Healthy Liver Chronic Hepatitis B Hepatitis B Virus

Healthy Liver

Chronic Hepatitis B
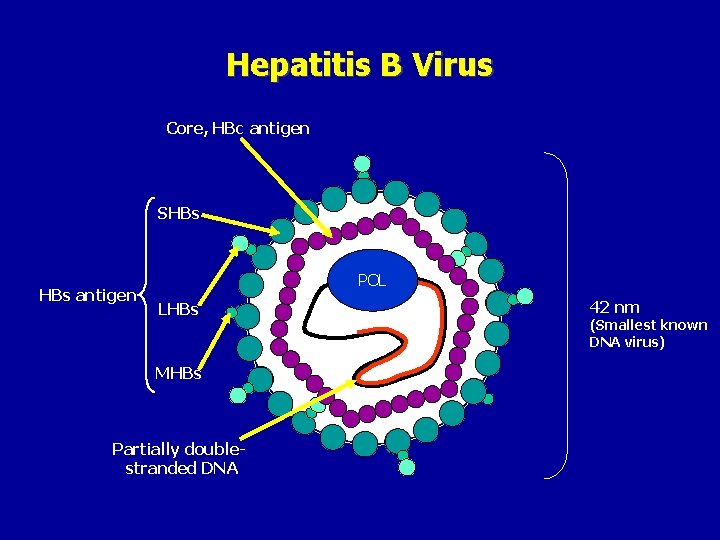
Hepatitis B Virus Core, HBc antigen SHBs antigen POL LHBs MHBs Partially doublestranded DNA 42 nm (Smallest known DNA virus)
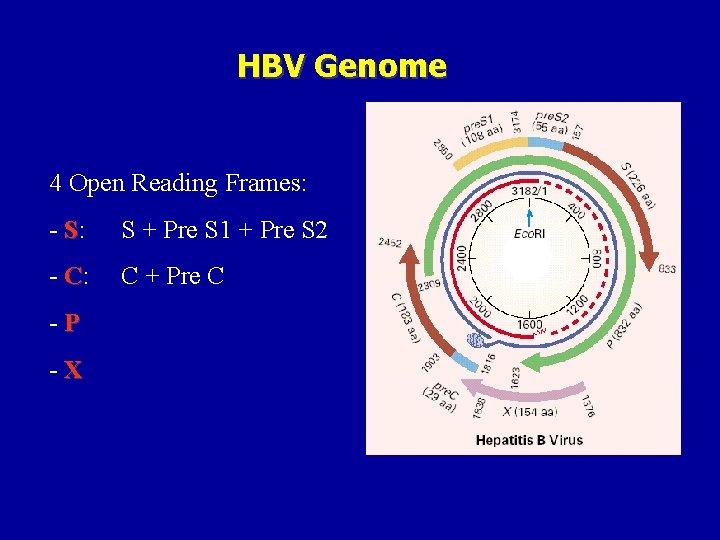
HBV Genome 4 Open Reading Frames: - S: S + Pre S 1 + Pre S 2 - C: C + Pre C -P -X
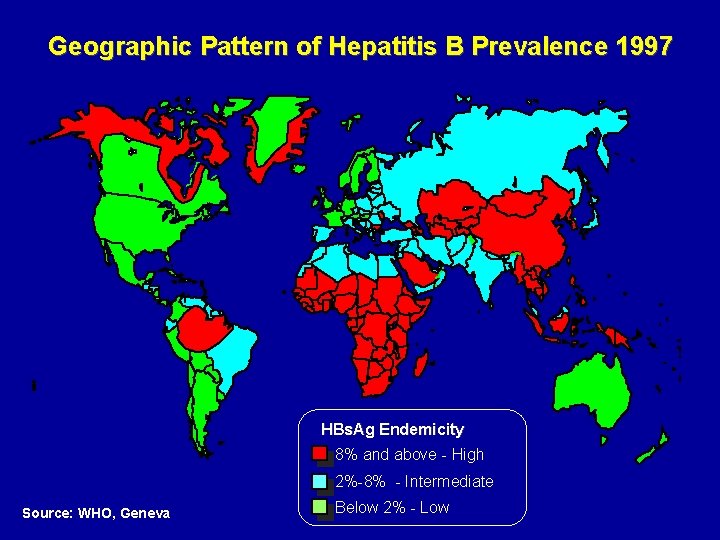
Geographic Pattern of Hepatitis B Prevalence 1997 HBs. Ag Endemicity 8% and above - High 2%-8% - Intermediate Source: WHO, Geneva Below 2% - Low

Prevalence of HBV in Syria • Intermediate HBV endemicity At least 4 % of chronic infection Statistics of Blood Transfusion Centers • Total Population of Syria: 18 866 000 (2002) at least 750 000 Syrian with chronic HBV infection

Mode of Transmission of HBV in Syria • • Infected blood transfusion or blood products Needle stick injuries: HCW - injection drug users Hemodialysis Sexual transmission: heterosexual - homosexual Horizontal transmission: childhood - family member Vertical Transmission (mother to newborn) Unsafe Procedures: ear piercing-tattooing -barbering.
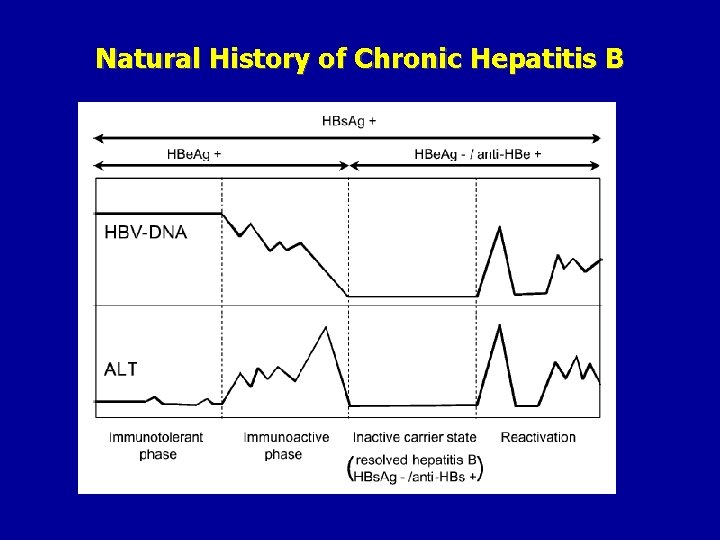
Natural History of Chronic Hepatitis B
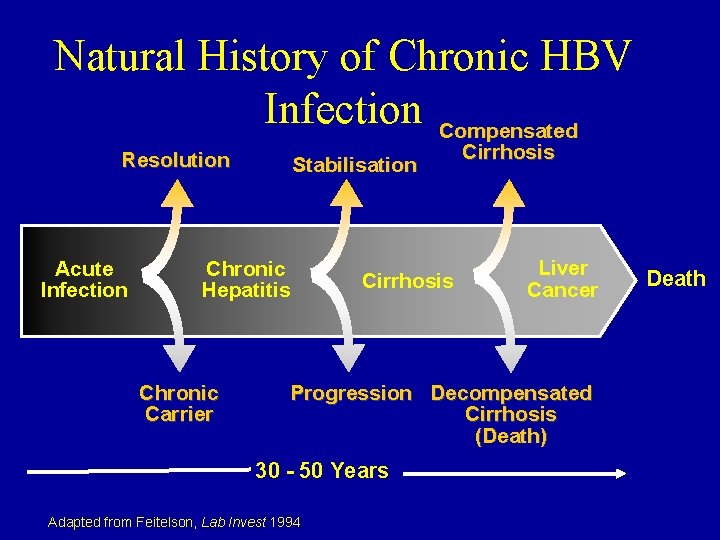
Natural History of Chronic HBV Infection Compensated Resolution Acute Infection Stabilisation Chronic Hepatitis Chronic Carrier Cirrhosis Liver Cancer Progression Decompensated Cirrhosis (Death) 30 - 50 Years Adapted from Feitelson, Lab Invest 1994 Death

Chronic Hepatitis B 1. HBs Ag + > 6 months 2. Serum HBV DNA >2. 000 or 20. 000 IU/m. L 3. Persistent or intermittent elevation in ALT/AST 4. Liver biopsy showing chronic hepatitis necroinflammatory score 4 *

Types of Chronic HBV Infections • Wild type HBe Ag + chronic hepatitis • Mutant type HBe Ag – chronic hepatitis More prevalent in Syria Up to 70 %

HBe Ag + & HBe Ag – Chronic Hepatitis HBe Ag + HBs Ag HBe Ag Anti-HBe HBV DNA (IU/m. L) (copie/m. L) ALT Necro-inflammation + + – > 20. 000 > 100000 Elevated + HBe Ag – + > 2. 000 >10000 Elevated +

Initial Evaluation of patients with chronic HBV 1. History & physical examination 2. Laboratory tests for liver disease CBC Hepatic panel PT 3. Tests for HBV replication: HBe. Ag /anti-HBe HBV DNA by PCR 4. R/O other causes of liver disease: anti-HCV anti HDV

Indications of Liver Biopsy • HBs Ag + • Chronic or intermittent elevations of ALT • Candidate for treatment HBV DNA > 2. 000 or 20. 000 IU/m. L • No contraindications for treatment
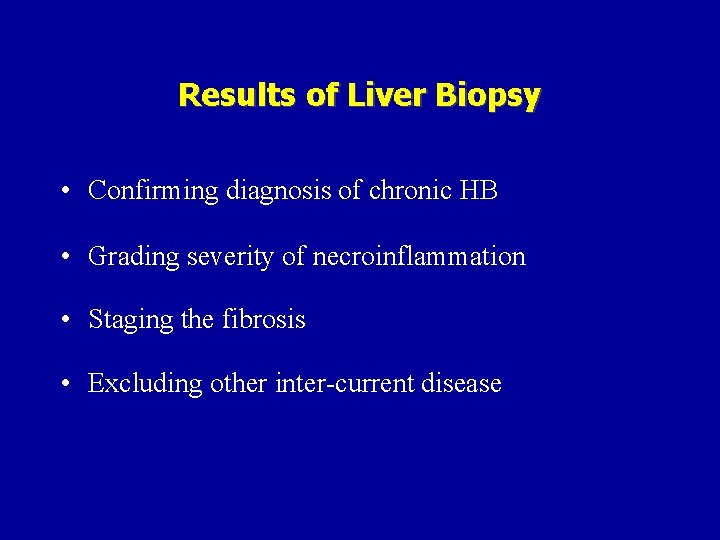
Results of Liver Biopsy • Confirming diagnosis of chronic HB • Grading severity of necroinflammation • Staging the fibrosis • Excluding other inter-current disease

Scoring Systems for Chronic Hepatitis Maximum Comments grade score stage score HAI (Knodell) 18 4 Original scoring system Sheuer 4 4 1 st to separate stage & grade METAVIR 4 4 Excellent Modified HAI (Ishak) 18 6 Most widely used in USA & UK
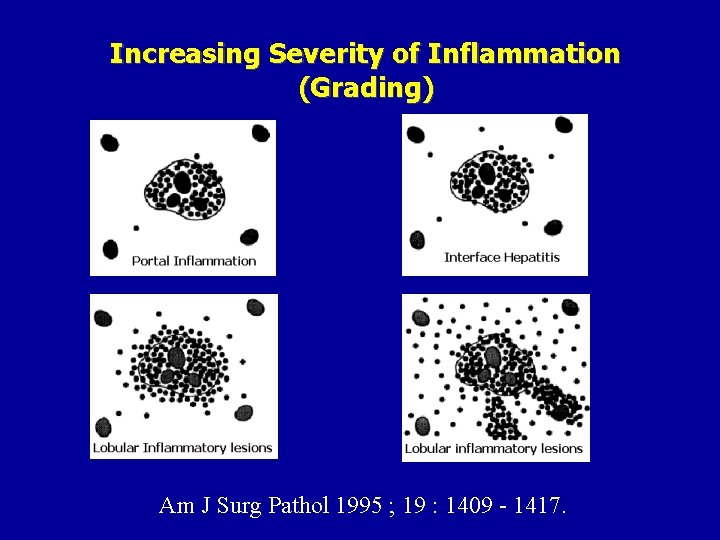
Increasing Severity of Inflammation (Grading) Am J Surg Pathol 1995 ; 19 : 1409 - 1417.
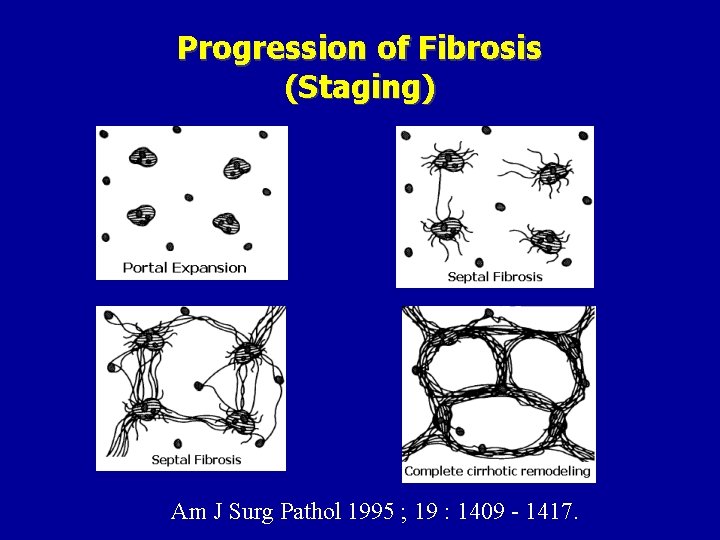
Progression of Fibrosis (Staging) Am J Surg Pathol 1995 ; 19 : 1409 - 1417.

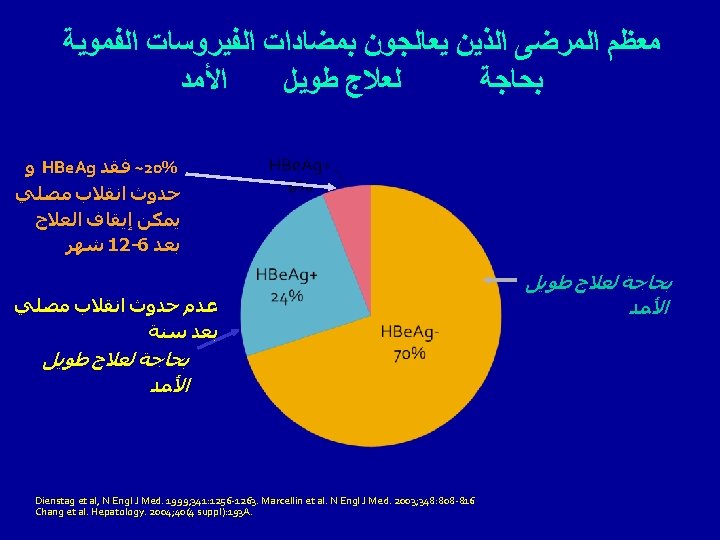
Goal of Treatment of Chronic HBV • HBe Ag + Seroconversion of HBe Ag to anti-HBe • HBe Ag – HBV DNA < 2. 000 IU/m. L Disappearance of HBs Ag is not the goal


How & when to assess the response to treatment?

Category of Response Biochemical (BR) Decrease in serum ALT to normal range Virological (VR) HBe Ag + Loss of HBe Ag – HBV DNA <104 copies/ml Histological (HR) Decrease in HAI by at least 2 points compared to pre-treatment liver biopsy Complete (CR) Biochemical & virological response & loss of HBs Ag

What are the predictive factors of response to treatment?

Predictive Factors of Response to Therapy • High level of ALT • Low lever of HBV DNA • Female patients • Infection in adulthood • Severe necro-inflammation activity in liver biopsy

Treatment of HBe Ag + chronic hepatitis B • IFN • ADF • ENT • TDF First choice Contraindication to INF Intolerance to INF No response to INF Lamivudine resistant mutants (First choice: ESAL-2008)
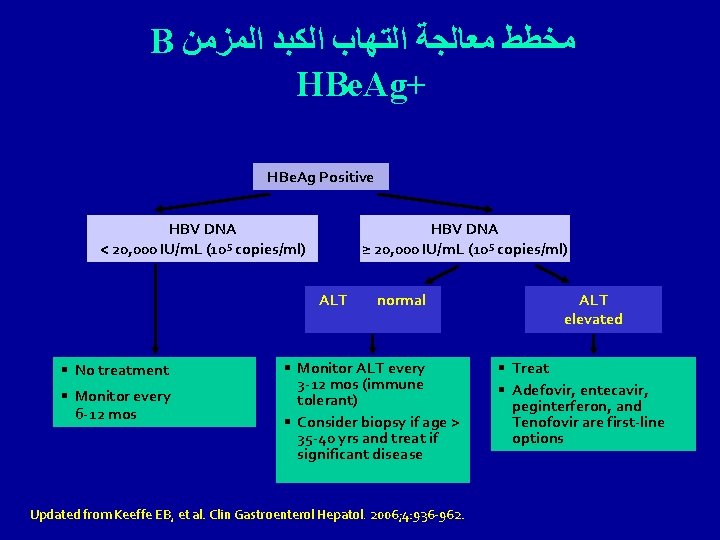
B ﻣﺨﻄﻂ ﻣﻌﺎﻟﺠﺔ ﺍﻟﺘﻬﺎﺏ ﺍﻟﻜﺒﺪ ﺍﻟﻤﺰﻣﻦ HBe. Ag+ HBe. Ag Positive HBV DNA < 20, 000 IU/m. L (105 copies/ml) HBV DNA ≥ 20, 000 IU/m. L (105 copies/ml) ALT § No treatment § Monitor every 6 -12 mos normal § Monitor ALT every 3 -12 mos (immune tolerant) § Consider biopsy if age > 35 -40 yrs and treat if significant disease Updated from Keeffe EB, et al. Clin Gastroenterol Hepatol. 2006; 4: 936 -962. ALT elevated § Treat § Adefovir, entecavir, peginterferon, and Tenofovir are first-line options

Treatment of HBe Ag – chronic hepatitis B • IFN • ADF • ENT • TDF First choice Contraindication to INF Intolerance to INF No response to INF Lamivudine resistant mutants(YMDD) YMDD In view of the need for long term treatment, IFN or ADF, ENT, TDF is preferred
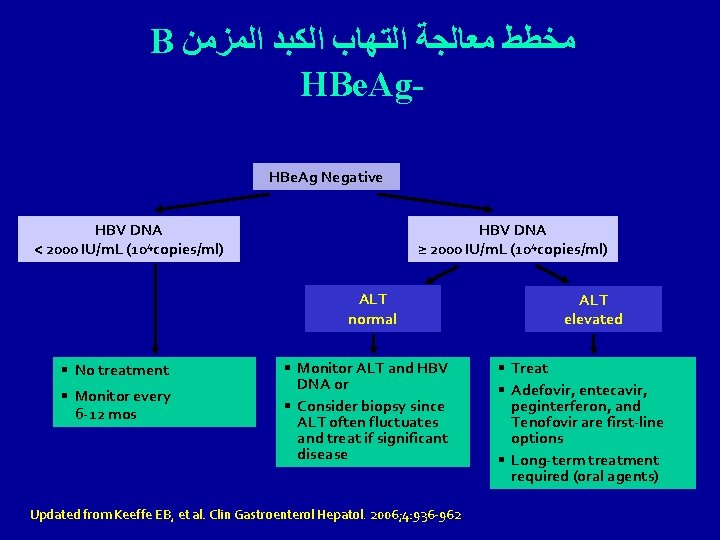
B ﻣﺨﻄﻂ ﻣﻌﺎﻟﺠﺔ ﺍﻟﺘﻬﺎﺏ ﺍﻟﻜﺒﺪ ﺍﻟﻤﺰﻣﻦ HBe. Ag Negative HBV DNA < 2000 IU/m. L (104 copies/ml) HBV DNA ≥ 2000 IU/m. L (104 copies/ml) ALT normal § No treatment § Monitor every 6 -12 mos § Monitor ALT and HBV DNA or § Consider biopsy since ALT often fluctuates and treat if significant disease Updated from Keeffe EB, et al. Clin Gastroenterol Hepatol. 2006; 4: 936 -962. ALT elevated § Treat § Adefovir, entecavir, peginterferon, and Tenofovir are first-line options § Long-term treatment required (oral agents)
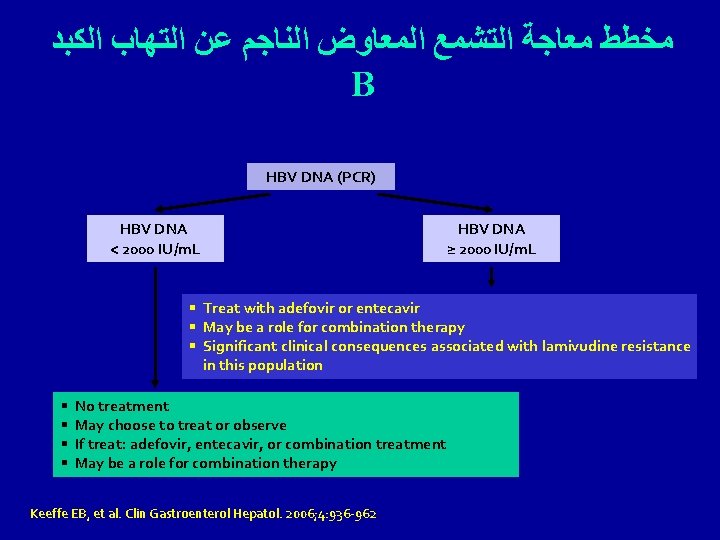
ﻣﺨﻄﻂ ﻣﻌﺎﺟﺔ ﺍﻟﺘﺸﻤﻊ ﺍﻟﻤﻌﺎﻭﺽ ﺍﻟﻨﺎﺟﻢ ﻋﻦ ﺍﻟﺘﻬﺎﺏ ﺍﻟﻜﺒﺪ B HBV DNA (PCR) HBV DNA < 2000 IU/m. L HBV DNA ≥ 2000 IU/m. L § Treat with adefovir or entecavir § May be a role for combination therapy § Significant clinical consequences associated with lamivudine resistance in this population § § No treatment May choose to treat or observe If treat: adefovir, entecavir, or combination treatment May be a role for combination therapy Keeffe EB, et al. Clin Gastroenterol Hepatol. 2006; 4: 936 -962.


Side Effects of Interferon • • • Influenza-like symptoms Alopecia Neutropenia & thrombocytopenia Depression Induction of autoimmune disease (thyroid, …. ) Cardiac complication: MI, AP Erythema at injection site Loss of libido Diabetes mellitus

Contraindications of Interferon • Current psychosis or a history of psychosis • Uncontrolled depressive illness • Presence of active auto-immune disease • Neutropenia or thrombocytopenia • Decompensated cirrhosis • Symptomatic heart disease • Uncontrolled seizures

Follow-up of Patients on IFN Therapy • CBC ½ dose Stop • Thyroid tests q 2 weeks to 8 weeks then q 8 weeks WBC < 1 500 / mm Neutrophils < 750 / mm Platelets < 50 000 / mm WBC < 1 000 / mm Neutrophils < 500 / mm Platelets < 25 000 / mm q 3 - 4 months

Inactive HBs Ag Carrier State* 1. HBs Ag + > 6 months 2. HBe Ag – , anti-HBe + 3. Serum HBV DNA < 20. 000 IU/m. L 4. Persistently normal ALT/AST levels 5. Liver biopsy: absence of significant hepatitis necroinflammatory score < 4 * Previously described as ‘healthy’ carrier state

Follow-up of Inactive HBs Ag Carrier State • ALT q 6 -12 months • If ALT >1 -2 x ULN: check serum HBV DNA level & exclude other causes of liver disease • Consider screening for HCC in relevant population FP & US every 6 months

How & to whom you give the vaccine of HBV?

Schedule of Vaccine Standard Rapid Accelerated 0, 1, 6 months 0, 1, 2 months 0, 7, 21 days Higher antibody levels after 3 rd dose Rapid protection 4 th dose at 12 months for those at intermediate risk for those at high-risk

Indications of HBV vaccine • Universal All infants All children & adolescents not vaccinated • High risk group Health care workers Household contacts CRF & hemodialysis patients Repeated blood transfusions Homosexuals Sexual partners of HBV carriers Illicit injection drug users

Adverse Events of Vaccine • Minimal reactions Local pain: only local pain more frequent in PCT Mild & transient fever: mostly lasting only 24 h • Anaphylaxis Incidence 1 / 600 000 vaccine doses Epinephrine should always be available No severe or fatal anaphylactic reaction reported • Demyelinating diseases Not support for causal relationship

Efficacy of HBV Vaccine & Age Efficacy of the vaccine depends on age Newborns 100 % < 20 years 95 % < 40 years 90 %
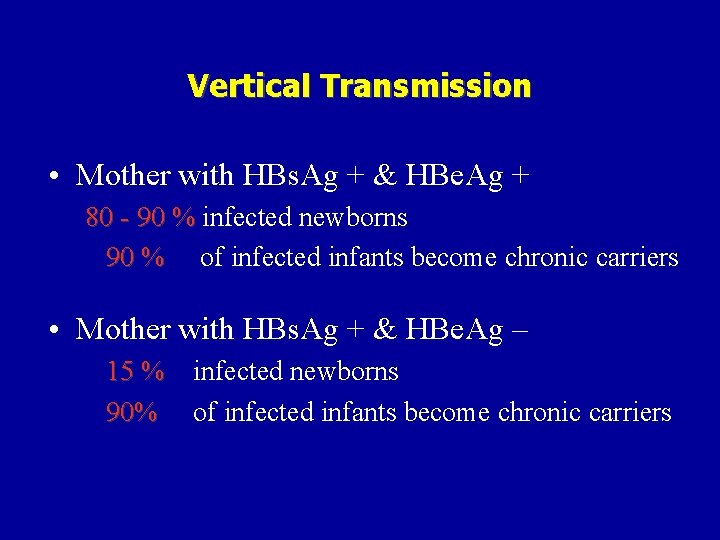
Vertical Transmission • Mother with HBs. Ag + & HBe. Ag + 80 - 90 % infected newborns 90 % of infected infants become chronic carriers • Mother with HBs. Ag + & HBe. Ag – 15 % infected newborns 90% of infected infants become chronic carriers

How to prevent the vertical transmission of HBV?

Active & Passive Immunization • Immediately after Delivery 1 st dose Vaccine in 2 different sites HBIG (200 IU) • 1 month after delivery 2 nd dose Vaccine • 2 month after delivery 3 rd dose Vaccine

Treatment of HBV Infection in Children Treated as adults with modification of doses • IFN: 6 MU / m 2 3 times / week No more than 10 MU per dose • LAM 3 mg / kg / day No more than 100 mg / day • ADF: Not yet approved for treatment in children

Treatment of HBV infection in CRF & KT Data on all 4 agents are limited There are no guideline or consensus • IFN: • LAM: • ADF: • TDF: CRF Indicated with surveillance KT Can lead to rejection CRF Indicated with surveillance KT Indicated Limited data with this drug CRF Indicated with surveillance KT Indicated
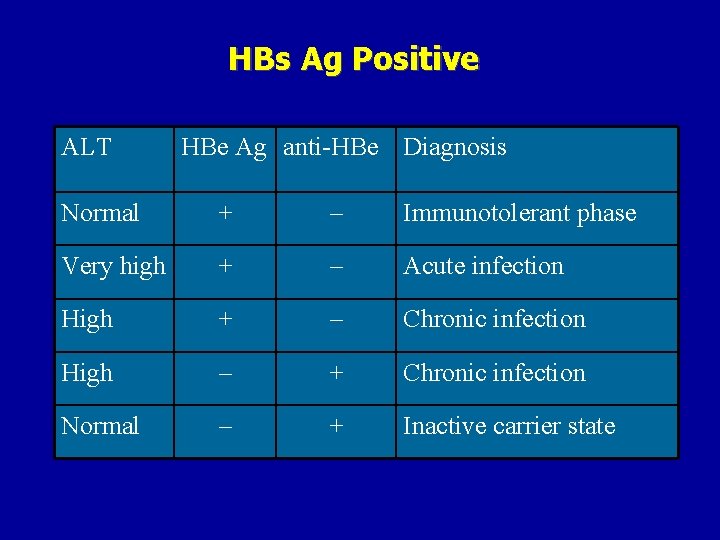
HBs Ag Positive ALT HBe Ag anti-HBe Diagnosis Normal + – Immunotolerant phase Very high + – Acute infection High + – Chronic infection High – + Chronic infection Normal – + Inactive carrier state
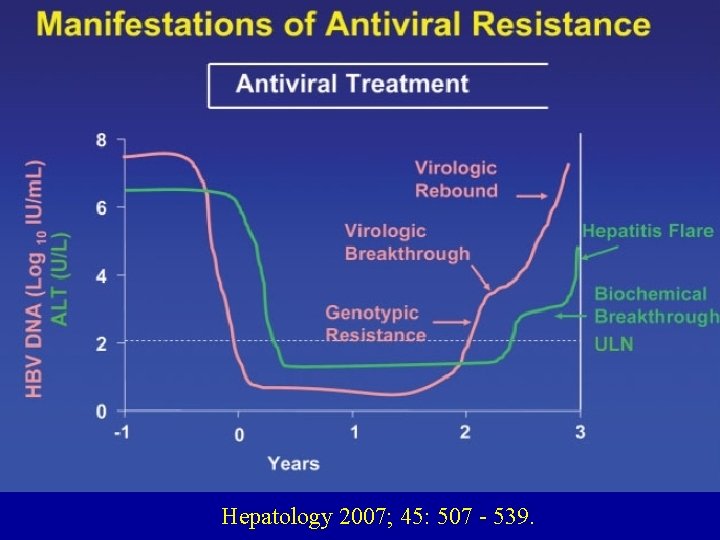
Hepatology 2007; 45: 507 - 539.

Chronic Hepatitis C
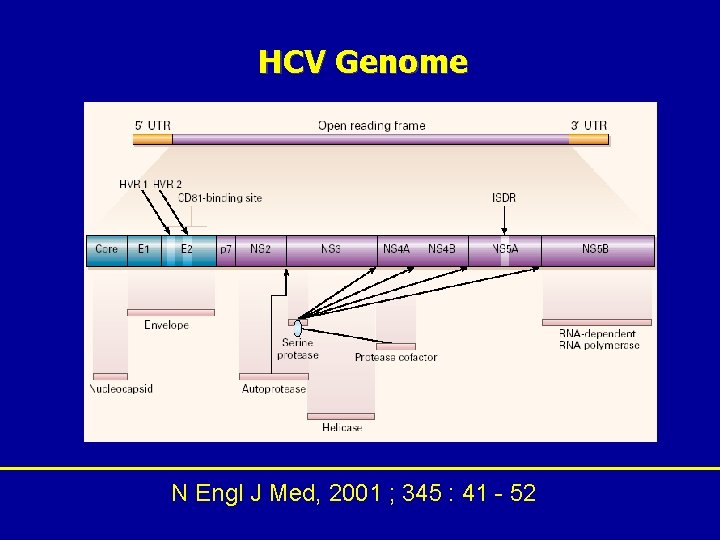
HCV Genome N Engl J Med, 2001 ; 345 : 41 - 52
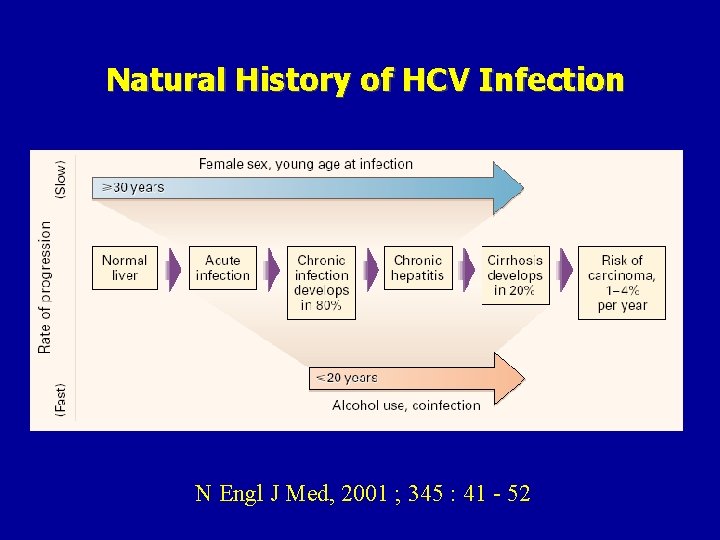
Natural History of HCV Infection N Engl J Med, 2001 ; 345 : 41 - 52

HCV Infection Worldwide Genotype Distribution 1 a, 1 b 2 a, 2 b, 2 c, 3 a 1 a, 1 b 2 a, 2 b, 3 a 2 a 4 4 1 b, 3 a 1 b 1 b, 6 3 b 1 a, 1 b, 2 b, 3 a 5 a 1 b, 3 a Fang et al. Clin Liver Dis. 1997
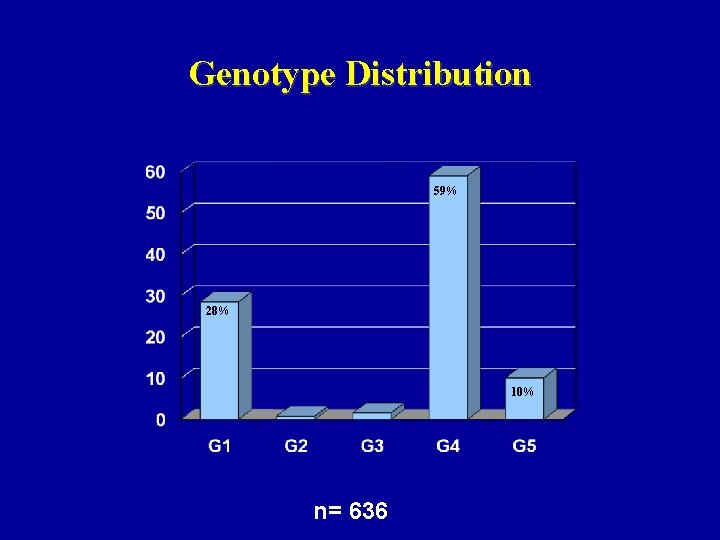
Genotype Distribution 59% 28% 10% n= 636
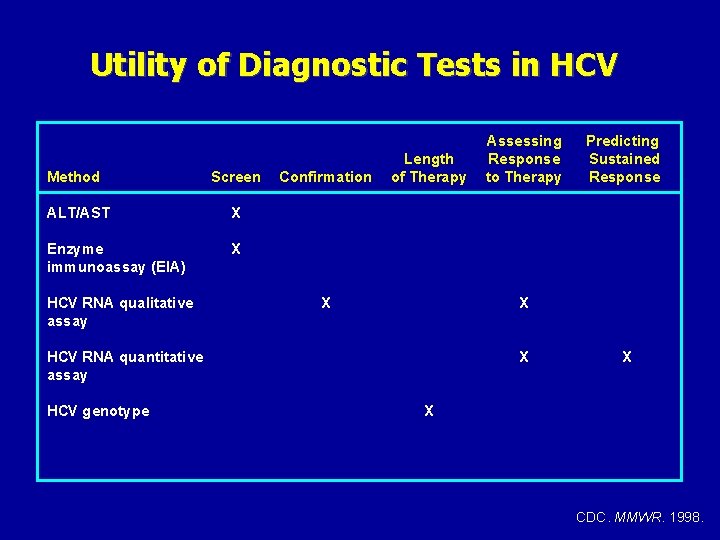
Utility of Diagnostic Tests in HCV Method Screen ALT/AST X Enzyme immunoassay (EIA) X HCV RNA qualitative assay Confirmation Length of Therapy X Predicting Sustained Response X HCV RNA quantitative assay HCV genotype Assessing Response to Therapy X X X CDC. MMWR. 1998.

Interpretation of Hepatitis C Testing Anti HCV RNA Interpretation – – No infection + + HCV present + – Resolved infection Treated HCV < detectable level – + AIDS Hemodialysis Early infection Cleveland Clinic Journal of Medicine 2003 ; 70 : S 7 - S 13.

Algorithm for Laboratory Investigation of Suspected HCV Infection Cleveland Clinic Journal of Medicine 2003 ; 70 : S 7 S 13.

Goals of Therapy in CHC Primary objective = cure Secondary objective=delay/prevent • No virus 1 • Reduce progression of fibrosis 1 • Arrest progression (necrosis/fibrosis) • Reduce progression to cirrhosis 2 • No symptoms • Prevent decompensation • Prevent HCC 2 1. Worman. Hepatitis C: Sourcebook 2002; 2. Peters et al. Medscape HIV/AIDS e. Journal. 2002; 8(1).
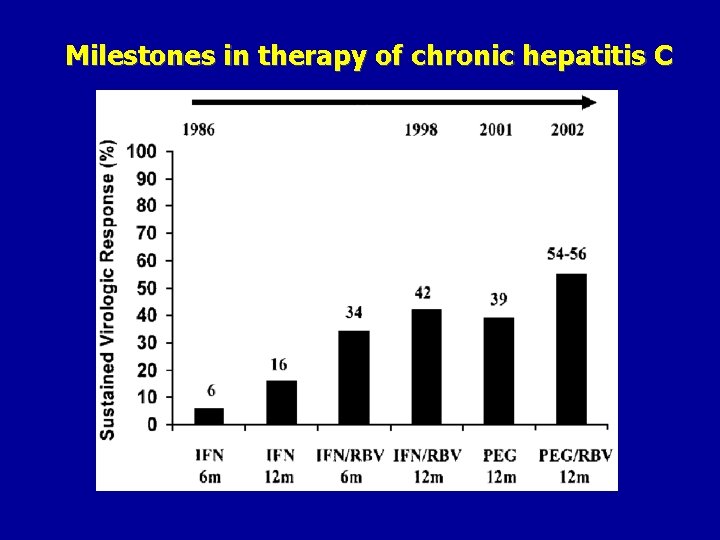
Milestones in therapy of chronic hepatitis C

Available Drugs in CHC • IFN: 3 M units, 3 times weekly • Peg-IFN : - Peg-IFN 2 a - Peg-IFN 2 b 180 gm weekly 1. 5 gm/kg weekly • Ribavirin : 800 - 1200 mg daily • Combination therapy: therapy IFN or Peg-IFN with Ribavirin • Monotherapy: Special cases
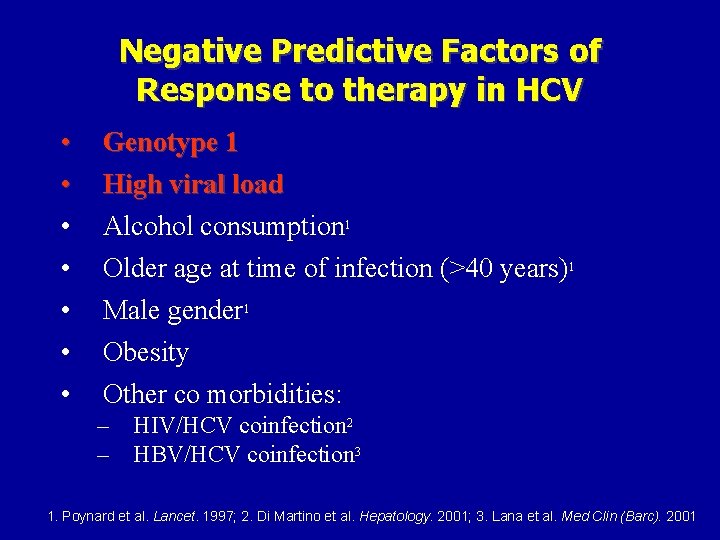
Negative Predictive Factors of Response to therapy in HCV • • Genotype 1 High viral load Alcohol consumption 1 Older age at time of infection (>40 years)1 Male gender 1 Obesity Other co morbidities: – HIV/HCV coinfection 2 – HBV/HCV coinfection 3 1. Poynard et al. Lancet. 1997; 2. Di Martino et al. Hepatology. 2001; 3. Lana et al. Med Clin (Barc). 2001

Treatment of CHC Genotype 1/4 Peg-IFN 2 a 180 gm weekly Peg-IFN 2 b 1. 5 gm/kg weekly Ribavirin 800 -1200 mg daily For 12 weeks – EVR HCV-RNA PCR or +
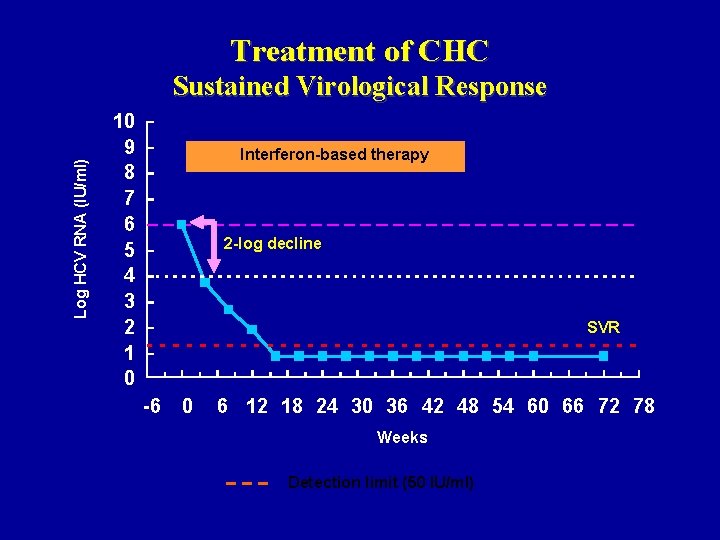
Treatment of CHC Log HCV RNA (IU/ml) Sustained Virological Response 10 9 8 7 6 5 4 3 2 1 0 Interferon-based therapy 2 -log decline SVR -6 0 6 12 18 24 30 36 42 48 54 60 66 72 78 Weeks Detection limit (50 IU/ml)

Treatment of CHC Genotype 2/3 Peg-IFN 2 a 180 gm weekly or Peg-IFN 2 b 1. 5 gm/kg weekly + Ribavirin 800 mg daily For 24 weeks - No need for EVR - SVR: 80 -82% Or IFN 3 Million Unit 3 / week + Ribavirin 1000 -1200 mg/d For 48 weeks if EVR achieved - SVR: 79%
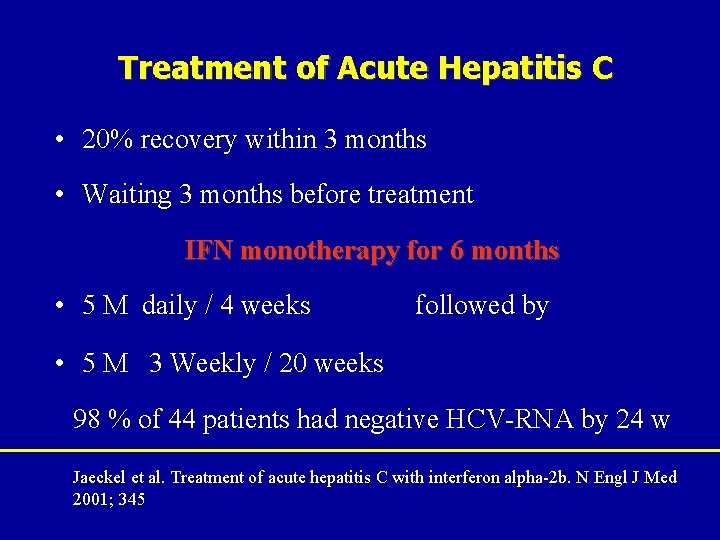
Treatment of Acute Hepatitis C • 20% recovery within 3 months • Waiting 3 months before treatment IFN monotherapy for 6 months • 5 M daily / 4 weeks followed by • 5 M 3 Weekly / 20 weeks 98 % of 44 patients had negative HCV-RNA by 24 w Jaeckel et al. Treatment of acute hepatitis C with interferon alpha-2 b. N Engl J Med 2001; 345

HCV & Renal failure/dialysis • High prevalence of HCV in dialysis units in Syria • Anti-HCV may be negative • Ribavirin is contra-indicated • IFN monotherapy is the treatment • Response to treatment is higher in CRF patients for the same genotype

HCV & Renal failure/dialysis • A tentative of viral eradication should be given before kidney transplantation • If failed and if the liver functions are stable & the liver biopsy doesn’t show advanced liver disease, kidney transplantation is authorized

Treatment of Cirrhosis in CHC • • • We treat if: Bilirubine < 1. 5 mg/dl Serum albumin > 3. 4 g/dl INR < 1. 5 WBC > 1500 Platelets > 75000 No ascitis, no encephalopathy SVR: 43%
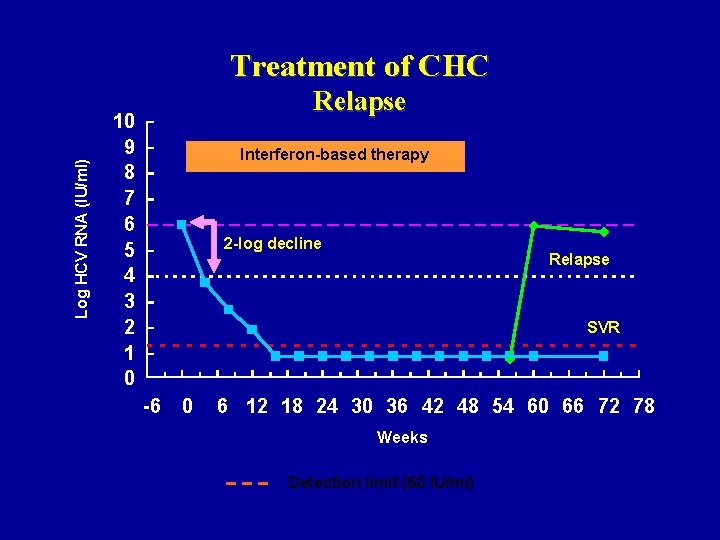
Log HCV RNA (IU/ml) Treatment of CHC Relapse 10 9 8 7 6 5 4 3 2 1 0 Interferon-based therapy 2 -log decline Relapse SVR -6 0 6 12 18 24 30 36 42 48 54 60 66 72 78 Weeks Detection limit (50 IU/ml)
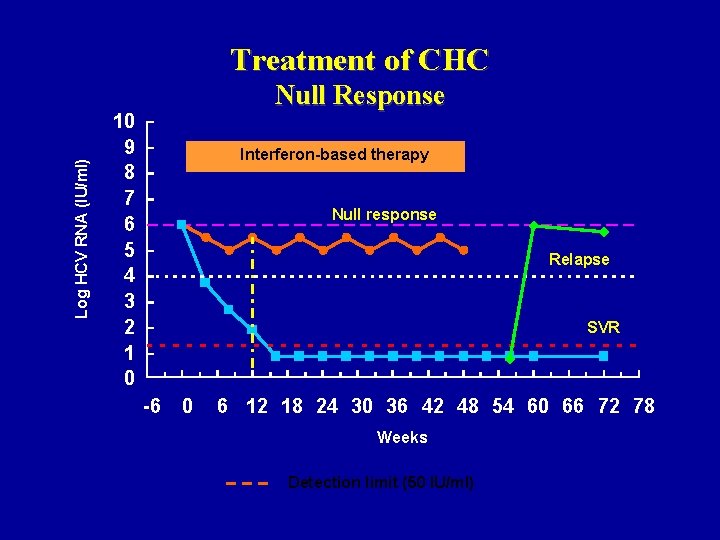
Log HCV RNA (IU/ml) Treatment of CHC Null Response 10 9 8 7 6 5 4 3 2 1 0 Interferon-based therapy Null response Relapse SVR -6 0 6 12 18 24 30 36 42 48 54 60 66 72 78 Weeks Detection limit (50 IU/ml)
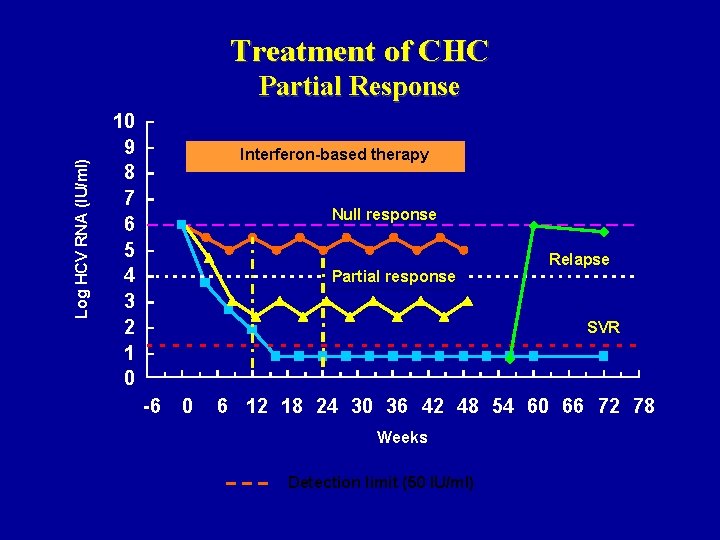
Treatment of CHC Log HCV RNA (IU/ml) Partial Response 10 9 8 7 6 5 4 3 2 1 0 Interferon-based therapy Null response Partial response Relapse SVR -6 0 6 12 18 24 30 36 42 48 54 60 66 72 78 Weeks Detection limit (50 IU/ml)
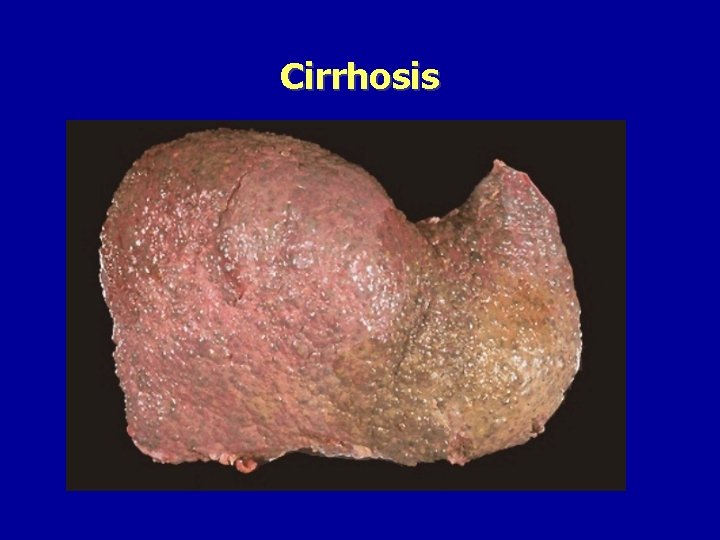
Cirrhosis

- Slides: 73