Header Transfusion Medicine Meghan Delaney DO MPH Subhead

Header Transfusion Medicine Meghan Delaney, DO, MPH Subhead
Overview of Transfusion Medicine 1. Blood products • Indications 2. Selection of blood products • Compatibility testing 3. Adverse events • Transfusion transmitted disease • Transfusion reactions • Complications ?

Blood Products and Plasma-derived products Blood products Red blood cells Platelets Plasma Cryoprecipitate Plasma derived products Albumin IVIG Coagulation factors
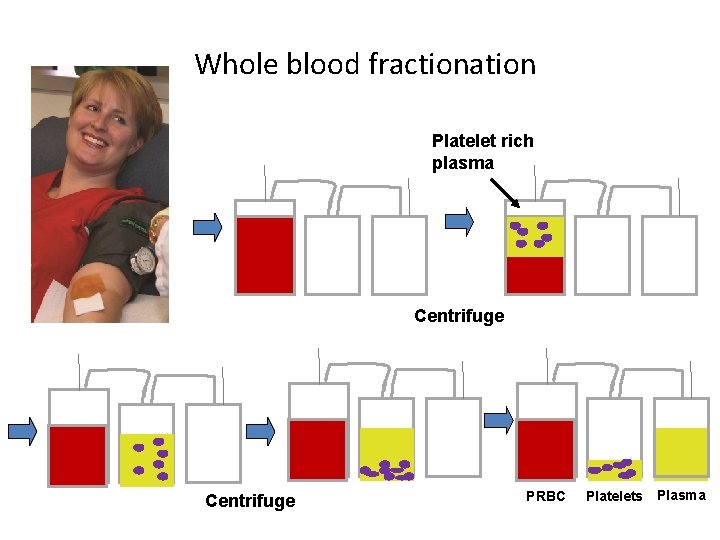
Whole blood fractionation Platelet rich plasma Centrifuge PRBC Platelets Plasma
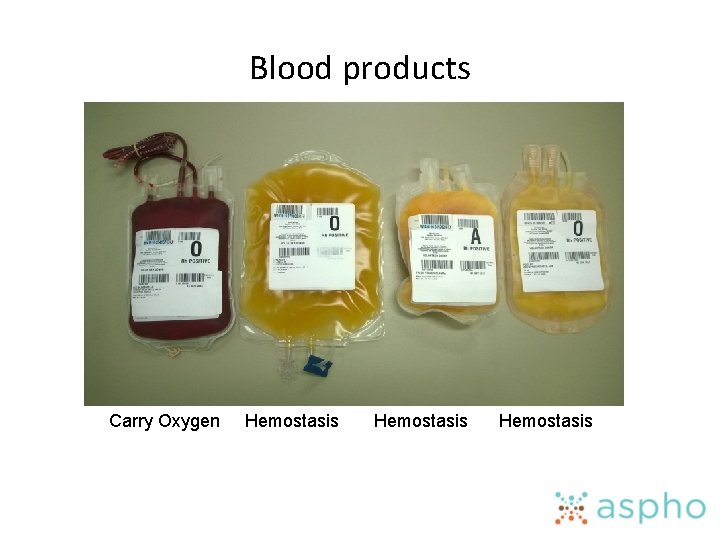
Blood products Carry Oxygen Hemostasis
Red blood cells • Use • Increase O 2 carrying capacity to tissues • Increase cell mass following significant hemorrhage • When other treatments for chronic anemia have failed • Not used • Wound healing • Shortening time to ambulation following surgery
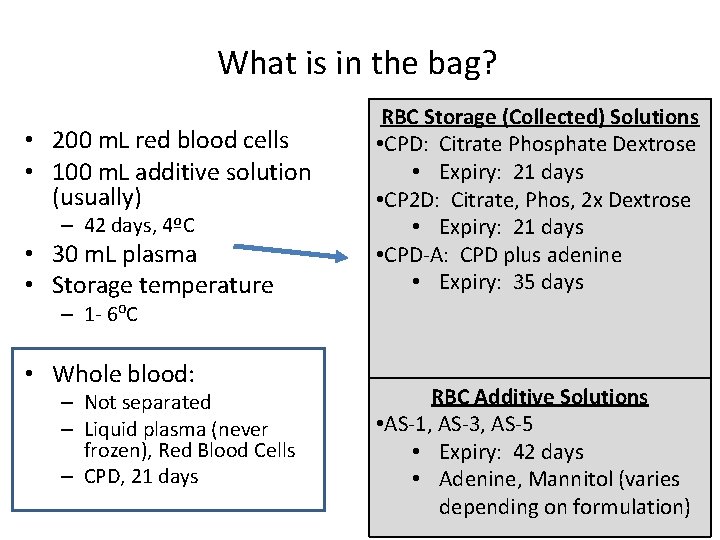
What is in the bag? • 200 m. L red blood cells • 100 m. L additive solution (usually) – 42 days, 4ºC • 30 m. L plasma • Storage temperature RBC Storage (Collected) Solutions • CPD: Citrate Phosphate Dextrose • Expiry: 21 days • CP 2 D: Citrate, Phos, 2 x Dextrose • Expiry: 21 days • CPD-A: CPD plus adenine • Expiry: 35 days – 1 - 6⁰C • Whole blood: – Not separated – Liquid plasma (never frozen), Red Blood Cells – CPD, 21 days RBC Additive Solutions • AS-1, AS-3, AS-5 • Expiry: 42 days • Adenine, Mannitol (varies depending on formulation)

What is in the bag? • Solutes: – Adenine metabolism • 95% phosphoribosyl transferase ok • 5% xanthine oxidase dihydroxyadenine (DOA) insolulble precipitate crystals can block renal tubules and cause liver toxicity – Mannitol – Glucose – Sodium Philps, FS et al. J Pharm Exp Ther, 1952 • Storage lesion – Lose membrane deformability – Loss of 2, 3 -DPG – Acidification – Oxidative damage (Hb) – Elevated K+

Evidence Based Indications: RBC Threshold in Pediatric Critical Care (TRIP-ICU) 637 PICU Patients Hgb < 9. 5 Stable, critically ill Had anemia within 7 days of ICU admission Random 320 420 Restrictive Transfusion Liberal Transfusion RBC if Hgb <7 RBC if Hgb <9. 5 Progressive MODS 12% La. Croix, NEJM, 2007 Progressive MODS 12% p=NS 54% did not receive 2% did not receive a transfusion Restrictive: 44% fewer transfusions
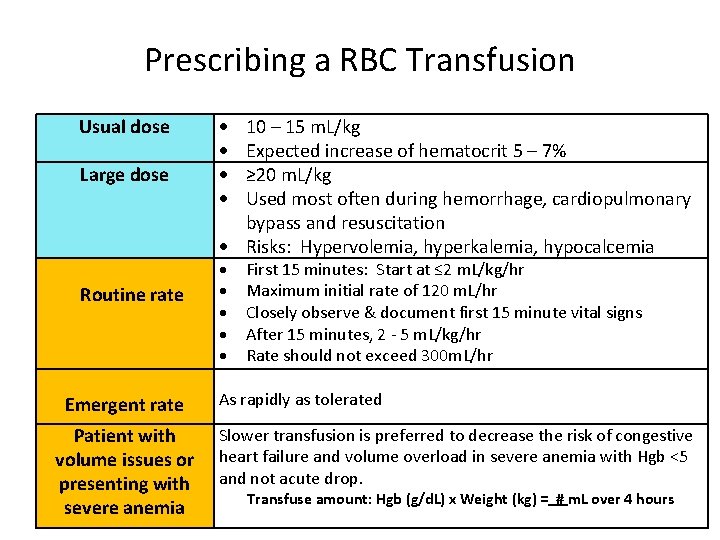
Prescribing a RBC Transfusion Usual dose Large dose Routine rate Emergent rate Patient with volume issues or presenting with severe anemia 10 – 15 m. L/kg Expected increase of hematocrit 5 – 7% ≥ 20 m. L/kg Used most often during hemorrhage, cardiopulmonary bypass and resuscitation Risks: Hypervolemia, hyperkalemia, hypocalcemia First 15 minutes: Start at ≤ 2 m. L/kg/hr Maximum initial rate of 120 m. L/hr Closely observe & document first 15 minute vital signs After 15 minutes, 2 - 5 m. L/kg/hr Rate should not exceed 300 m. L/hr As rapidly as tolerated Slower transfusion is preferred to decrease the risk of congestive heart failure and volume overload in severe anemia with Hgb <5 and not acute drop. Transfuse amount: Hgb (g/d. L) x Weight (kg) = # m. L over 4 hours

Platelets • Function – Initial phase of coagulation • Use – Treat thrombocytopenia – Platelet function disorders • Not used – Pre-procedure prophylaxis to prevent serious bleeding – Expand intravascular compartment Apheresis Platelets
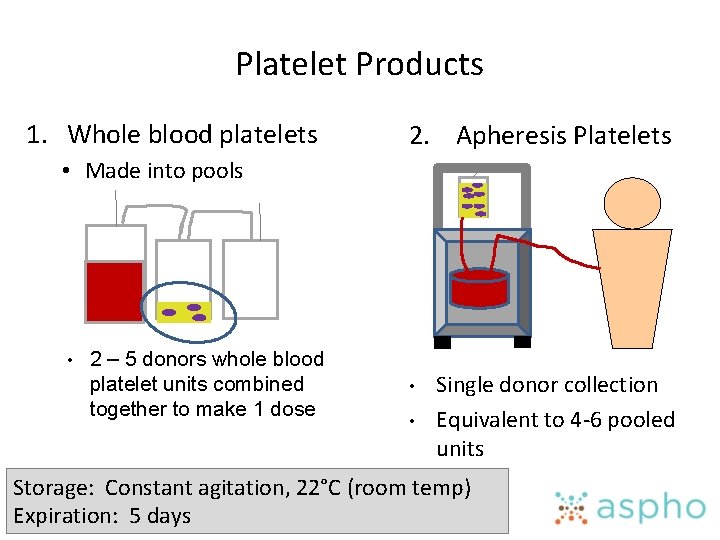
Platelet Products 1. Whole blood platelets 2. Apheresis Platelets • Made into pools • 2 – 5 donors whole blood platelet units combined together to make 1 dose • • Single donor collection Equivalent to 4 -6 pooled units Storage: Constant agitation, 22°C (room temp) Expiration: 5 days
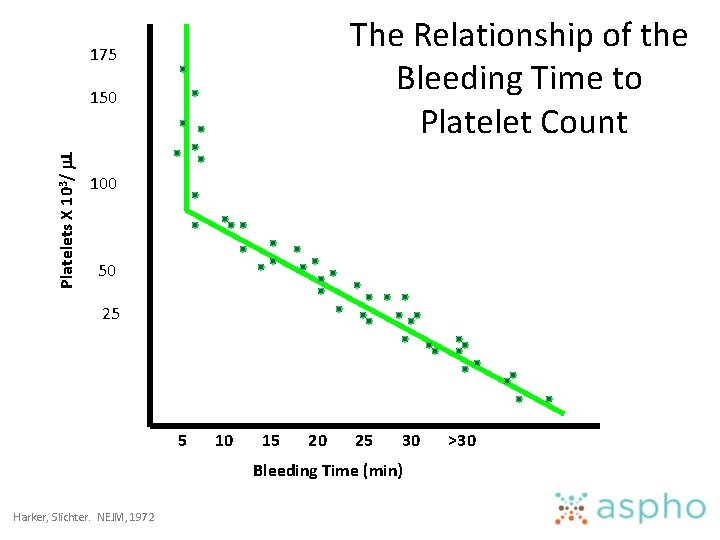
The Relationship of the Bleeding Time to Platelet Count 175 Platelets X 103/ L 150 100 50 25 5 10 15 20 25 30 Bleeding Time (min) Harker, Slichter. NEJM, 1972 >30
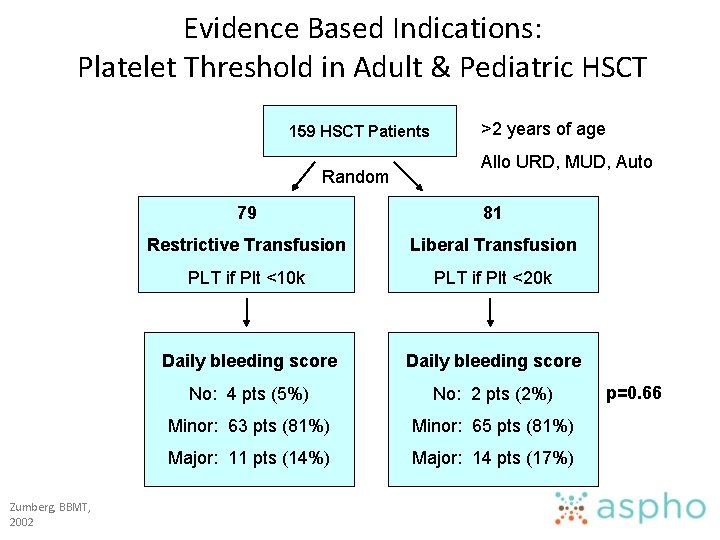
Evidence Based Indications: Platelet Threshold in Adult & Pediatric HSCT 159 HSCT Patients Random Zumberg, BBMT, 2002 >2 years of age Allo URD, MUD, Auto 79 81 Restrictive Transfusion Liberal Transfusion PLT if Plt <10 k PLT if Plt <20 k Daily bleeding score No: 4 pts (5%) No: 2 pts (2%) Minor: 63 pts (81%) Minor: 65 pts (81%) Major: 11 pts (14%) Major: 14 pts (17%) p=0. 66
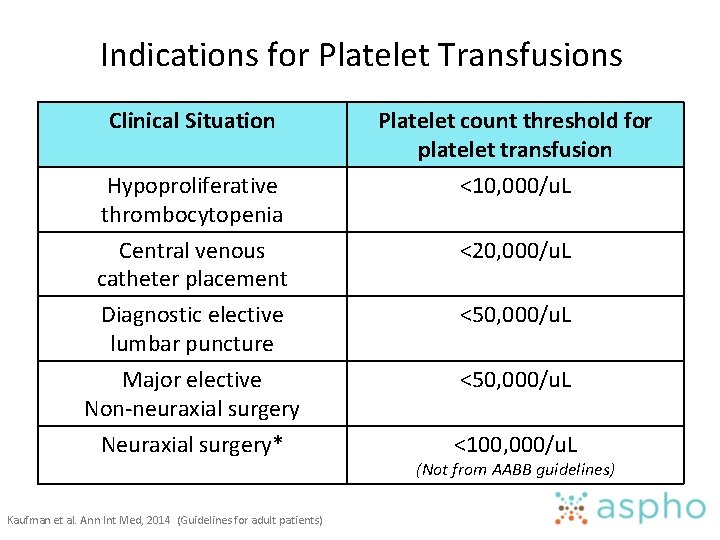
Indications for Platelet Transfusions Clinical Situation Platelet count threshold for platelet transfusion Hypoproliferative thrombocytopenia <10, 000/u. L Central venous catheter placement <20, 000/u. L Diagnostic elective lumbar puncture <50, 000/u. L Major elective Non-neuraxial surgery <50, 000/u. L Neuraxial surgery* <100, 000/u. L Kaufman et al. Ann Int Med, 2014 (Guidelines for adult patients) (Not from AABB guidelines)
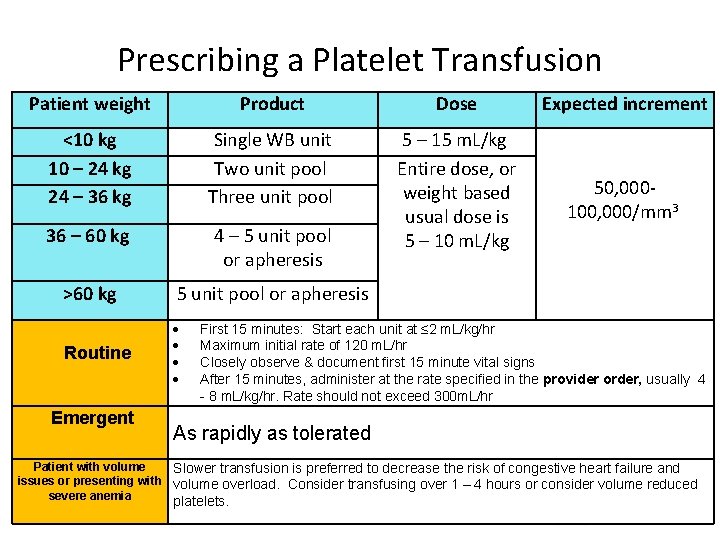
Prescribing a Platelet Transfusion Patient weight Product Dose Expected increment <10 kg 10 – 24 kg 24 – 36 kg Single WB unit Two unit pool Three unit pool 36 – 60 kg 4 – 5 unit pool or apheresis 5 – 15 m. L/kg Entire dose, or weight based usual dose is 5 – 10 m. L/kg 50, 000100, 000/mm 3 >60 kg 5 unit pool or apheresis Routine Emergent First 15 minutes: Start each unit at ≤ 2 m. L/kg/hr Maximum initial rate of 120 m. L/hr Closely observe & document first 15 minute vital signs After 15 minutes, administer at the rate specified in the provider order, usually 4 - 8 m. L/kg/hr. Rate should not exceed 300 m. L/hr As rapidly as tolerated Patient with volume Slower transfusion is preferred to decrease the risk of congestive heart failure and issues or presenting with volume overload. Consider transfusing over 1 – 4 hours or consider volume reduced severe anemia platelets.

Plasma • Use – Correct coagulopathy • Multiple factor deficiencies – Patients facing hemostatic challenge • Liver disease • • DIC • Warfarin reversal • • Massive transfusion • • Not used – – Specific factor deficiencies (!) Volume expansion Enhance wound healing Hypogammaglobulinemia All of the proteins of the coagulation, fibrinolytic and complement systems FFP frozen storage: ≤ -18° C or ≤ -65° C FFP expiry: 1 year FFP (thawed) expiry: 24 hours • Thawed plasma storage: 1 -6⁰C • Thawed plasma expiry: 5 days

Indication for plasma transfusion • Documented factor deficiency – AND active bleeding – OR about to have procedure • PT or PTT x 1. 5 times normal, or INR ≥ 1. 6 • Warfarin Reversal or Vitamin K deficiency – Significant bleeding or procedure eminent Fresh Frozen Plasma Thawed Plasma
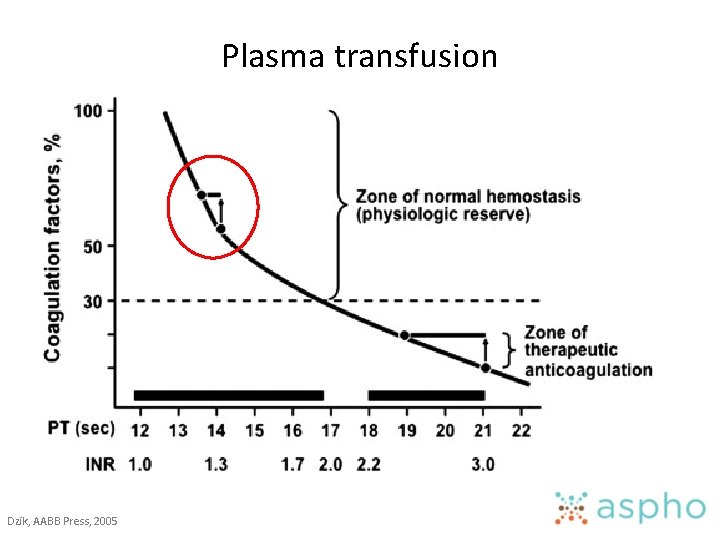
Plasma transfusion Dzik, AABB Press, 2005
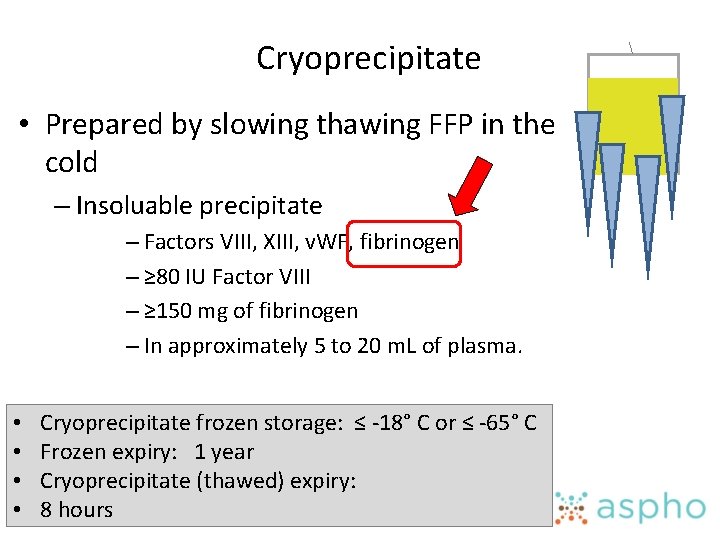
Cryoprecipitate • Prepared by slowing thawing FFP in the cold – Insoluable precipitate – Factors VIII, XIII, v. WF, fibrinogen – ≥ 80 IU Factor VIII – ≥ 150 mg of fibrinogen – In approximately 5 to 20 m. L of plasma. • • Cryoprecipitate frozen storage: ≤ -18° C or ≤ -65° C Frozen expiry: 1 year Cryoprecipitate (thawed) expiry: 8 hours

Cryoprecipitate Indications & Dosing • Indications – Hypofibrinogenemia – < 100 - 150 mg – DIC – Obstetrical bleeding – Massive transfusion • Dosing • One ”pool” = 5 -10 donor units = one adult sized patient dose • 2 – 5 m. L/kg • 1 bag / 10 kg of body weight
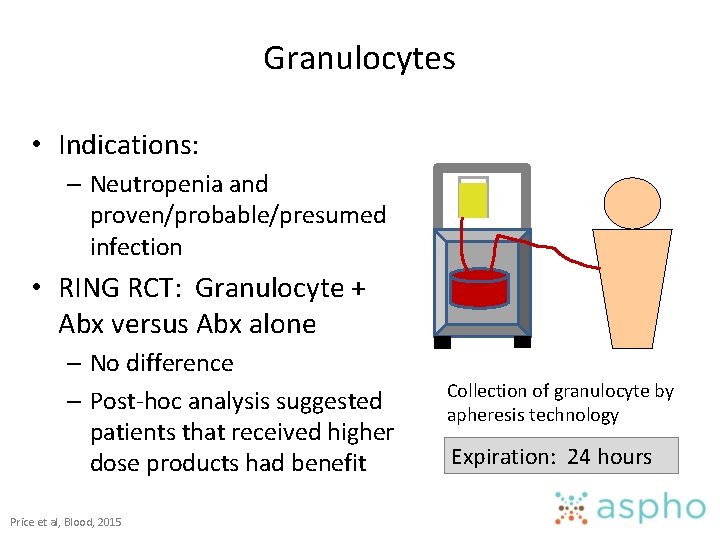
Granulocytes • Indications: – Neutropenia and proven/probable/presumed infection • RING RCT: Granulocyte + Abx versus Abx alone – No difference – Post-hoc analysis suggested patients that received higher dose products had benefit Price et al, Blood, 2015 Collection of granulocyte by apheresis technology Expiration: 24 hours
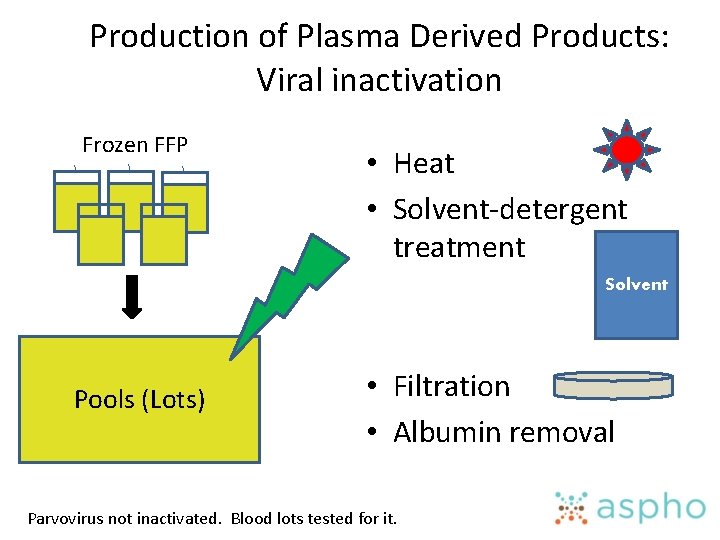
Production of Plasma Derived Products: Viral inactivation Frozen FFP • Heat • Solvent-detergent treatment Solvent Pools (Lots) • Filtration • Albumin removal Parvovirus not inactivated. Blood lots tested for it.
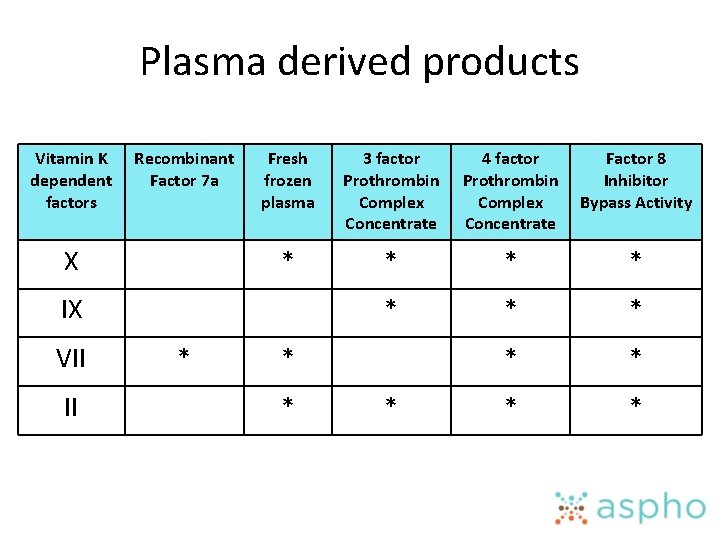
Plasma derived products Vitamin K dependent factors Recombinant Factor 7 a X Fresh frozen plasma 3 factor Prothrombin Complex Concentrate 4 factor Prothrombin Complex Concentrate Factor 8 Inhibitor Bypass Activity * * * IX VII II * *
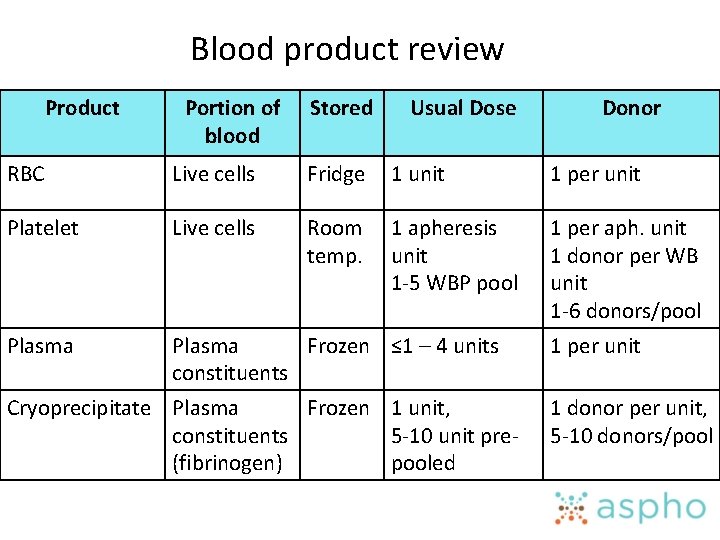
Blood product review Product Portion of blood Stored RBC Live cells Fridge Platelet Live cells Room temp. Donor 1 unit 1 per unit Red blood cell 1 apheresis unit 1 -5 WBP pool Plasma Usual Dose Plasma Frozen ≤ 1 – 4 units constituents Cryoprecipitate Plasma Frozen 1 unit, constituents 5 -10 unit pre(fibrinogen) pooled 1 per aph. unit 1 donor per WB unit 1 -6 donors/pool 1 per unit 1 donor per unit, 5 -10 donors/pool
Overview of Transfusion Medicine 1. Blood products • Indications 2. Selection of blood products • Compatibility testing 3. Adverse events • Transfusion transmitted disease • Transfusion reactions • Complications ?
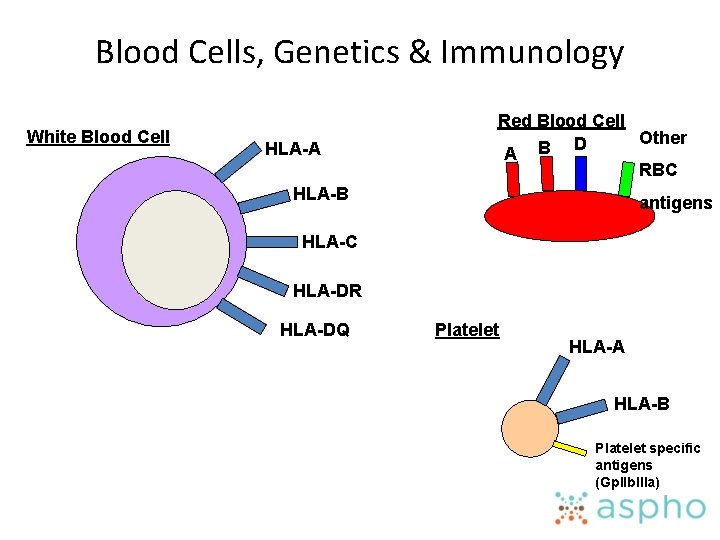
Blood Cells, Genetics & Immunology White Blood Cell HLA-A Red Blood Cell Other D B A RBC HLA-B antigens HLA-C HLA-DR HLA-DQ Platelet HLA-A HLA-B Platelet specific antigens (Gp. IIb. IIIa)

Blood Groups Red Blood Cell Other D B A RBC antigens • Genetically related antigens on red blood cell surface • There are 30 blood groups systems >300 different antigens • Most well known blood group? ABO Rh (Rhesus) • Second most well known? • Other clinically important ones? Duffy, Kidd, Kell
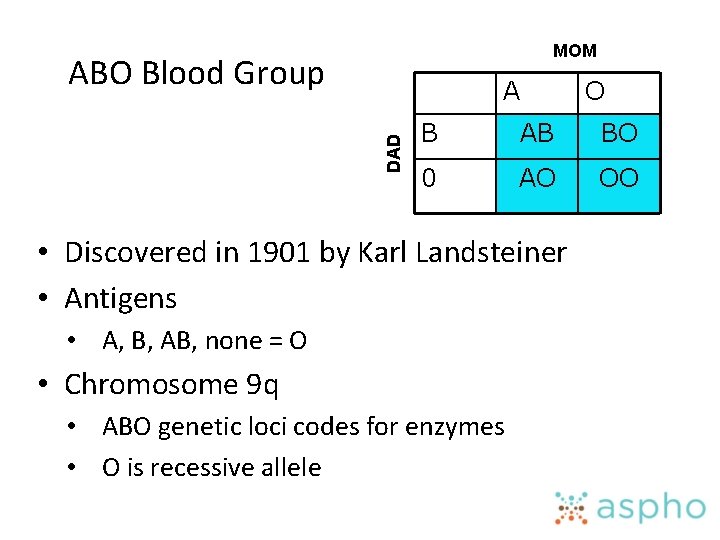
MOM ABO Blood Group DAD A O B AB BO 0 AO OO • Discovered in 1901 by Karl Landsteiner • Antigens • A, B, AB, none = O • Chromosome 9 q • ABO genetic loci codes for enzymes • O is recessive allele
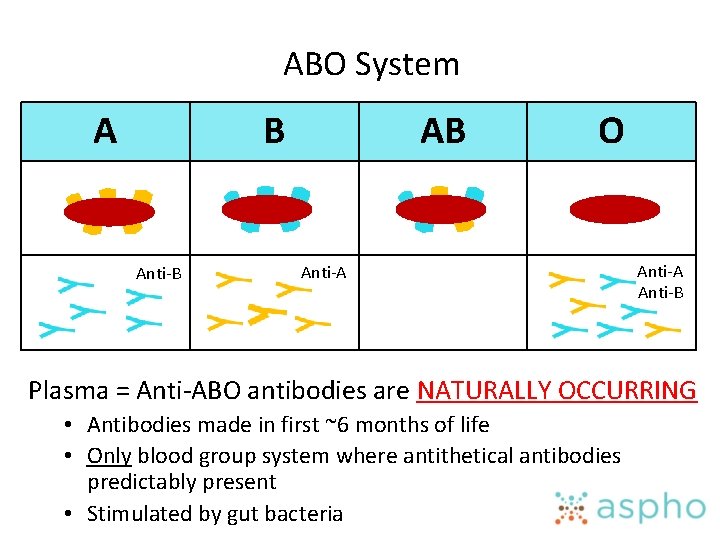
ABO System A B Anti-B AB O Anti-A Anti-B Plasma = Anti-ABO antibodies are NATURALLY OCCURRING • Antibodies made in first ~6 months of life • Only blood group system where antithetical antibodies predictably present • Stimulated by gut bacteria
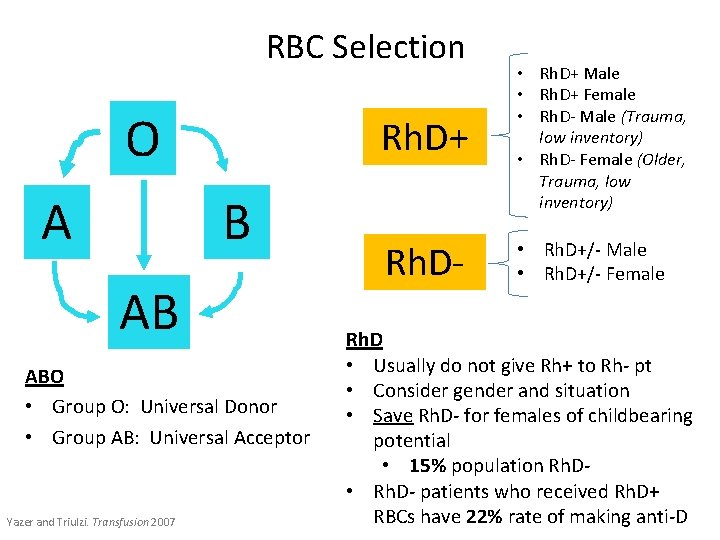
RBC Selection O A B AB ABO • Group O: Universal Donor • Group AB: Universal Acceptor Yazer and Triulzi. Transfusion 2007 Rh. D+ • Rh. D+ Male • Rh. D+ Female • Rh. D- Male (Trauma, low inventory) • Rh. D- Female (Older, Trauma, low inventory) Rh. D- • Rh. D+/- Male • Rh. D+/- Female Rh. D • Usually do not give Rh+ to Rh- pt • Consider gender and situation • Save Rh. D- for females of childbearing potential • 15% population Rh. D • Rh. D- patients who received Rh. D+ RBCs have 22% rate of making anti-D
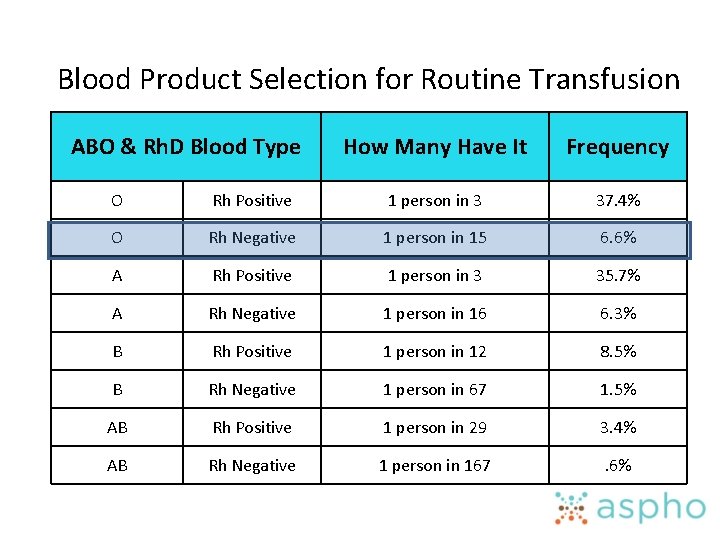
Blood Product Selection for Routine Transfusion ABO & Rh. D Blood Type How Many Have It Frequency O Rh Positive 1 person in 3 37. 4% O Rh Negative 1 person in 15 6. 6% A Rh Positive 1 person in 3 35. 7% A Rh Negative 1 person in 16 6. 3% B Rh Positive 1 person in 12 8. 5% B Rh Negative 1 person in 67 1. 5% AB Rh Positive 1 person in 29 3. 4% AB Rh Negative 1 person in 167 . 6%
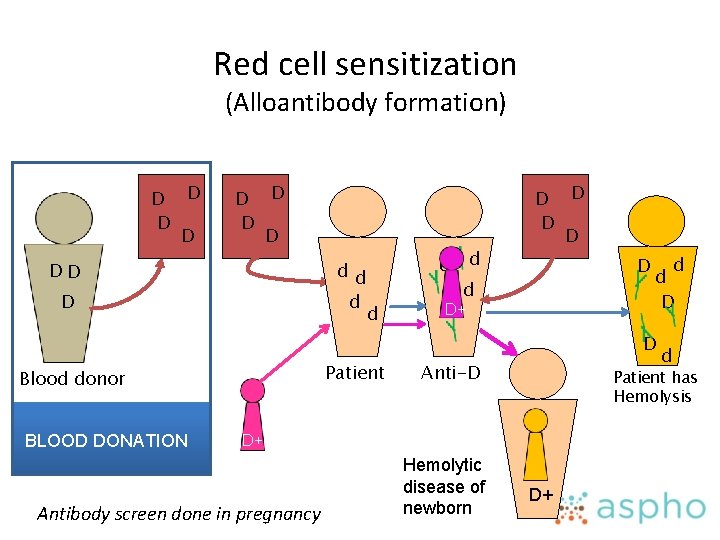
Red cell sensitization (Alloantibody formation) D D D D DD dd d D d d d D D d d d. D D d d D+ D Patient Blood donor BLOOD DONATION Anti-D Patient has Hemolysis D+ Antibody screen done in pregnancy Hemolytic disease of newborn d D+
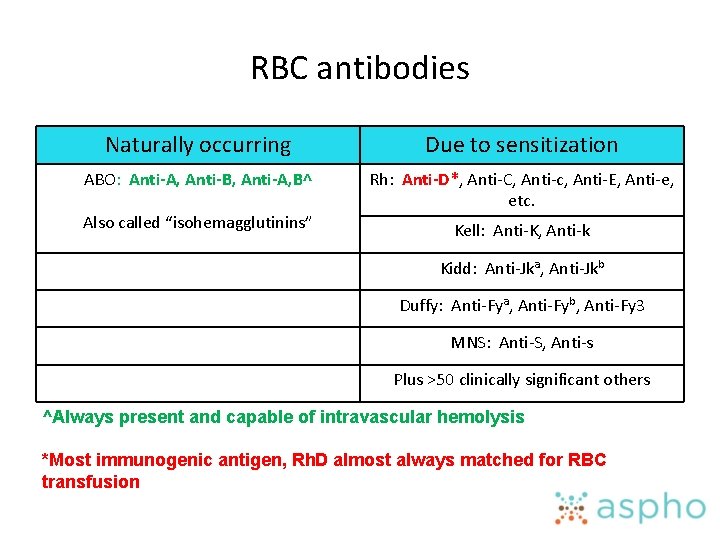
RBC antibodies Naturally occurring Due to sensitization ABO: Anti-A, Anti-B, Anti-A, B^ Rh: Anti-D*, Anti-C, Anti-c, Anti-E, Anti-e, etc. Also called “isohemagglutinins” Kell: Anti-K, Anti-k Kidd: Anti-Jka, Anti-Jkb Duffy: Anti-Fya, Anti-Fyb, Anti-Fy 3 MNS: Anti-S, Anti-s Plus >50 clinically significant others ^Always present and capable of intravascular hemolysis *Most immunogenic antigen, Rh. D almost always matched for RBC transfusion
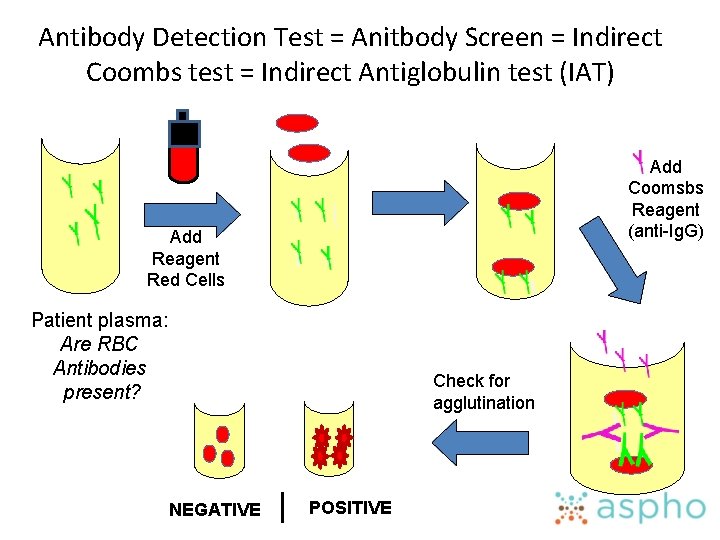
Antibody Detection Test = Anitbody Screen = Indirect Coombs test = Indirect Antiglobulin test (IAT) Add Coomsbs Reagent (anti-Ig. G) Add Reagent Red Cells Patient plasma: Are RBC Antibodies present? Check for agglutination NEGATIVE POSITIVE

Reagent RBC Antibody detection test: Antigram 1 2 3 Patient sera with reagent red blood cells with known antigen expression check for agglutination

Antibody identification: Antigram Extends time for crossmatch RBCs to be ready Patient sera with reagent red blood cells with known antigen expression check for agglutination

Type and Screen Type and Cross • Blood Type: ABO, Rh • Type & Screen PLUS – Reserve # RBC units ordered • Antibody Screen – Crossmatch units – Test patient plasma for • Multiple antibodies or Ab to high presence of RBC frequency antigens may require: alloantibodies – Uncommon RBC units: ↑time – If RBC antibodies present, – Rare RBC units: Frozen antigen negative RBC are YES: required Do not give • Sample active (expiration) – 72 hours NO: Ok to – If <4 months active for one give hospitalization usually
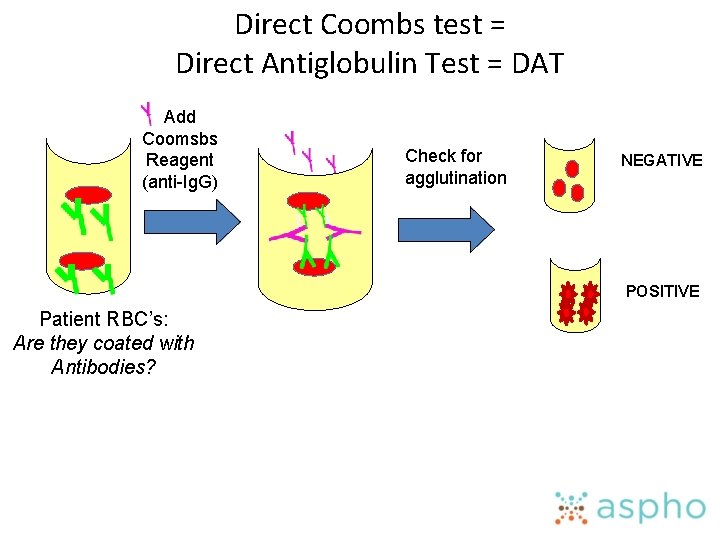
Direct Coombs test = Direct Antiglobulin Test = DAT Add Coomsbs Reagent (anti-Ig. G) Check for agglutination NEGATIVE POSITIVE Patient RBC’s: Are they coated with Antibodies?

Platelet product compatibility ABO • • • Plasma compatibility: Concern of ABO HTR Universal donor: AB When giving incompatible plasma (Group O donor) to Group A pt • Blood bank will choose low titer anti-A or volume reduce platelet product Rh. D Patient ABO Compatible Plasma O O, A, B, AB A A, AB B B, AB AB AB • Not expressed on platelets • Very small amount of RBC in platelet product • Rh. D- patients who receive Rh. D+ platelets have 1. 4% rate of forming anti-D • Rh. Ig can prevent; often not used in adult or low risk settings Cid et al, BJH, 2014
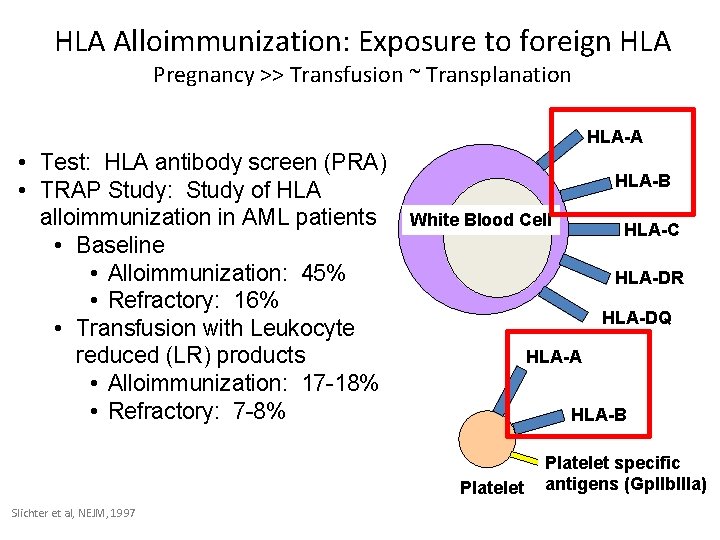
HLA Alloimmunization: Exposure to foreign HLA Pregnancy >> Transfusion ~ Transplanation HLA-A • Test: HLA antibody screen (PRA) • TRAP Study: Study of HLA alloimmunization in AML patients • Baseline • Alloimmunization: 45% • Refractory: 16% • Transfusion with Leukocyte reduced (LR) products • Alloimmunization: 17 -18% • Refractory: 7 -8% HLA-B White Blood Cell HLA-DR HLA-DQ HLA-A HLA-B Platelet Slichter et al, NEJM, 1997 HLA-C Platelet specific antigens (Gp. IIb. IIIa)
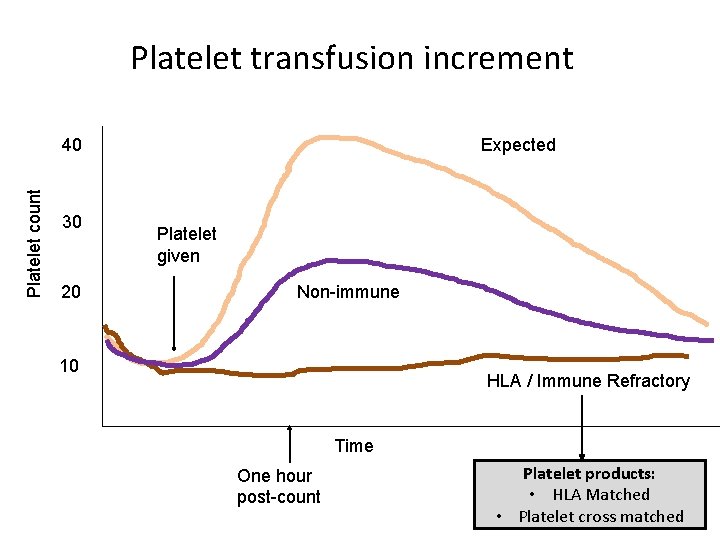
Platelet transfusion increment Platelet count 40 30 20 Expected Platelet given Non-immune 10 HLA / Immune Refractory Time One hour post-count Platelet products: • HLA Matched • Platelet cross matched
Overview of Transfusion Medicine 1. Blood products • Indications 2. Selection of blood products • Compatibility testing 3. Adverse events • Transfusion transmitted disease • Transfusion reactions • Complications ?
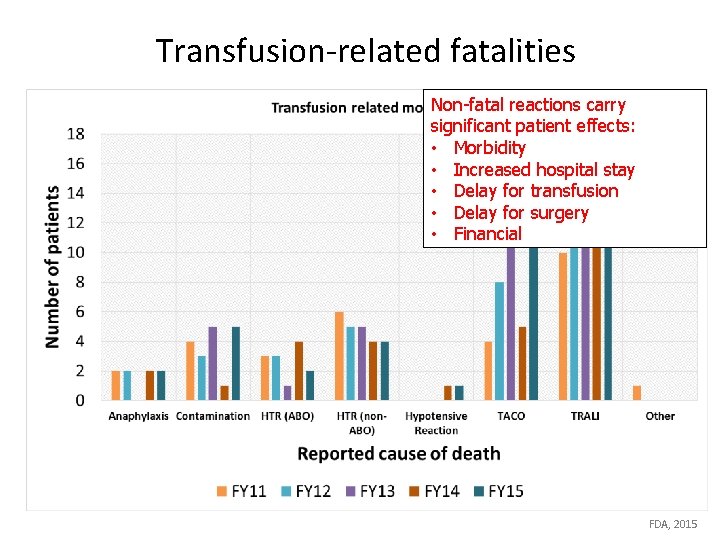
Transfusion-related fatalities Non-fatal reactions carry significant patient effects: • Morbidity • Increased hospital stay • Delay for transfusion • Delay for surgery • Financial FDA, 2015
Transfusion Transmitted Disease Republished with permission of John Wiley & Sons, from Transfusion, cover image, 49, 2 S, 2009, permission conveyed through Copyright Clearance Center, Inc.
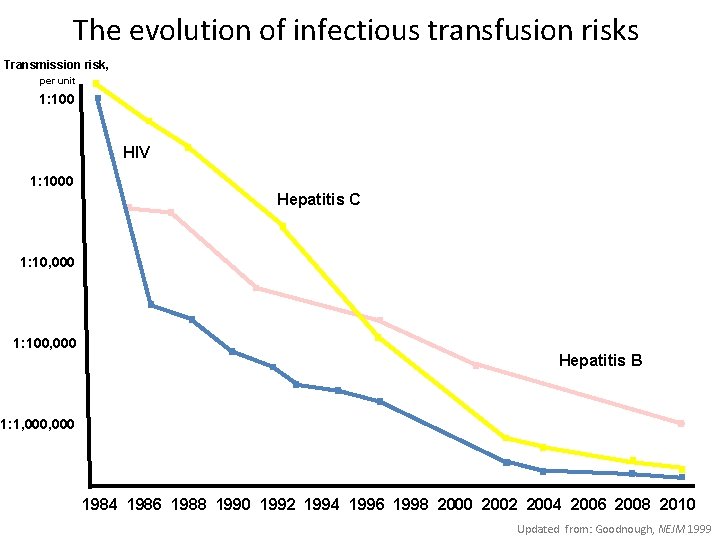
The evolution of infectious transfusion risks Transmission risk, per unit 1: 100 HIV 1: 1000 Hepatitis C 1: 10, 000 1: 100, 000 Hepatitis B 1: 1, 000 1984 1986 1988 1990 1992 1994 1996 1998 2000 2002 2004 2006 2008 2010 Updated from: Goodnough, NEJM 1999

Residual infection risks • HIV – 1: 2. 9 – 4. 7 million • Hepatitis C – 1: 1, 500, 000 • • – 1: 2, 000 Wide confidence intervals 1: 200, 000 Motor vehicle accident (injury) • • HTLV I & II 1: 5, 000 Anesthesia (death) • • Hepatitis B – 1: 250, 000 -1. 5 mil Lightening strike (death) • • 1: 1, 000 The risk of contracting an infection during a stay at a U. S. hospital • • 1: 15
Only volunteer donors Replacement blood donors wait to donate on behalf of a specific patient that required blood during their hospitalization, Ghana.

Blood donor questions & testing Screening for donor risk factors (1) Disease we test for • HIV, HBV, HCV, Syphilis, Chagas, WNV, HTLV • Sepsis (2) Disease we cannot test for • • variant Jacob-Creutzfeldt “classical” Jacob-Creutzfeldt Malaria (we don’t test for) Diseases we don’t know about

Transfusion associated CMV • 50% or more of blood donors CMV are seropositive – ~1% thought to be infectious – Usually transfusion associated CMV of no clinical consequence • Certain categories of immunocompromised patients can have progressive disease – Fetusus / newborns • Pregnant mothers – Bone marrow transplant patients / candidates – Solid organ transplant patients – Seronegative AIDS patients

CMV Safe Blood Products • Prevent transfusion associated-CMV disease in patients at risk • ”CMV safe” products made by two methods – Donor negative for CMV antibodies (CMV negative) OR – Blood product leukoreduced • CMV cellular restricted pathogen

Directed donation • Appealing for families because they (or the patient) hopes the directed donation will be • Safer than the community blood supply • Decrease the number of donor exposures • Volunteer vs. Directed • No difference in deferral rates or test results • No difference in HIV+ • DD higher hepatitis+ • Not able to be used emergently • Irradiation required: GVHD Cordell, Transfusion, 1986 Starkey, JAMA, 1989

Pediatric hospital study: Directed donation • Pediatric center with >22, 000 blood components transfused/year – Directed donation requests (219/year) • 99 excluded from donation – Compatibility, urgency of need, parental risk factors or illness • 133 of 219 requests had blood available (61%) Wales, J Pediatric Surgery, 2001
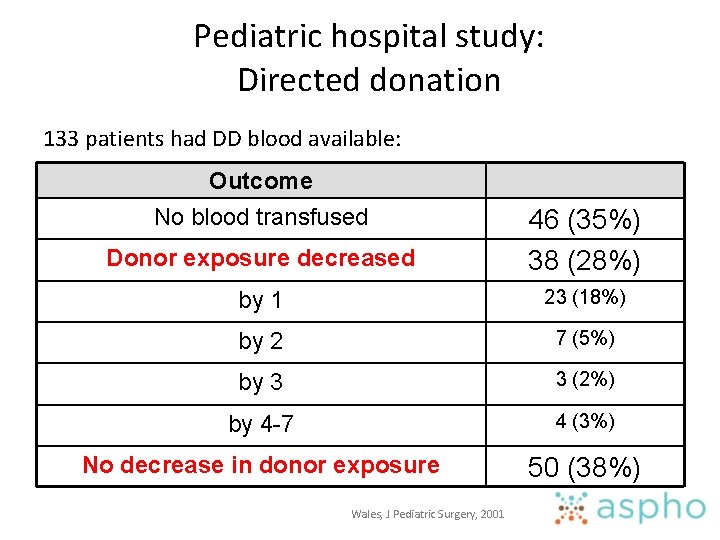
Pediatric hospital study: Directed donation 133 patients had DD blood available: Outcome No blood transfused Donor exposure decreased 46 (35%) 38 (28%) by 1 23 (18%) by 2 7 (5%) by 3 3 (2%) by 4 -7 4 (3%) No decrease in donor exposure 50 (38%) Wales, J Pediatric Surgery, 2001
Overview of Transfusion Medicine 1. Blood products • Indications 2. Selection of blood products • Compatibility testing 3. Adverse events • Transfusion transmitted disease • Transfusion reactions • Complications ?

Relative Risk of Transfusion Reactions • Allergic, hives – 1: 500 • Febrile – 1: 1000 • Sepsis – 1: 10, 000 • TRALI – 1: 5000 to 1: 100, 000 • Hemolytic – 1: 250, 000 to 1: 600, 000 What’s the Risk?

Transfusion Reactions • Most importantly, what do you do? – STOP the transfusion – Keep the IV line open – Perform clerical check of product and patient – Signs & Symptoms • Follow and document vital signs • Supportive treatment – Anti-pyretics, anti-histamines, anti-inflammatories • Send for pertinent labs – Note type of product – Notify and send patient tube and product to blood bank
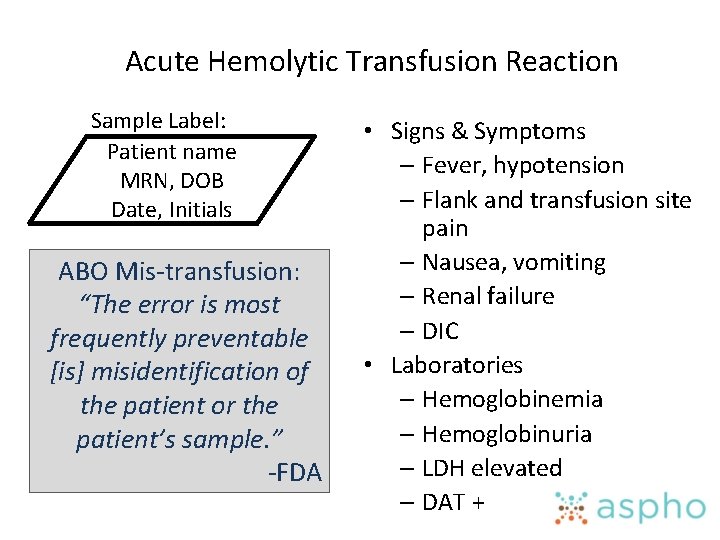
Acute Hemolytic Transfusion Reaction Sample Label: Patient name MRN, DOB Date, Initials ABO Mis-transfusion: “The error is most frequently preventable [is] misidentification of the patient or the patient’s sample. ” -FDA • Signs & Symptoms – Fever, hypotension – Flank and transfusion site pain – Nausea, vomiting – Renal failure – DIC • Laboratories – Hemoglobinemia – Hemoglobinuria – LDH elevated – DAT +
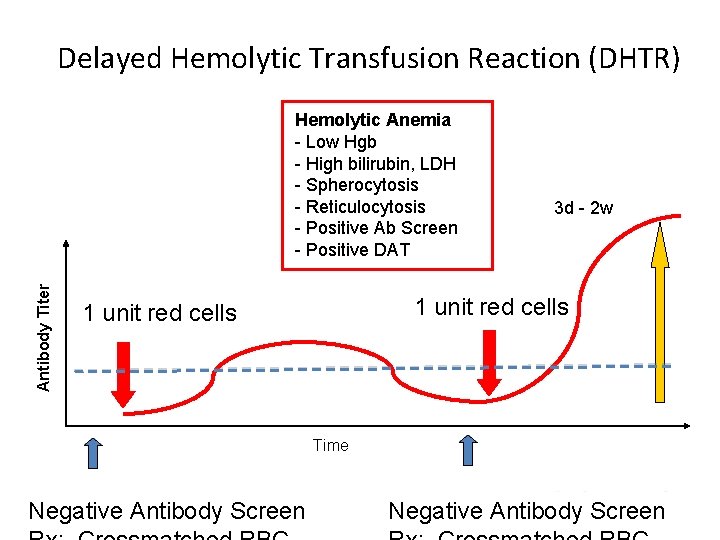
Delayed Hemolytic Transfusion Reaction (DHTR) Antibody Titer Hemolytic Anemia - Low Hgb - High bilirubin, LDH - Spherocytosis - Reticulocytosis - Positive Ab Screen - Positive DAT 3 d - 2 w 1 unit red cells Time Negative Antibody Screen
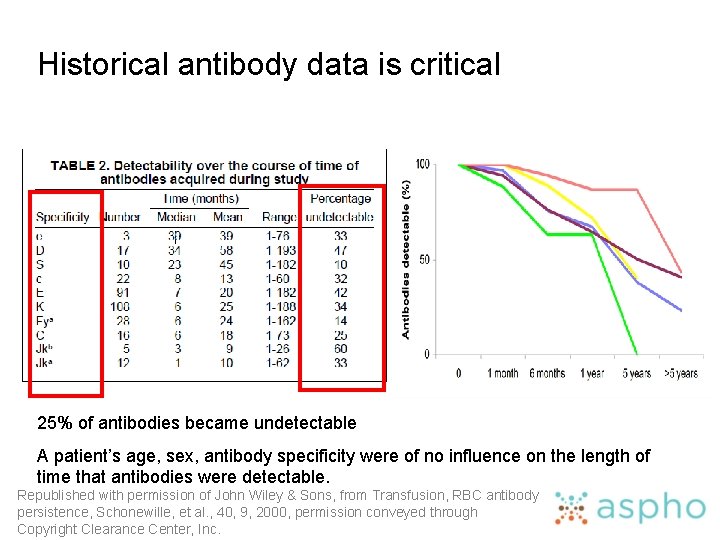
Historical antibody data is critical 25% of antibodies became undetectable A patient’s age, sex, antibody specificity were of no influence on the length of time that antibodies were detectable. Republished with permission of John Wiley & Sons, from Transfusion, RBC antibody persistence, Schonewille, et al. , 40, 9, 2000, permission conveyed through Copyright Clearance Center, Inc.
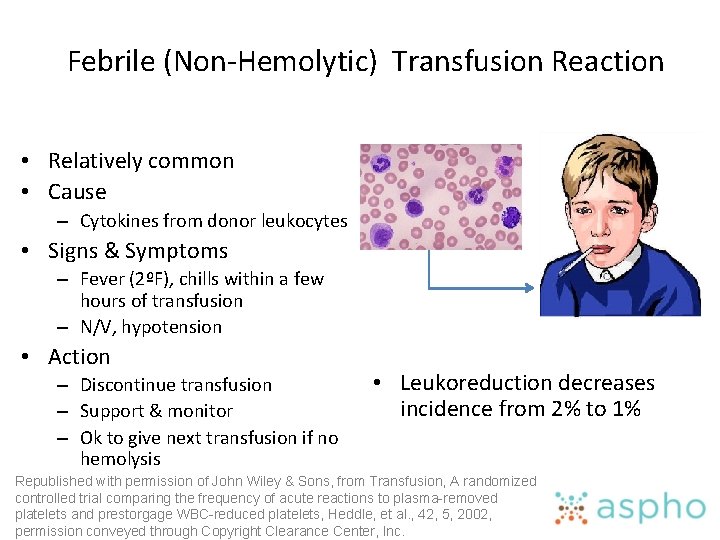
Febrile (Non-Hemolytic) Transfusion Reaction • Relatively common • Cause – Cytokines from donor leukocytes • Signs & Symptoms – Fever (2ºF), chills within a few hours of transfusion – N/V, hypotension • Action – Discontinue transfusion – Support & monitor – Ok to give next transfusion if no hemolysis • Leukoreduction decreases incidence from 2% to 1% Republished with permission of John Wiley & Sons, from Transfusion, A randomized controlled trial comparing the frequency of acute reactions to plasma-removed platelets and prestorgage WBC-reduced platelets, Heddle, et al. , 42, 5, 2002, permission conveyed through Copyright Clearance Center, Inc.
Leukoreduction • Filtration of blood products to remove WBC – Decrease risk of febrile rections from 2% 1% – Decrease risk of allosensitization – Decrease risk of CMV transmission • Cellular restricted pathogen Heddle et al, TRANSFUSION, 2002 FILTER
Urticarial, Allergic & Anaphylaxis URTICARIAL (Common) ANAPHYLACTIC (Rare) • Hypersensitivity Reactions – Proteins in donor plasma cause hypersensitivity reaction in recipient
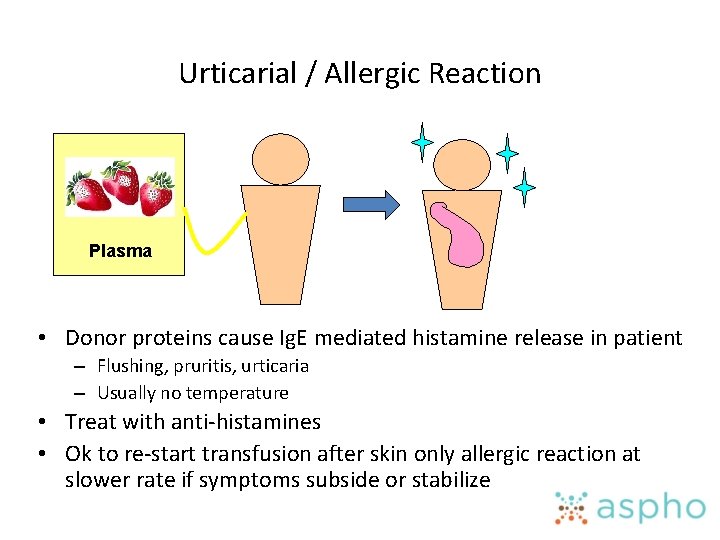
Urticarial / Allergic Reaction Plasma • Donor proteins cause Ig. E mediated histamine release in patient – Flushing, pruritis, urticaria – Usually no temperature • Treat with anti-histamines • Ok to re-start transfusion after skin only allergic reaction at slower rate if symptoms subside or stabilize
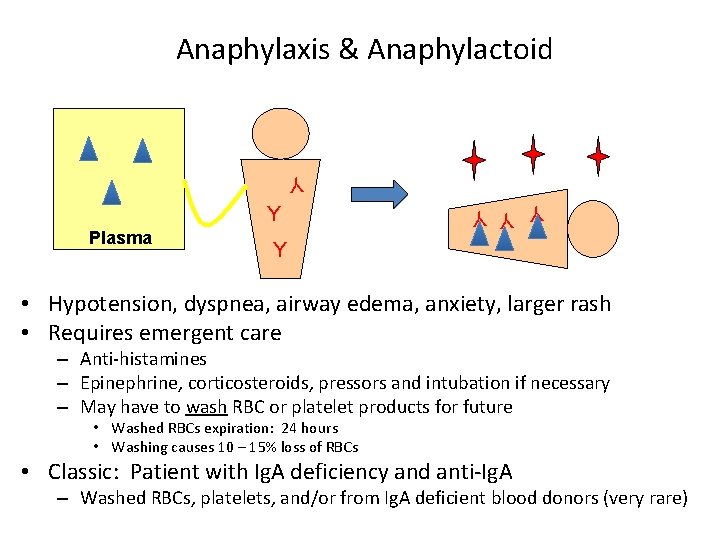
Anaphylaxis & Anaphylactoid Y Plasma Y Y Y • Hypotension, dyspnea, airway edema, anxiety, larger rash • Requires emergent care – Anti-histamines – Epinephrine, corticosteroids, pressors and intubation if necessary – May have to wash RBC or platelet products for future • Washed RBCs expiration: 24 hours • Washing causes 10 – 15% loss of RBCs • Classic: Patient with Ig. A deficiency and anti-Ig. A – Washed RBCs, platelets, and/or from Ig. A deficient blood donors (very rare)

Sepsis • Blood components contaminated by bacteria – Blood donor skin flora – Blood donor unrecognized bacteremia – Environmental or product handling during processing • Most common in platelets because stored at room temp – Up to 1 in 1, 300 platelet transfusions • Red blood cell contamination uncommon – Gram negative rods, Yersinia spp. • Always let blood bank know if septic reaction suspected so products can be cultured
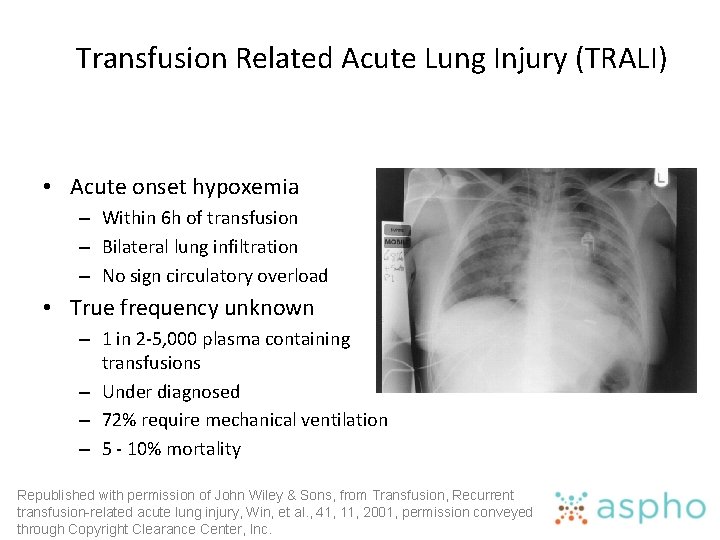
Transfusion Related Acute Lung Injury (TRALI) • Acute onset hypoxemia – Within 6 h of transfusion – Bilateral lung infiltration – No sign circulatory overload • True frequency unknown – 1 in 2 -5, 000 plasma containing transfusions – Under diagnosed – 72% require mechanical ventilation – 5 - 10% mortality Republished with permission of John Wiley & Sons, from Transfusion, Recurrent transfusion-related acute lung injury, Win, et al. , 41, 11, 2001, permission conveyed through Copyright Clearance Center, Inc.
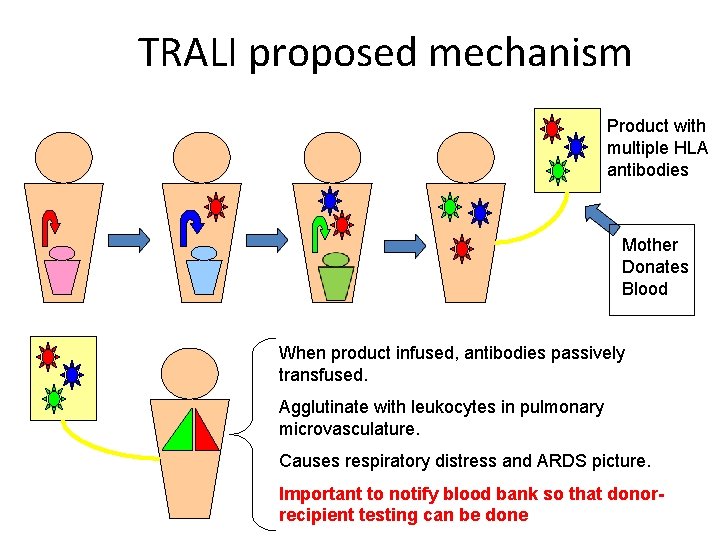
TRALI proposed mechanism Product with multiple HLA antibodies Mother Donates Blood When product infused, antibodies passively transfused. Agglutinate with leukocytes in pulmonary microvasculature. Causes respiratory distress and ARDS picture. Important to notify blood bank so that donorrecipient testing can be done
Overview of Transfusion Medicine 1. Blood products • Indications 2. Selection of blood products • Compatibility testing 3. Adverse events • Transfusion transmitted disease • Transfusion reactions • Complications ?

Transfusion Associated GVHD • Engraftment and proliferation of donor lymphocytes in transfusion recipient. • Who’s at risk? • Immunocompetent patient transfused with HLA haploidentical product (blood relative) • Neonatal exchange transfusions • Severely immunocompromised patients • Donor cells attack • Hematopoietic cells refractory pancytopenia • Other organ systems: Fever, enterocolitis, rash, hepatitis • Usually fatal: 90 -100%
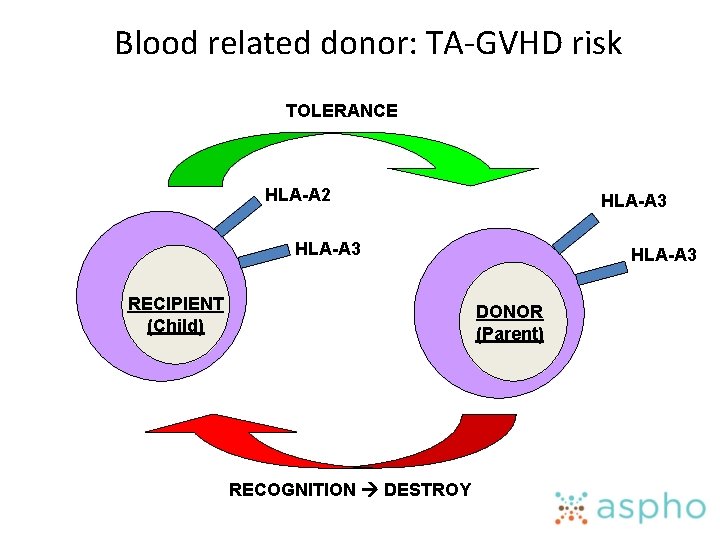
Blood related donor: TA-GVHD risk TOLERANCE HLA-A 2 HLA-A 3 RECIPIENT (Child) HLA-A 3 DONOR (Parent) RECOGNITION DESTROY

Irradiation • Protects patient from Transfusion Associated Graft Versus Host Disease (TA-GVHD) • Inactivates lymphocyte division in blood product • 2, 500 c. Gy (cesium) applied to blood product • Patients that need irradiated blood products: • • • Fetusus / newborns (usually up to 4 months of age) Bone marrow transplant patients / candidates Hematological malignancy Congenital immunodeficiency (Di. George Syndrome, etc) Blood relative blood donations
Risk: Massive transfusion and K+ K+ K+ Republished with permission from Carl (Rod) Nave, Department of Physics and Astronomy, Georgia State University, hyperphysics. phy-astr. gsu. edu
![[K+] during storage [K+] m. Eq/L Irradiation Red cell product age (days) [K+] during storage [K+] m. Eq/L Irradiation Red cell product age (days)](http://slidetodoc.com/presentation_image_h2/a3131e23c47f5b92effc9912a547af7e/image-74.jpg)
[K+] during storage [K+] m. Eq/L Irradiation Red cell product age (days)
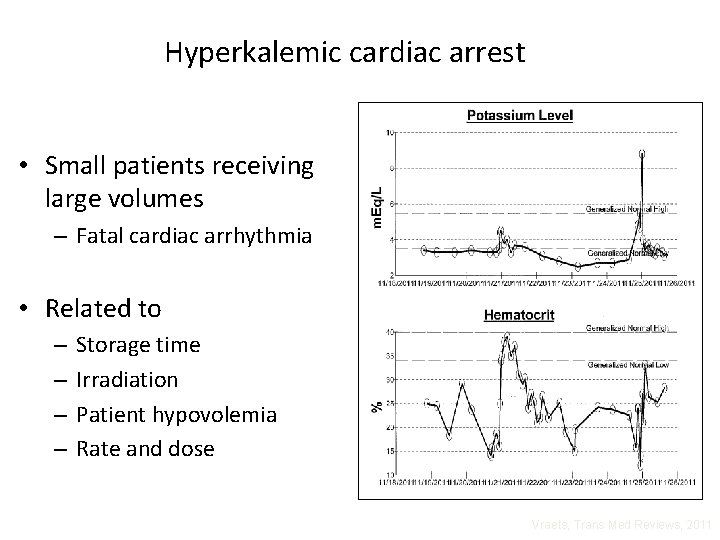
Hyperkalemic cardiac arrest • Small patients receiving large volumes – Fatal cardiac arrhythmia • Related to – – Storage time Irradiation Patient hypovolemia Rate and dose Vraets, Trans Med Reviews, 2011

Conclusions: Transfusion Medicine 1. Blood products • Indications, dosing 2. Selection of blood products • Compatibility testing • Antibodies, crossmatching 3. Adverse events • Transfusion transmitted disease • Transfusion reactions • Complications

Thank you We did it !
- Slides: 77