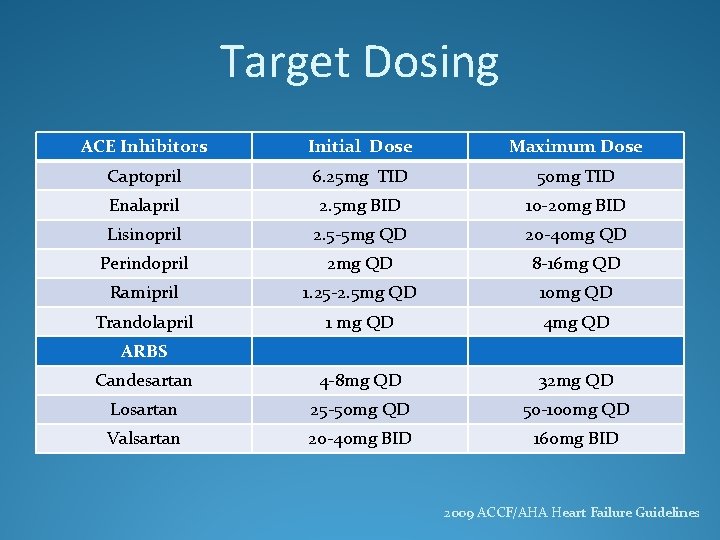
Target Dosing ACE Inhibitors Initial Dose Maximum Dose Captopril 6. 25 mg TID 50 mg TID Enalapril 2. 5 mg BID 10 -20 mg BID Lisinopril 2. 5 -5 mg QD 20 -40 mg QD Perindopril 2 mg QD 8 -16 mg QD Ramipril 1. 25 -2. 5 mg QD 10 mg QD Trandolapril 1 mg QD 4 mg QD Candesartan 4 -8 mg QD 32 mg QD Losartan 25 -50 mg QD 50 -100 mg QD Valsartan 20 -40 mg BID 160 mg BID ARBS 2009 ACCF/AHA Heart Failure Guidelines

Adverse Effects �Hyperkalemia �Increased Serum Creatinine �Angioedema �Cough (ACE inhibitors) �Hypotension
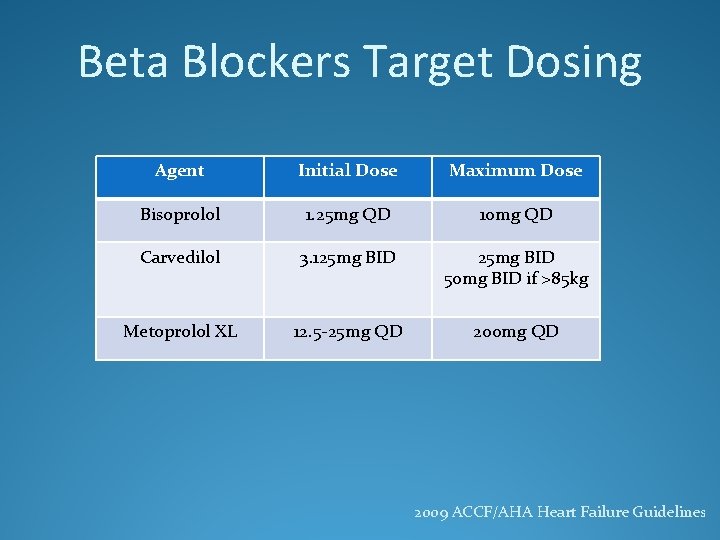
Beta Blockers Target Dosing Agent Initial Dose Maximum Dose Bisoprolol 1. 25 mg QD 10 mg QD Carvedilol 3. 125 mg BID 50 mg BID if >85 kg Metoprolol XL 12. 5 -25 mg QD 2009 ACCF/AHA Heart Failure Guidelines

Beta Blockers �Mechanism- Antagonist at beta-receptors �Metoprolol and Bisoprolol are β 1 selective �Carvedilol is an α and β blocker (nonselective) �Rationale: Inhibit adverse effects of Sympathetic nervous system �NE can induce cardiac hypertrophy, trigger apoptosis �Arrhythmias �Titrate slowly to avoid worsening heart failure symptoms and fluid retention �Adverse effects �Fatigue, bradycardia, heart block, hypotension

COMET �Randomized to Carvedilol or Metoprolol IR �n= 3029 patients with CHF NYHA Class II-IV on ACE inhibitors and diuretics. �Titrated to Carvedilol 25 mg BID or Metoprolol 50 mg BID. �Results: Mortality 34% (Car) vs 40% (Met), p = 0. 002 �Carvedilol had less cardiovascular deaths Lancet 2003; 362: 7 -13.

Hydralazine/Nitrates �Mechanism- Hydralazine dilates arterioles while Nitrates dilate veins �Dosing- Fixed combo of 37. 5 mg Hydr/20 mg ISDN TID, titrate to 75 mg Hydr/40 mg ISDN TID. Dosing may differ with separate products. �Adverse effects: �Hydralazine- Drug–induced lupus, headaches, GI �Isosorbide Dinitrate- headaches, need nitrate free period of 10 hours.
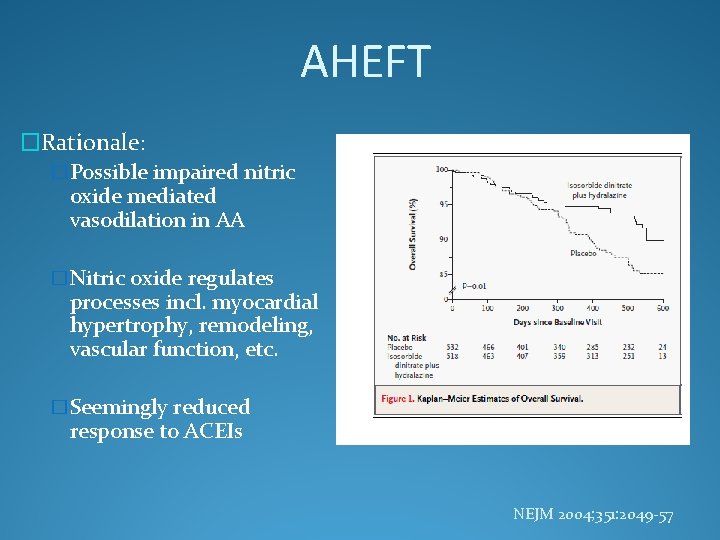
AHEFT �Rationale: �Possible impaired nitric oxide mediated vasodilation in AA �Nitric oxide regulates processes incl. myocardial hypertrophy, remodeling, vascular function, etc. �Seemingly reduced response to ACEIs NEJM 2004; 351: 2049 -57

Aldosterone Antagonists �Mechanism- Antagonist for Aldosterone �Aldosterone can cause myocardial and vascular fibrosis, direct vascular damage, baroreceptor dysfunction �Dosing �Spironolactone- target dose of 25 mg -50 mg per day �Eplerenone- target dose of 50 mg per day �Adverse effects: �Gynecomastia and menstrual disturbances (Spi >> Epler) �Hyperkalemia
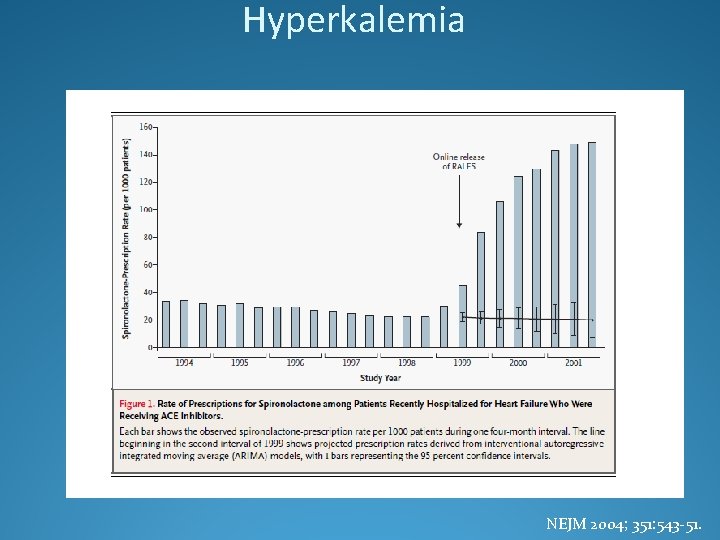
Hyperkalemia NEJM 2004; 351: 543 -51.
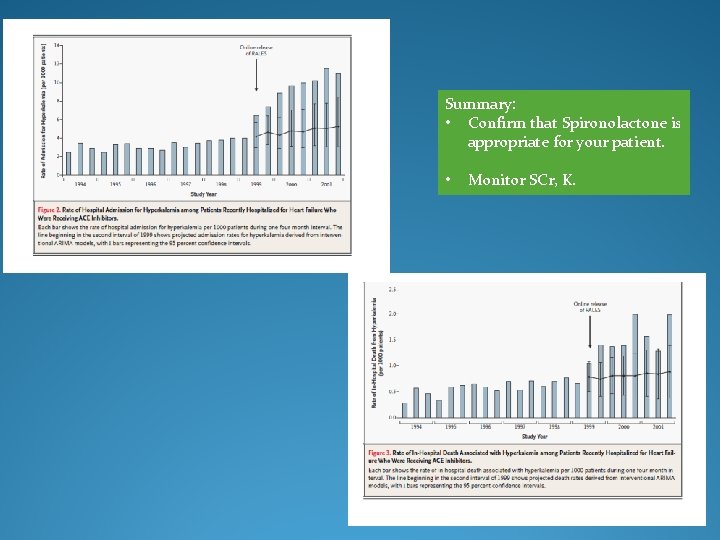
Summary: • Confirm that Spironolactone is appropriate for your patient. • Monitor SCr, K.

Guidelines for Minimizing Risk �Determine that GFR or Cr. Cl > 30 m. L/min �Do not administer if K > 5 meq/L �Initial dose of Spironolactone 12. 5 mg or Eplerenone 25 mg. �Risk of hyperkalemia is increased with concomitant use of higher dose of ACEIs. �NSAIDs or COX-2 inhibitors should be avoided. �Potassium supplements should be reduced or stopped. �Close monitoring: Check in 3 days, in 1 week, then at least monthly for first 3 months. �Diarrhea or other causes of dehydration should be addressed quickly. 2009 ACCF/AHA Heart Failure Guidelines

Digoxin �Mechanism- Inhibit Na/K ATPase �Positive inotrope �Reduces sympathetic outflow from CNS �Reduces renal tubular reabsorption of Na. �Dose- based on levels (0. 5 - 1 ng/m. L) � 0. 125 mg – 0. 25 mg daily for normal renal function �Renally eliminated �Drug interactions with PGP inhibitors: Amiodarone, Clarithromycin, Cyclosporine, Quinidine, Verapamil
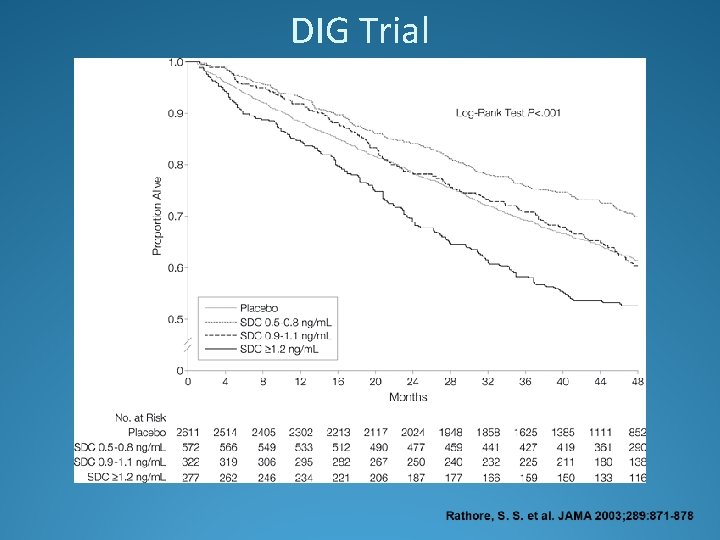
DIG Trial

Digoxin �Adverse effects �Heart block, etopic and re-entrant cardiac rhythms �GI: anorexia, nausea, vomiting �Neurological: Visual disturbances, confusion � Overt toxicity is commonly associated with levels >2 ng/m. L � Toxicity may occur at lower levels. � Risk factors: Hypokalemia, Hypomagnesemia

Questions
- Slides: 67