Healthcare Associated Infections HAI Data Reporting and Validation






































- Slides: 42

Healthcare Associated Infections (HAI) Data Reporting and Validation

Continuing Education Disclosures • Successful completion of this continuing education activity requires that you: • • Complete registration and sign in sheet Attend entire educational activity Participate in education activities Complete participant evaluation • This educational activity received no commercial support. • The speakers and Planning Committee for this event have disclosed no financial interests. • Accredited status does not imply endorsement of any commercial products or services by the Department of State Health Services, Continuing Education Service; Texas Medical Association; or American Nurse Credentialing Center • The speakers did not disclose the use of products for a purpose other than what it had been approved for by the Food and Drug Administration. • Complete and submit the evaluation by the end of the session. 6/16/2021 Texas Department of State Health Services 2

Objectives • Describe the healthcare associated infection (HAI) validation process • Discuss common data reporting errors and how to avoid them. 6/16/2021 Texas Department of State Health Services 3

Texas Requirements Overview • Reporting Requirements • Differences between Texas and Centers for Medicaid and Medicare Services (CMS) requirements • Future of Texas HAI reporting 6/16/2021 Texas Department of State Health Services 4

Texas Requirements Central line-associated bloodstream infections (CLABSI) in the following special care settings: adult, pediatric and/or adolescent ICUs & NICUs (Level II/III & Level III Nurseries). Catheter associated urinary tract infections (CAUTI) in the following special care settings: adult, pediatric and/or adolescent ICUs. Surgical site infections (SSI) 6/16/2021 § CHILDREN’S HOSPITALS: Cardiac procedures, heart transplants, spinal surgery with instrumentation, and VP shunt procedures § ALL OTHER GENERAL HOSPITALS & ASCs: Colon surgeries, hip & knee arthroplasties, abdominal & vaginal hysterectomies, vascular procedures, and coronary artery bypass grafts Texas Department of State Health Services 5

Centers for Medicaid and Medicare Services (CMS) Reporting Requirements 6/16/2021 6

Reporting Overview Alerts regarding data & reports View reports & make comments 6/16/2021 7

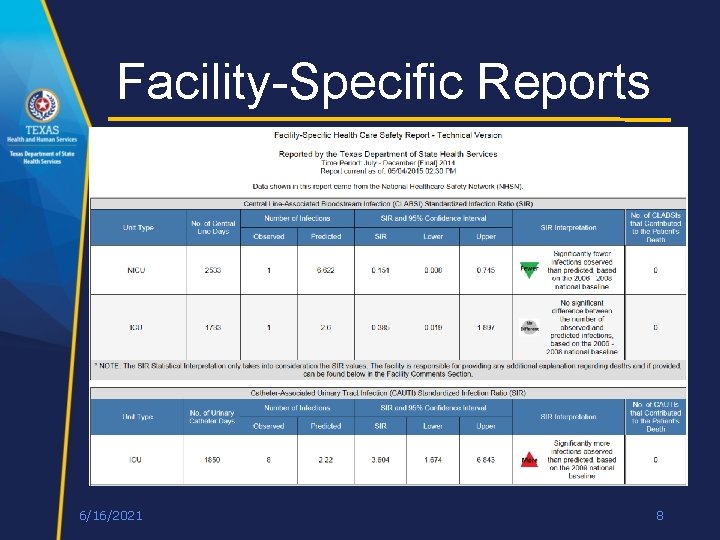
Facility-Specific Reports 6/16/2021 8

Future of Reporting • Changes to Facility-Specific Report due to converting to new Standardized Infection Ratio (SIR) data • Inpatient/Outpatient SSI data will change to Pediatric and Adult SSI data • New Oncology Intensive Care Unit (ICU) row for CAUTI and CLABSI • Alignment with CMS reporting requirements • On hold until next legislative session 6/16/2021 9

National Healthcare Safety Network (NHSN) Updates 2017 Changes to the SIR reports • SIR Risk Models • SIR trending – incorporating old and new baselines • Endocarditis • Patient Injection Definition • Other Miscellaneous Changes • Frequently Asked NHSN Questions 6/16/2021 10

Frequent Questions QUESTION: TRUE OR FALSE. Surgeries that were closed Primarily and by Other means will BOTH be included in the 2015 SIR. 11

Frequent Questions QUESTION: TRUE OR FALSE. Surgeries that were closed Primarily and by Other means will BOTH be included in the 2015 SIR. • True. The old baseline used to exclude surgeries that were not primarily closed. The new 2015 baseline will include both CLOSURE = PRIMARY and CLOSURE = OTHER. 12

Standardized Infection Ratio (SIR) Risk Models • NHSN updated all risk models for 2015 baseline • New SIR “Bible” released: • https: //www. cdc. gov/nhsn/pdfs/psanalysis-resources/nhsn-sirguide. pdf 6/16/2021 13

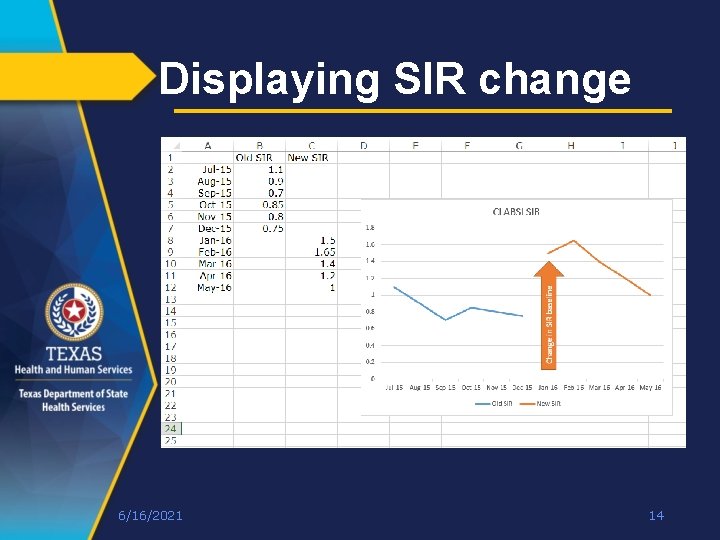
Displaying SIR change 6/16/2021 14

Endocarditis The Infection Window Period (IWP) for Endocarditis is defined as the 21 days during which all site specific criteria are met. Includes date of first positive diagnostic test the 10 calendar days before and the 10 calendar days after. The Repeat Infection Timeframe (RIT) includes the REMAINDER of the patient’s current admission. 6/16/2021 15

Patient Injection A positive blood specimen meeting LCBI criteria that is accompanied by DOCUMENTATION of observed or suspected patient injection into vascular lines is not a CLABSI. A BSI RIT will be created. - Does not cover patient’s family or friends injecting into line. - Does not meet if only “line manipulation” is documented. 6/16/2021 16

Frequent Questions QUESTION 1: If you have more then one diagnostic test used to meet criteria on different dates, which do you use to determine the Infection Window Period? 17

Frequent Questions QUESTION 1: If you have more then one diagnostic test used to meet criteria on different dates, which do you use to determine the Infection Window Period? Use the most localized test. Example: If you have a lab specimen and an imaging test, you would use the lab specimen. 18

Misc. changes 1. Imaging test evidence “suggestive” of infection a. This may include fluid collection 2. Emergency definition updated to match what is documented to be an emergency/urgent procedure in medical record. 3. Group B strep CLABSIs should NOT be reported during neonate’s first 6 days of life. 4. Summary can be found: https: //www. cdc. gov/nhsn/pdfs/pscma nual/psc-january-2017 -revisions. pdf 6/16/2021 19

Validation Overview • Current Process for Validation CLABSI CAUTI SSI • Targeted Assessment and Prevention Reports • Validation Results • Future of Texas HAI validation 6/16/2021 Texas Department of State Health Services 20

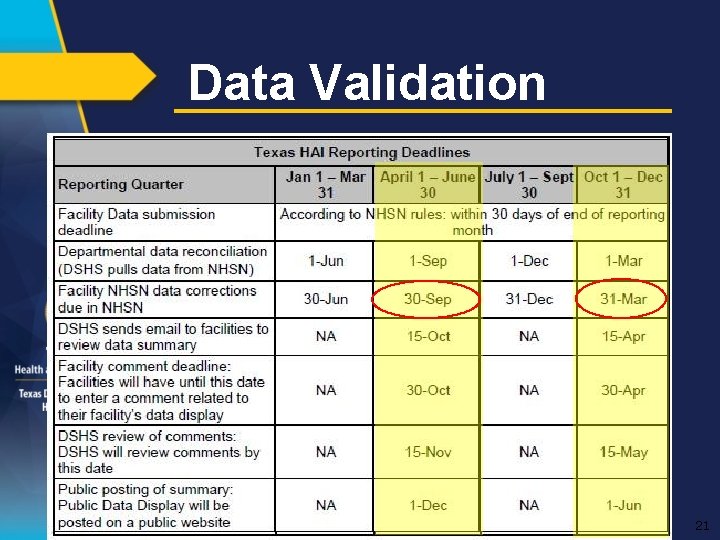
Data Validation 21

Historically… Ø Prior to current validation methods, only reported events were reviewed and validated by DSHS. Ø 97 -99% of events were reported accurately. Those responsible for reporting, mostly Infection Preventionists, had a good grasp of requirements and definitions. Ø Facilities that were audited had very robust IP programs that were good at “finding” and identifying HAIs. 22

CLABSI/CAUTI Validation Ø Facility selection process modeled after the NHSN CLABSI Validation Protocol Ø CDC recommends targeted validation in order to investigate and correct potential deficiencies in an efficient manner. Ø NHSN recommends 21 facilities be chosen via targeted selection and 5% of the remaining facilities selected randomly. For Texas, this is approximately 35 -40 facilities per HAI. 23

Facility Selection: Details Ø 21 in the top 33% of facilities with highest number of expected/predicted infections are selected. Ø Top 7 facilities with SIRs above the median Ø Top 7 with SIRs at or below the median, but above 0 Ø Top 7 with SIRs = 0 Ø 5% of all remaining facilities are randomly selected (~10 -15). 24

Record Selection Selected facilities are required to submit a line list of all positive cultures from the given audit period (6 months). From the line list, DSHS will select: a. Up to 20 patient records of NHSN reported b. Up to 40 patient records of unreported candidate events NOTE: the entire patient’s medical record will be reviewed, not just the data around a single positive culture. 25

Summary of Validation Process 1. Facility notified and line list requested 2. DSHS selects medical records for review and sends to facility 3. Select site visit date and send Facility Audit Survey for completion by facility prior to site visit. 4. Notify CEO/Administrator, DSHS Regulatory and Regional/Local Health Departments about upcoming visit 5. Review Facility Audit Survey and perform site visit • Introductions/Entrance Interview • Partially “Blind” Chart Review • Debriefing/Conclusions 6. Send Validation Summary Report to IPs, CEO/Admin and other staff as needed. 26

Surgical Site Infection (SSI) Validation Ø SSI: Identify facilities based on Standardized Infection Ratio (SIR): If Statistically Significantly High Ø 2 Audit Tiers: Ø First Time High SIR – no high SIR for same HAI for previous time period) Ø Subsequent High SIR – high SIR for same HAI for two reporting periods in a row 27

SSI Validation Ø First Time High SSI SIR Ø Purpose: To ensure facility is applying the CDC definitions correctly and to verify the number of infections reported to DSHS. § Audits for those facilities with significantly high SIRs to verify data reported meet NHSN HAI criteria § Review of reported SSI events only § Record Review & IP/Administration staff Interview 28

SSI Validation § Subsequent High SIR Investigations: § Purpose: DSHS will aid facilities in prevention efforts and provide consultation/support as needed. § Conducted by Epidemiologists with Certification in Infection Control and Prevention (CIC certification) § Phone consultation to review interventions taken and action plans in place at facility to determine if site visit is warranted § If site visit needed, CIC HAI Epidemiologist will come to facility and may perform environmental rounds, interview floor staff, observe procedures/patient care activities, review policies and patient records. 29

Validation Results For validation of data reported between Jan 2015 – July 2016: 1. CLABSI: a. 2, 326 records reviewed for CLABSI b. 86 (3. 7%) Discrepancies noted 2. SSI: a. 309 records reviewed for SSI b. 14 (4. 5%) Discrepancies noted 3. CAUTI: a. 472 records reviewed for CAUTI b. 9 (1. 9%) Discrepancy noted 30

CAUTI Common Errors • Date of Event: IP uses date of positive culture instead of date of first sign/symptom • Foley wasn’t in place for > 2 days • Infection attributed to wrong unit • Facility didn’t look for symptoms that can be used with catheter is not in place (e. g. urgency, frequency) after foley was removed. 6/16/2021 31

CLABSI Common Errors • #1: Inappropriately attributing as secondary to pneumonia (PNEU) or intra abdominal infection (IAB). • Used non-definitive imaging without clinical correlation • NOTE: clinical correlation is physician documentation of treatment for primary infection • Documentation of central line or symptom missed initially or unable to be found at validation • Definition of hypotension – if no policy will default to electronic flags/alert thresholds. 6/16/2021 32

Frequent Questions QUESTION 2: Can a secondary Blood Stream Infection (BSI) have a Repeat Infection Timeframe (RIT)? 33

Frequent Questions QUESTION 2: Can a secondary Blood Stream Infection (BSI) have a Repeat Infection Timeframe (RIT)? • No. Only primary Blood Stream Infections create a BSI RIT. Secondary BSIs do not create a BSI RIT. 34

Frequent Questions QUESTION 3: Can you use non-definitive chest x-rays (e. g. opacities/infiltrates noted but pneumonia not specified as cause) to meet pneumonia criteria? 35

Frequent Questions QUESTION 3: Can you use non-definitive chest x-rays (e. g. opacities/infiltrates noted but pneumonia not specified as cause) to meet pneumonia criteria? • If there is any documentation in the medical record that correlates the nondefinitive findings on imaging with what is clinically happening with the patient and that documentation suggests it is pneumonia and there is treatment for pneumonia then the imaging tests would be eligible for use in meeting the imaging portion of the PNEU definitions. 36

SSI Common Errors • Wrong depth of SSI reported • EX: Superficial reported when it should have been Deep • Subsequent SSI for same patient not reported • Remember, SSIs do not have repeat infection timeframes. • Joint (JNT) Infection reported instead of Periprosthetic Joint Infection (PJI) for SSIs following knee or hip replacements. 6/16/2021 37

Frequent Questions QUESTION 4: TRUE OR FALSE. If there was an infection present at the time of surgery (PATOS), then I do not have to report it in NHSN. 38

Frequent Questions QUESTION 4: TRUE OR FALSE. If there was an infection present at the time of surgery (PATOS), then I do not have to report it in NHSN. • FALSE. You will still report these infections as PATOS = YES. But they will not be included in the 2015 SIR calculation. 39

Validation Updates • DSHS began utilizing Go. To. Meeting to conduct remote audits • To mitigate “validation fatigue” facilities with > 10% discrepancies will be re-audited the subsequent time period • Coming soon! SSI validation using NHSN guidance 40

Current Audit Team Candace Campbell, MPH DSHS Epidemiologist Candace. Campbell@dshs. texas. gov Office Phone: 512. 776. 6488 Nesreen Gusbi, MPH(c) DSHS HAI/PAE Specialist Nesreen. Gusbi@dshs. texas. gov Office Phone: 512. 776. 2588 6/16/2021 41

Thank you Jennifer Vinyard, MPH, CIC Email: Jennifer. Vinyard@dshs. texas. gov Office: 512 -776 -3773 6/16/2021 42