Healthcare Worker Safety More than replacing devices April


















- Slides: 20

Healthcare Worker Safety; More than replacing devices April 1 st, 2008

Sharps and Needlestick Injury Facts • 70, 000 - 107, 000 incidents each year in Canada • 10% of incidents are high risk • Incident rate rising – e. g. 136% increase in losttime injuries in Ontario from 1998 to 2001 • 58% of sharps injuries are unreported=

Sharps Injuries have a Serious Personal Impact • 6 – 12 months waiting to know test results – serious impact on individuals and families • Treatment is debilitating • Job impacts • HCV is a serious health risk • Chronic infection in 75% - 85% of patients • Active liver disease in 70% of these patients

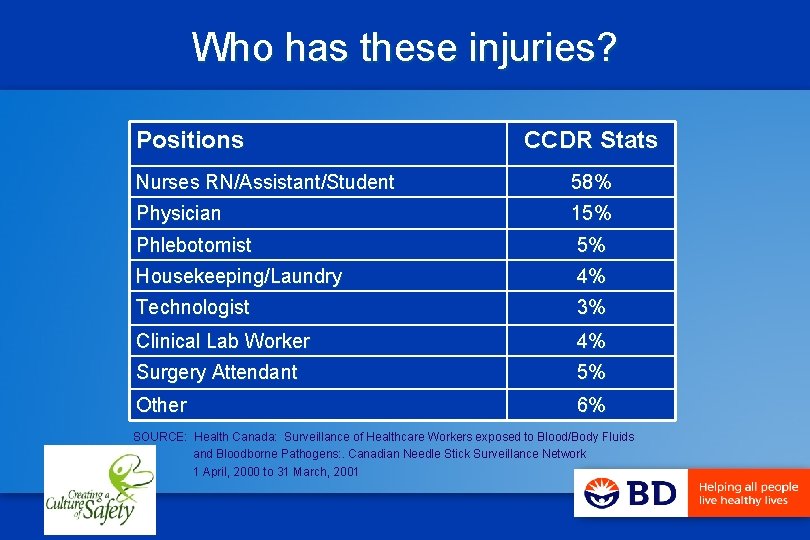
Who has these injuries? Positions CCDR Stats Nurses RN/Assistant/Student 58% Physician 15% Phlebotomist 5% Housekeeping/Laundry 4% Technologist 3% Clinical Lab Worker 4% Surgery Attendant 5% Other 6% SOURCE: Health Canada: Surveillance of Healthcare Workers exposed to Blood/Body Fluids and Bloodborne Pathogens: . Canadian Needle Stick Surveillance Network 1 April, 2000 to 31 March, 2001

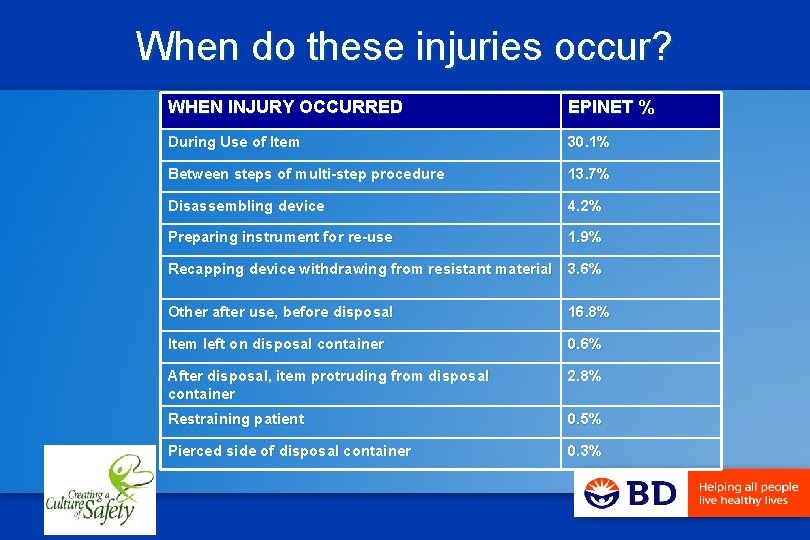
When do these injuries occur? WHEN INJURY OCCURRED EPINET % During Use of Item 30. 1% Between steps of multi-step procedure 13. 7% Disassembling device 4. 2% Preparing instrument for re-use 1. 9% Recapping device withdrawing from resistant material 3. 6% Other after use, before disposal 16. 8% Item left on disposal container 0. 6% After disposal, item protruding from disposal container 2. 8% Restraining patient 0. 5% Pierced side of disposal container 0. 3%

Significant Injury Reduction is Possible Reduction in Injuries: • CDC: • 75% reduction in injuries • U of Virginia - EPINET: • 85% reduction from engineering controls • 95% reduction from comprehensive approach • Toronto East General: • 80% reduction in total injuries • 100% reduction in blood collection injuries

National Survey - Manitoba • First to legislate mandated environment – announced in Sept 04 • Bill 23 – The Workplace Safety and Health Amendment Act (Needles in Medical Workplaces) • Passed into law effective January 2006 • Change process is currently underway – 3 year plan lead by WRHA

National Survey - Saskatchewan • Minister of Labour announced mandated environment at SFL Congress Sept 04 • Regulations passed fall 2005 – requirement in 2006 • Mandates use of SEMD in all workplaces where there is the risk of a sharps injury

National Survey – Nova Scotia • New regulations passed Summer ’ 06 – Safe Needles in Healthcare Workplaces Act • • • Mandates use of SEMD Applies to all health care workplaces Fines up to $250 k

National Survey - Ontario • Ministry of Labour conducted audits fall 2004 – generated inconsistent activity • April 05 Martell tables private member’s bill mandating SEMD • April 05 Min of Health announces $11. 6 million one-time funding – funds allocated for every acute care facility • August ’ 07 announcement of regulation (474/07) to mandate use of SEMD – hospitals in ’ 08, all other workplaces in ‘ 09

Quebec, New Brunswick, Newfoundland PEI • Quebec – approach still to be defined • New Brunswick – NBNU, NBGEU, CUPE have campaign underway • Newfoundland – stakeholder report into Mo. L + Mo. H • PEI – 100% converted to SEMD

National Survey – B. C. Leads the Way • Updated regulations in 1998. Hierarchy of risk reduction beginning with engineering controls required the use of SEMD but did not specify this • Fines to VIHA prompted province-wide conversion • Updated regulations Jan 1, 2008 mandates SEMD – device that provides the “highest level of safety”

National Survey – B. C. Leads the Way • New regulations – “highest level of safety” and broad definition of biohazards including pandemic influenza • Unique roles and dialogue between stakeholders – BCNU, Work. Safe BC, Health Regions and OHSAH • Broad implementation of sharps devices – Concurrent implementation at FHA • WHITE system + VIHA Exposure Control Plan

National Survey – B. C. • If more than one type of safety-engineered hollow bore needle or safety-engineered medical sharp is available in commercial markets, the needle or sharp that provides the highest level of protection from accidental parenteral contact must be used. • Based upon information from manufacturers, independent testing agencies, objective product evaluation, or other reliable sources. • Vendors must provide training.

BCNU Focus Group Findings 1. Time available to do training and education is compromising change efforts. 2. Management must provide active support for the change process. 3. Successful implementation of sharps safety technology can improve the overall culture of safety.

Culture of Safety: Still Much To Do VIHA Survey Statistics: Have you reviewed the VIHA BBF Exposure Control Plan? 20% Have you reviewed the VIHA BBF Exposure Control Plan with your staff? 9% For what percentage of your employees that may be occupationally exposed to blood and other body fluids, do you have documentation indicating that each employee has received specific BBF Education/Training? 25%

From adoption to compliance • Compliance still inconsistent within certain provinces: e. g. Alberta, British Columbia • Challenges within certain areas: – Pharmacy – Anaesthesia – Nuc. Med. , ENT

Key Success Factors • • Executive level leadership/sponsorship is essential Proactive Clinical Leadership (Risk Management and/or OHSS) Robust Business Plan Implementation plan that considers broader change management challenges. Framed within context of creating a culture of safety • Strong Education and Training (safety and clinical) • Sharing best practices – creation of reference sites

Comprehensive Approach • U. of Virginia: 83% reduction from engineering controls. 94% reduction from comprehensive approach • Elements of Comprehensive Approach • Exposure Control Plan • Use of Safety-engineered medical devices • Training & Education about risks and prevention • Sharps Injury Log • Post Exposure Control Plan • Creation of a culture of safety – organizational capacity

Healthcare Worker Safety; More than replacing devices April 1 st, 2008