HEALING OF SPECIALISED TISSUE BONE Bone healing can

HEALING OF SPECIALISED TISSUE
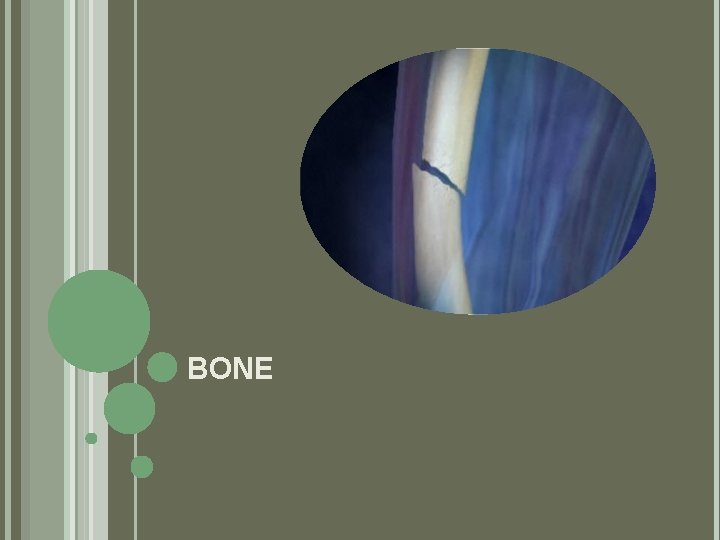
BONE

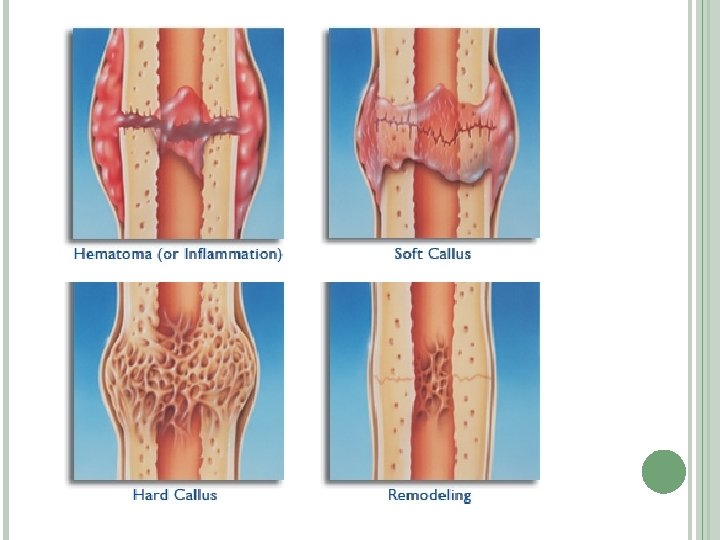
Bone healing can be described in 6 stages : 1. Stage of hematoma formation 2. Stage of liquefaction 3. Stage of neovascularisation 4. Stage of soft callus formation 5. Stage of hard callus formation 6. Stage of remodelling
Stage of hematoma formation: Accumulation of blood at fracture site Also contains devitalized soft tissue, dead bone, and necrotic marrow Stage of liquefaction: Degradation of nonviable products at the fracture site

Stage of neovascularisation: Revascularization starts from adjacent normal bone, with new blood vessels growing into the fracture site Similar to the formation of granulation tissue in soft tissue Clinically : signs and symptoms characteristic of inflammation
Stage of soft callus formation: Occurs 3 to 4 days following injury Soft tissue is deposited where neovascularization has taken place : soft callus The soft callus : �Forms a bridge between the fractured bone segments �Serves as an internal splint, preventing damage to the newly laid blood vessels �Achieves a fibrocartilaginous union. Clinically : End of pain and inflammatory signs.
Stage of hard callus formation: Mineralization of the soft callus and conversion to bone May take up to 2 to 3 months and leads to complete bony union. Clinically : allows weight bearing and appears healed on x rays Stage of remodeling : Excessive callus is reabsorbed Marrow cavity is recanalized. Allows correct transmission of forces and restores the contours of the bone.

OTHER DIFFERENCES Bone morphogenic proteins (BMPs) is a type of Tumor growth Factor It stimulates the differentiation of mesenchymal cells into chondroblasts and osteoblasts, Directly affects bone and cartilage repair
CARTILAGE
Consists of cells (chondrocytes) surrounded by an extracellular matrix made up of several proteoglycans, collagen fibers, and water. Very avascular Depends on diffusion for transmission of nutrients across the matrix. Perichondrium is hypervascular and contributes to the nutrition of the cartilage. Therefore, injuries to cartilage may be associated with permanent defects due to the meager and tenuous blood supply

Healing depends on depth of injury Healing varies in : 1. Superficial injuries 2. Deeper injuries

SUPERFICIAL CARTILAGE INJURY Disruption of the proteoglycan matrix and injury to the chondrocytes. No inflammatory response There is increase in synthesis of proteoglycan and collagen But, the healing power of cartilage is often inadequate

Hence superficial injuries: i. Have incomplete overall regeneration ii. Are slow to heal iii. Result in persistent structural defects

DEEP CARTILAGE INJURY Involves the underlying bone and soft tissue. So there is exposure of vascular channels of the surrounding damaged tissue This helps in the formation of granulation tissue. Heals better than superficial injuries !!
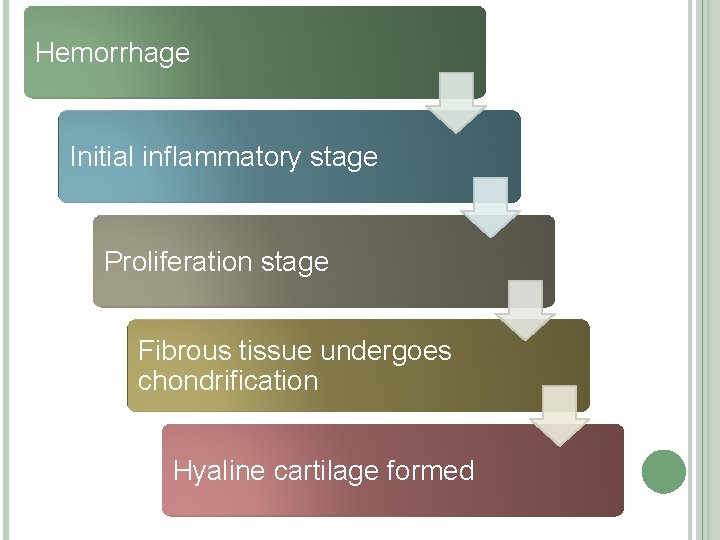
Hemorrhage Initial inflammatory stage Proliferation stage Fibrous tissue undergoes chondrification Hyaline cartilage formed
TENDONS AND LIGAMENTS

Tendon : links muscle and bone Ligament : links bone and bone Consist of parallel bundles of collagen interspersed with spindle cells Can be subjected to a variety of injuries, such as laceration, rupture, and contusion. As underlying bone or muscles are mobile, the damaged ends usually separate.

Healing progresses in a similar fashion as in other areas of the body ie via the 3 stages Matrix is characterized by accumulation of type I and III collagen along with increased water, DNA, and glycosaminoglycan content. As the collagen fibers are organized, transmission of forces across the damaged portion can occur. Restoration of the mechanical integrity may never be equal to that of the undamaged tendon.

. Healing 1. 2. depends on following factors : Tendon vasculature � Hypovascular tendons tend to heal with less motion and more scar formation Tenocytes � Specialised cells that are very metabolically active and have a large regenerative potential
NERVE

Nerve injuries are very common There are three types of nerve injuries: 1. Neurapraxia : temporary interruption of conduction without loss of axonal continuity 2. Axonotemesis : loss of the relative continuity of the axon and its covering of myelin, but preservation of the connective tissue framework of the nerve 3. Neurotemesis : complete transection
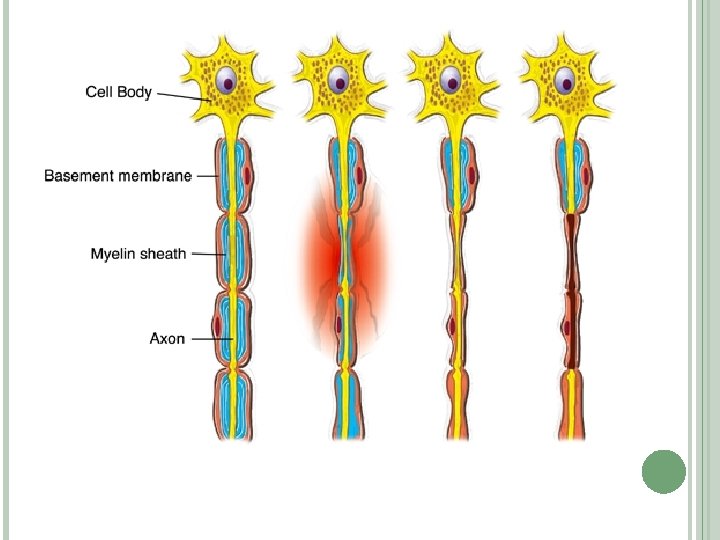

Times of degeneration in nerve: Distal to wound �Wallerian degeneration : Phagocytes remove the degenerating axons and myelin sheath from the distal stump Proximal to wound �Traumatic degeneration upto last node of Ranvier

Pattern a) b) c) of nerve regeneration: survival of axonal cell bodies regeneration of axons that grow across the transected nerve to reach the distal stump migration and connection of the regenerating nerve ends to the appropriate nerve ends or organ targets
FETAL TISSUE

Healing depends on age of gestation: Upto 3 rd trimester: �no scar formation at all From beginning of 3 rd trimester: �"transition wound" �scarless healing �loss of the ability to regenerate skin appendages Later : �scar formation �healing continues to be faster

Biggest difference : lack of scar formation! Reasons i. iii. iv. : wound environment, inflammatory responses, differential growth factor profiles wound matrix.

Wound Environment Sterile Temperature-stable Fluid Growth Factors Absence of TGF, which may have a significant role in scarring.

Inflammation Fetal wounds contain lower numbers of PMNs and macrophages Neutropenia Due to this immaturity of the fetal immune system there is reduced fetal inflammation

Wound Matrix Has excessive hyaluronic acid production Fetal fibroblasts produce more collagen than adult fibroblasts Increased level of hyaluronic acid aids in the orderly organization of collagen. Hence , enhanced healing and less postoperative adhesion formation

THANK YOU !!!
- Slides: 32