Header Brain Tumors Kenneth Subhead J Cohen MD

Header Brain Tumors Kenneth Subhead J. Cohen, MD, MBA
2 Background 2 nd most common group of cancers in children after the leukemias Most common group of solid neoplasms Estimated 4, 830 new cases in 2017 (0 -14 yrs old) ~ 73. 8% 5 year survival (0 -19 yrs of age)
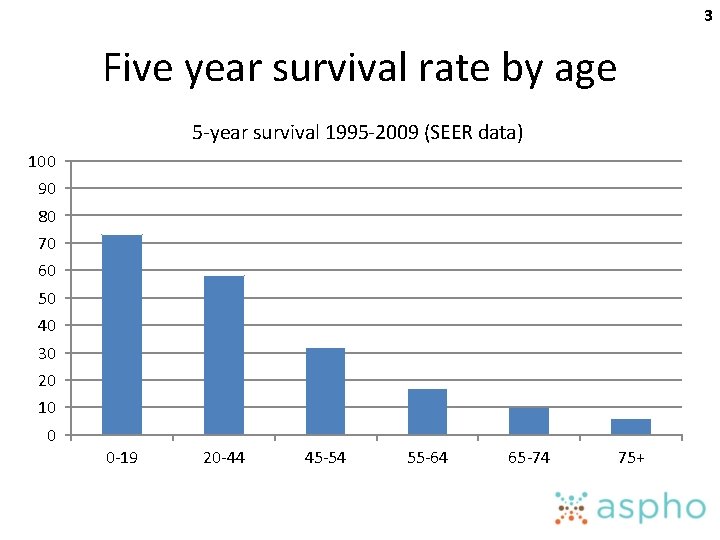
3 Five year survival rate by age 5 -year survival 1995 -2009 (SEER data) 100 90 80 70 60 50 40 30 20 10 0 0 -19 20 -44 45 -54 55 -64 65 -74 75+

4 Tumor Histology by Age (yrs) Most Common Histology Second Most Common Histology 0 -4 Embryonal/Medulloblastoma Pilocytic Astrocytoma 5 -9 Pilocytic Astrocytoma Diffuse astrocytoma, NOS 10 -14 Pilocytic Astrocytoma Neuronal/glial 15 -19 Pituitary Pilocytic astrocytoma 20 -34 Pituitary Meningioma 35 -44 Meningioma Pituitary 45+ Meningioma Glioblastoma

Brain Tumor Nomenclature: Histologic Classification NEUROGLIAL CELLS Astrocytes Oligodendrocytes Ependyma Choroid Plexus NEURONAL/EMBRYONAL CELLS Other stuff (e. g. microglia) 5

Brain Tumor Classification Former WHO Classification TUMORS OF NEUROEPITHELIAL TISSUE Tumors of Peripheral Nerves Tumors of the Meninges Lymphomas and Haemopoietic Neoplasms GERM CELL TUMORS OF THE SELLAR REGION Metastatic Tumors 6

Former WHO Classification: Tumors of Neuroepithelial Tissue Astrocytic tumors Oligodendroglial tumors Ependymal tumors Choroid plexus tumors Other neuroepithelial tumors Neuronal and mixed neuronal-glial tumors Tumors of the pineal region Embryonal tumors 7

WHO Classification 2016 (pediatric relevant categories) • • • Diffuse astrocytic and oligodendroglial tumors Other astrocytic tumors Ependymal tumors Choroid plexus tumors Neuronal and mixed neuronal-glial tumors Tumors of the pineal region Emryonal tumors Histiocytic tumors Germ cell tumors Tumors of the sellar region 8

Diffuse astrocytic and oligodendroglial tumors Diffuse astrocytoma Anaplastic astrocytoma Glioblastoma Diffuse midline glioma, H 3 K 27 M-mutant Oligodendroglioma Anaplastic oligodendroglioma 9

10 Low Grade vs. High Grade “Gliomas” “GLIOMAS” Astrocytomas: Pilocytic (“Low Grade”, WHO 1) Diffuse Astrocytoma (“Low Grade”, WHO 2) Anaplastic Astrocytoma (“High Grade”, WHO 3) Glioblastoma Multiforme (“High Grade”, WHO 4)

11 Other astrocytic tumors Pilocytic astrocytoma (includes most optic pathway tumors) Pilomyxoid astrocytoma Subependymal giant cell astrocytoma Pleomorphic xanthroastrocytoma (PXA) Anaplastic PXA

12 Ependymal tumors Myxopapillary ependymoma Ependymoma RELA fusion-positive Anaplastic ependymoma

13 Choroid plexus tumors Choroid plexus papilloma Atypical choroid plexus papilloma Choroid plexus carcinoma

14 Neuronal and mixed neuronal-glial tumors Dysembryoplastic neuroepithelial tumor (DNET) Ganglioma Desmoplastic infantile astrocytoma and ganglioma (DIG)

15 Tumours of the pineal region Pineal parenchymal tumor of intermediate differentiation Pineoblastoma

16 Embryonal tumors: Medulloblastomas, genetically defined Medulloblastoma, WNT-activated Medulloblastoma, SHH-activated and TP 53 -mutant Medulloblastoma, SHH-activated and TP 53 -wildtype Medulloblastoma, non-WNT/non-SHH Medulloblastoma, group 3 Medulloblastoma, group 4 Medulloblastoma, histologically defined Medulloblastoma, classic Medulloblastoma, desmoplastic/nodular Medulloblastoma with extensive nodularity Medulloblastoma, large cell/anaplastic Medulloblastoma, NOS

17 Embryonal tumors Embryonal tumor with multilayered rosettes, C 19 MCaltered Medulloepithelioma CNS neuroblastoma CNS ganglioneuroblastoma CNS embryonal tumor, NOS (aka PNET) Atypical teratoid/rhaboid tumor

18 Histiocytic tumors LCH Erheim-Chester disease Rosai-Dorfman disease Juvenile xanthogranuloma Histocytic sarcoma

19 Germ cell tumors Germinoma Embryonal carcinoma Yolk sac tumor Choriocarcinoma Teratoma Mature teratoma Immature teratoma Mixed germ cell tumor

20 Tumors of the sellar region Craniopharyngioma Adamantinomatous craniopharyngioma Papillary craniopharyngioma
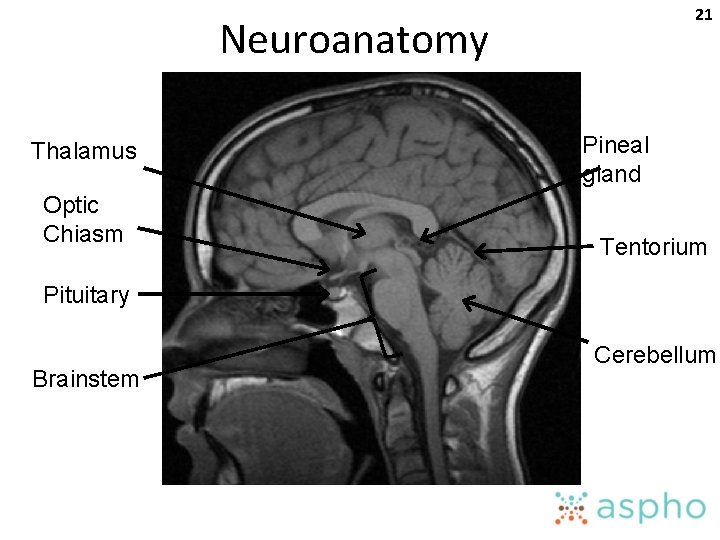
21 Neuroanatomy Thalamus Optic Chiasm Pineal gland Tentorium Pituitary Brainstem Cerebellum
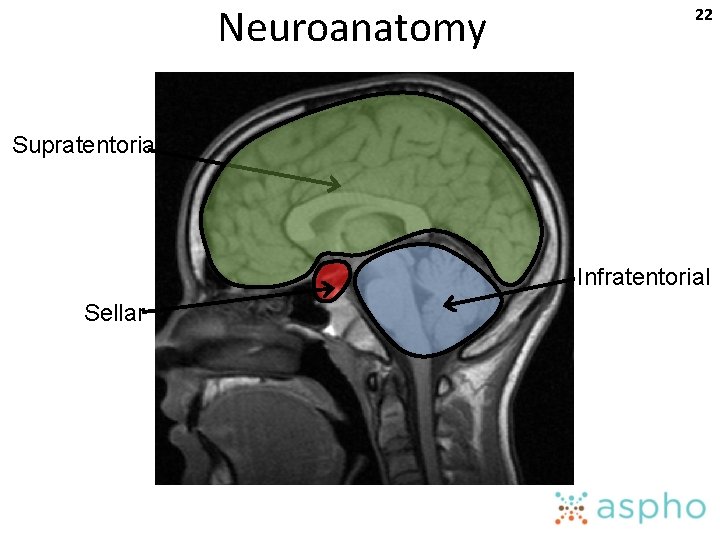
Neuroanatomy 22 Supratentorial Infratentorial Sellar
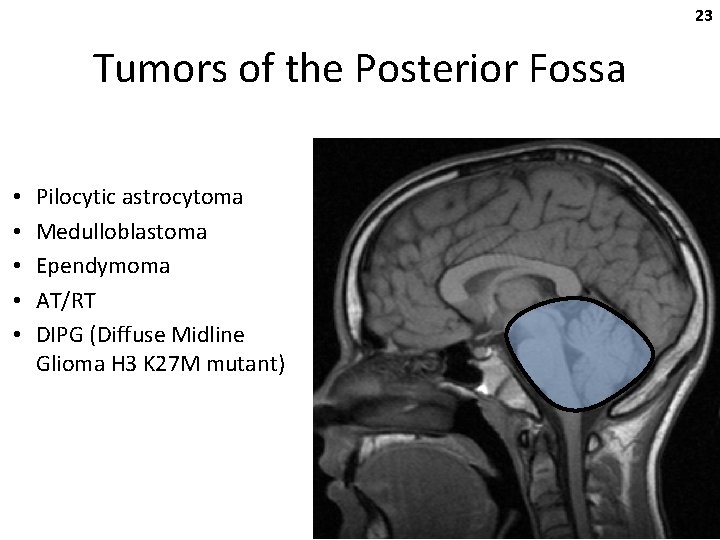
23 Tumors of the Posterior Fossa • • • Pilocytic astrocytoma Medulloblastoma Ependymoma AT/RT DIPG (Diffuse Midline Glioma H 3 K 27 M mutant)
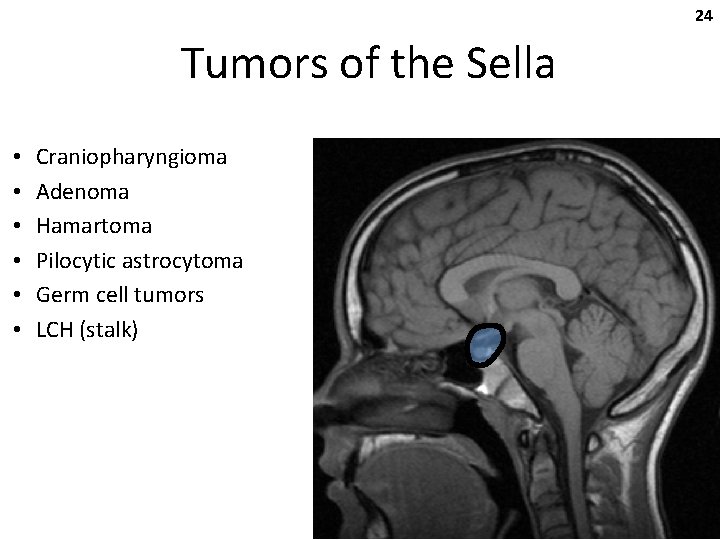
24 Tumors of the Sella • • • Craniopharyngioma Adenoma Hamartoma Pilocytic astrocytoma Germ cell tumors LCH (stalk)
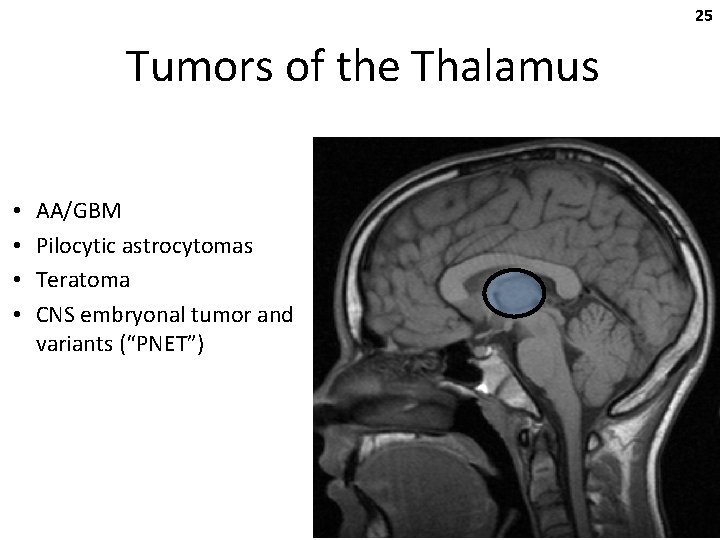
25 Tumors of the Thalamus • • AA/GBM Pilocytic astrocytomas Teratoma CNS embryonal tumor and variants (“PNET”)
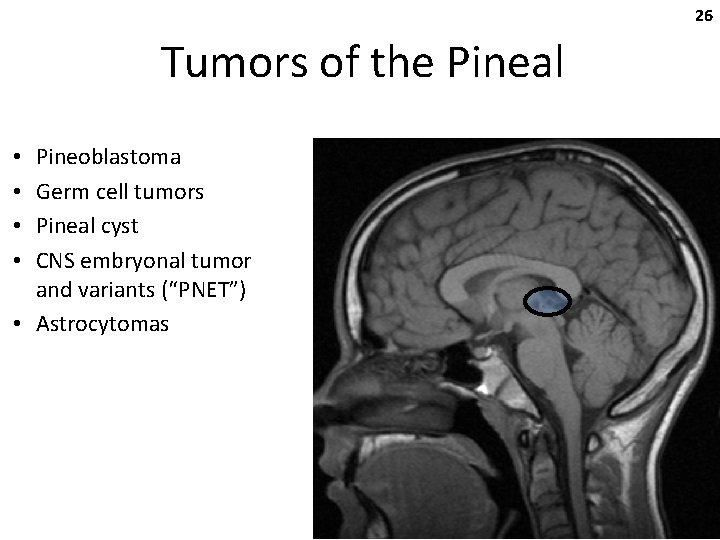
26 Tumors of the Pineal Pineoblastoma Germ cell tumors Pineal cyst CNS embryonal tumor and variants (“PNET”) • Astrocytomas • •
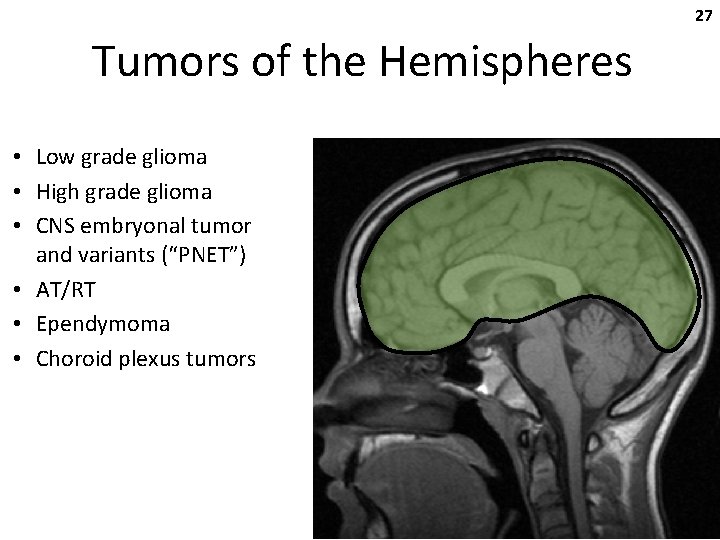
27 Tumors of the Hemispheres • Low grade glioma • High grade glioma • CNS embryonal tumor and variants (“PNET”) • AT/RT • Ependymoma • Choroid plexus tumors
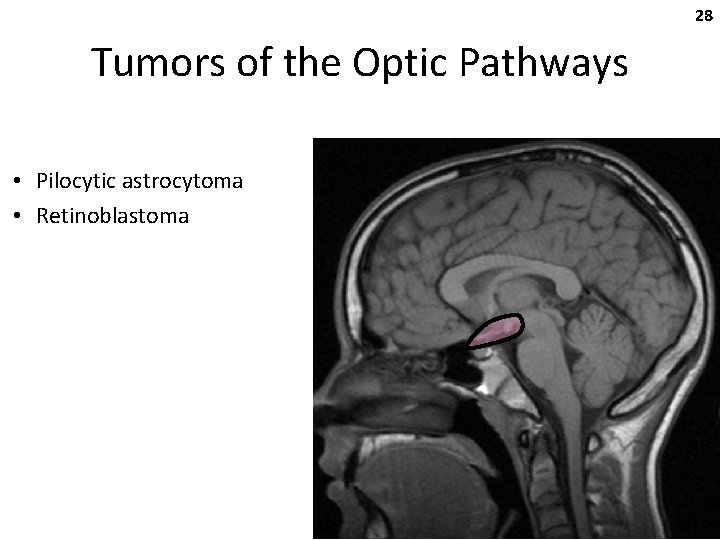
28 Tumors of the Optic Pathways • Pilocytic astrocytoma • Retinoblastoma

29 Signs and Symptoms 1. 2. 3. General and nonlocalizing symptoms Increased intracranial pressure Localizing signs
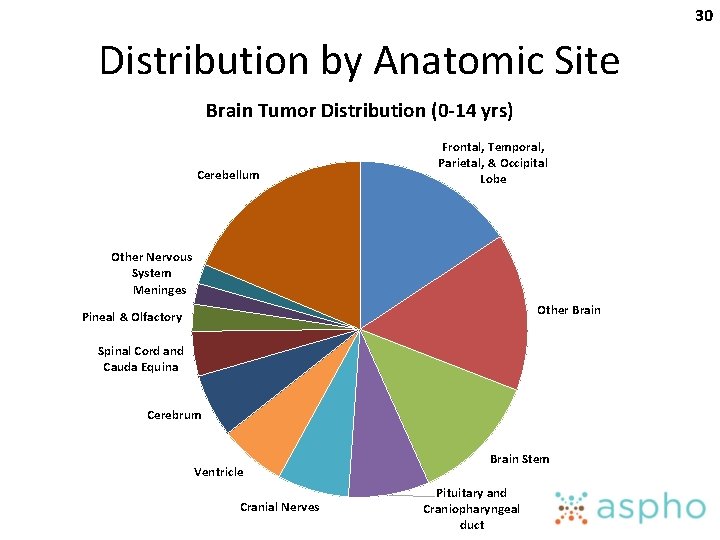
30 Distribution by Anatomic Site Brain Tumor Distribution (0 -14 yrs) Cerebellum Frontal, Temporal, Parietal, & Occipital Lobe Other Nervous System Meninges Other Brain Pineal & Olfactory Spinal Cord and Cauda Equina Cerebrum Ventricle Cranial Nerves Brain Stem Pituitary and Craniopharyngeal duct

31 General and Nonlocalizing Symptoms Headache Vomiting Behavioral changes (e. g. listlessness) Developmental delay Weight gain/loss Others (e. g. endocrine or autonomic dysfunction, failure to thrive)
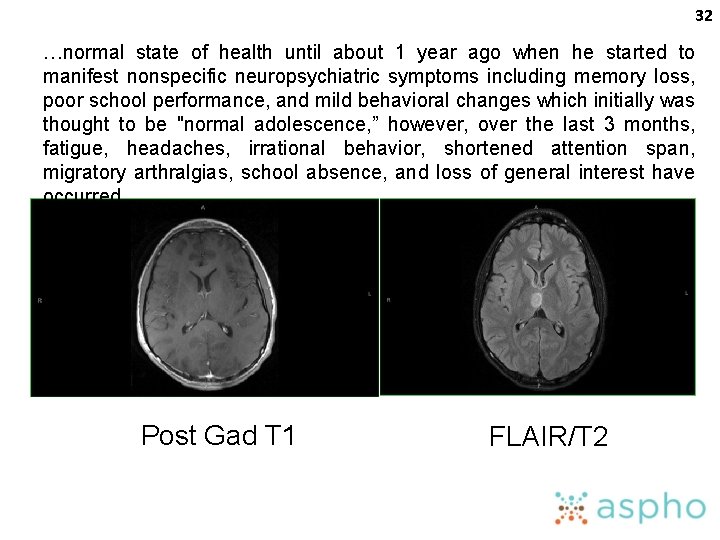
32 …normal state of health until about 1 year ago when he started to manifest nonspecific neuropsychiatric symptoms including memory loss, poor school performance, and mild behavioral changes which initially was thought to be "normal adolescence, ” however, over the last 3 months, fatigue, headaches, irrational behavior, shortened attention span, migratory arthralgias, school absence, and loss of general interest have occurred. Post Gad T 1 FLAIR/T 2

33 Signs and Symptoms 1. 2. 3. General and nonlocalizing symptoms Increased intracranial pressure Localizing signs

34 Increased Intracranial Pressure Headache, irritability, lethargy and vomiting Bulging fontanelle/separation of sutures Papilledema Parinaud syndrome Others (e. g. anisocoria, ataxia, head tilt)
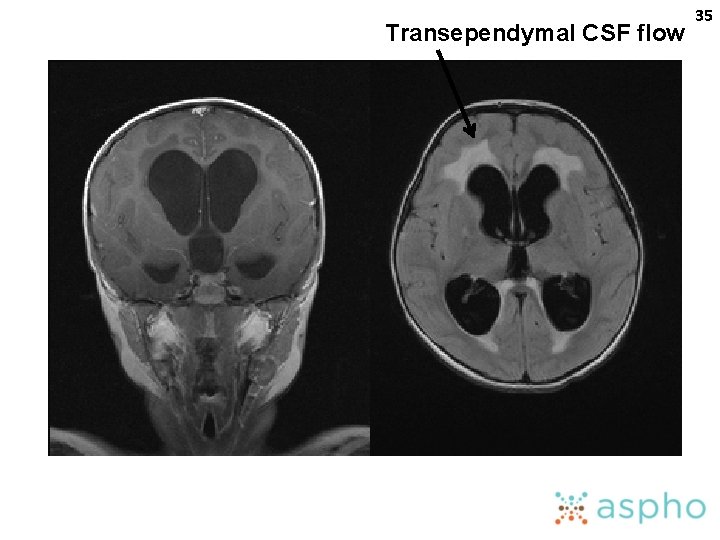
Transependymal CSF flow 35

36 Signs and Symptoms 1. 2. 3. General and nonlocalizing symptoms Increased intracranial pressure Localizing signs

37 Localizing Signs Anatomic Location Common signs and symptoms Frontal lobe Personality changes, decreased speech (Broca’s), seizures Temporal lobe Seizures, poor memory, language comprehension (Wernicke’s) Parietal lobe Decreased sense of touch/pain, poor spatial and visual perception, poor interpretation of language Occipital lobe Poor/Loss of Vision Cerebellum Ataxia, muscle movement, posture Brain stem Weakness, cranial neuropathies (III-XII), autonomic fxn Thalamus Weakness/motor control, consciousness, sleep/wake cycle Hypothalamus Autonomic function (temp regulation, thirst, hunger, etc) endocrinopathies
38
39
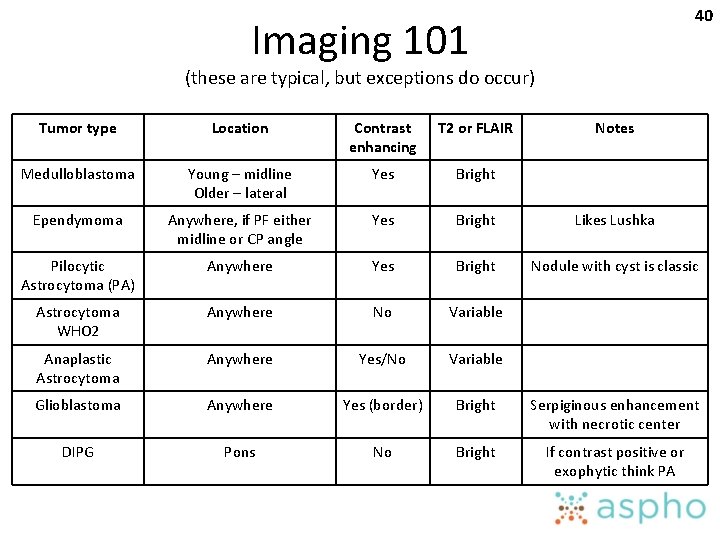
40 Imaging 101 (these are typical, but exceptions do occur) Tumor type Location Contrast enhancing T 2 or FLAIR Notes Medulloblastoma Young – midline Older – lateral Yes Bright Ependymoma Anywhere, if PF either midline or CP angle Yes Bright Likes Lushka Pilocytic Astrocytoma (PA) Anywhere Yes Bright Nodule with cyst is classic Astrocytoma WHO 2 Anywhere No Variable Anaplastic Astrocytoma Anywhere Yes/No Variable Glioblastoma Anywhere Yes (border) Bright Serpiginous enhancement with necrotic center DIPG Pons No Bright If contrast positive or exophytic think PA

Treatment Overview 41

Neurosurgery Generally, obligate for diagnosis Exceptions: DIPG, most NG-GCT Gross total resection is the ideal Debulking may be all that is feasible Provide other supportive measures e. g. shunts, Ommaya reservoirs 42

43 Radiation Therapy Potentially utilized for all CNS tumors Exceptions generally include: Choroid plexus papillomas Low grade gliomas (initially) Treatment in young children “Full dose” treatment ~5400 – 5940 c. Gy Craniospinal irradiation ~1800 - 3600 c. Gy

44 Chemotherapy Used as adjuvant therapy in most cases Particularly efficacious in: Germ cell tumors Embryonal tumors Expanding role in other tumors Blood-brain barrier may pose limitations

45 Chemotherapy Particular interest in use in young children to avoid XRT Novel therapeutics being developed Bypassing blood-brain barrier Local delivery systems (e. g. Gliadel) Osmotic pumps

46 Tumors of the Posterior Fossa Case History 18 yo male with a 3 mo. hx. of intermittent frontal headaches. At diagnosis, the patient presented with nausea and vomiting and was unable to get out of bed. The patient has a hx of diplopia. The patient also had some episodes of unsteady gait. The patient presented to the emergency room where a CT scan was obtained.

47 Tumors of the Posterior Fossa • • Medulloblastoma Ependymoma (common location) Cerebellar astrocytoma (usually pilocytic) AT/RT (increasingly recognized)

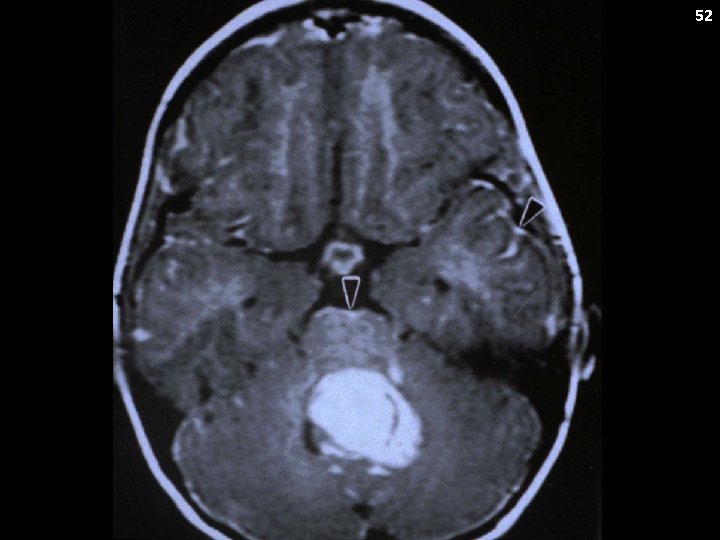
48 Medulloblastoma Clinical Presentation: Raised ICP (headache, vomiting) Ataxia Brain stem findings (sometimes) Metastatic Potential: Yes Neuraxis Systemic spread (very rare)
49 Medulloblastoma Staging: MRI brain/spine CSF cytopathology Bone marrow and/or bone scan (rarely) Laboratory Features: Histologic variants (e. g. desmoplastic, large cell/anaplastic) impact prognosis
50 Medulloblastoma: Risk Classification Features Standard Risk High Risk Extent of resection < 1. 5 cm 3 residual tumor > 1. 5 cm 3 residual tumor M-staging No metastases M+ Histology Anaplasia

51 Medulloblastoma: Genetics Subgroup Ages: (in parenthesis less common) Histologies Genetics Prognosis Wnt Children/(Adults) Classic, rarely large call anaplastic (LCA) Monosomy 6 Nuclear beta-catenin Turcot syndrome Excellent SHH Infants/(Children)/Ad ults Desmoplastic/ nodular, MBEN Classic, LCA PTCH 1/SMO/SUFU, GLI 2 amplification Gorlin syndrome (basal cell carcinoma syndrome) Fair Group 3 (Infants)/Children Classic, LCA i 17 q, MYC amplification Poor Group 4 (Infants)/Children/(A dults) Classic, LCA CDK 6 amplification MYCN amplification Fair Poor if SHH TP 53 mutant
52

53 Medulloblastoma: Treatment Standard Risk: CSI (2340 c. Gy) with PF boost to 54 Gy (± VCR) Adjuvant chemotherapy: A–A–B–A–A–B A cycles: Cisplatin/CCNU/Vincristine B cycles: Cyclophosphamide/Vincristine

Medulloblastoma: Treatment High Risk: Less uniformity in treatment Best resection possible CSI (3600 c. Gy) with PF boost to 54 Gy (± VCR) Adjuvant chemotherapy Typical example: Six cycles of: Cisplatin/Vincristine/CTX 54

55 Medulloblastoma: Prognosis Standard Risk: High Risk: 80 -85% EFS 20 -70% EFS

CNS Embryonal Tumor (CNS ET) and Pineoblastoma often presents with Parinaud syndrome. CNS ET varies based on location Staging is similar to medulloblastoma since these tumors can disseminate Despite molecular differences, both tumors are managed similarly to high risk medulloblastoma Prognosis is variable but generally outcome is poorer than for medulloblastoma 56

CNS Embryonal Tumor (CNS ET) and Pineoblastoma: Treatment is current similar to high risk medullo: Best resection possible CSI (3600 c. Gy) with PF boost to 54 Gy (± VCR) Adjuvant chemotherapy Typical example: Six cycles of: Cisplatin/Vincristine/CTX NOTE: The recent understanding that “PNET” is really multiple different tumors has made treatment decisions less clear 57
58 Embryonal tumor pointers All embryonal tumors like to disseminate – you need an LP and spinal MRI for staging This is a small round blue cell tumor – so in theory bone marrow and bone are at risk … this is rare and marrows are generally not indicated Recurrent tumor is very difficult to salvage, particularly if you have had prior XRT

59 Ependymoma Clinical Presentation: Can occur throughout CNS Dependent on location Raised ICP (esp 4 th ventricular tumors) Ataxia Metastatic Potential: Yes Neuraxis

60 Ependymoma Staging: MRI brain/spine CSF cytopathology Laboratory Features: Histologic variants (e. g. anaplastic variant) may impact prognosis

61 Ependymoma – Genetics Location Molecular Subgroup Histopathology Outcome Notes Posterior Fossa EPN-A Grade 2 or 3 Poor Cp. G island methylator phenotype (CIMP) positive ependymoma EPN-B Grade 2 or 3 Good CIMP-negative EPN-YAP 1 Grade 2 or 3 Good EPN-RELA Grade 2 or 3 Poor MPE Myxopapillary (Grade 1) Good EPN Grade 2 or 3 Good Supratentorial Spine C 11 orf 95 -RELA +
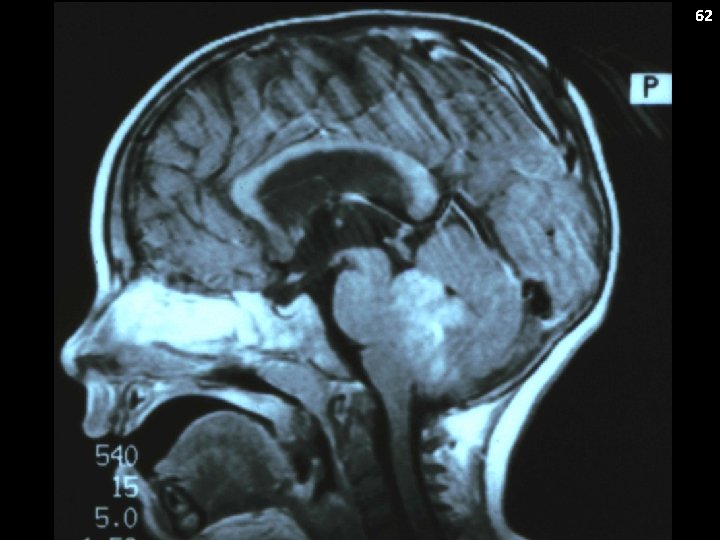
62

63 Ependymoma: Treatment Aggressive upfront resection (GTR “required”) For nonmetastatic tumors: Focal XRT often to a dose between 54 -59. 4 Gy (no CSI prophylaxis despite potential for metastatic spread) Role of chemotherapy uncertain Omitting XRT in select cases with favorable features is being studied For metastatic tumors: Focal XRT to a dose between 54 -59. 4 Gy and CSI (36 Gy) Consideration of chemotherapy

Ependymoma Prognosis 50 -70% EFS for nonmetastatic GTR cases Poor prognosis for metastatic cases 64

65 Ependymoma pointers This is a tumor that likes to spread to the spinal cord – you need an LP and spinal MRI for staging If you don’t get a good resection, it’s pretty hard to cure these patients (that’s the dogma anyhow) If the tumor recurs, you rarely have long term disease control despite many strategies (e. g. chemotherapy, re-operation, re-irradiation, radiosurgery)

66 Brain Stem Tumors: Case History The patient is an 8 -year-old male who presented to an ophthalmologist after complaints of diplopia for approximately 2 weeks. He was noted to have papilledema. He had also been noticed by his parents to be having some slurred speech during the prior 2 -3 days. MRI was consistent with a brain stem tumor centered in the pons that encased the basilar artery. He had evidence of hydrocephalus.

67 Brain stem tumors Almost any brain tumor histology can occur in the brain stem You need to be able to distinguish between: Diffuse midline glioma H 3 K 27 M mutant (reflecting Diffuse Intrinsic Pontine Glioma, DIPG) Low-grade astrocytoma of the brain stem

Diffuse midline glioma, H 3 K 27 M-mutant (Diffuse Intrinsic Pontine Glioma) Synonyms: “Brain stem glioma” Clinical Presentation: see case history above Metastatic potential: Yes, but rare at diagnosis Staging: MRI of brain only (if classic lesion) Biopsy for atypical imaging features Laboratory Features: Bx will reveal diffuse astrocytoma (WHO 2, 3 or 4) Genetics: 80% have histone H 3. 1 or H 3. 3 mutations 68
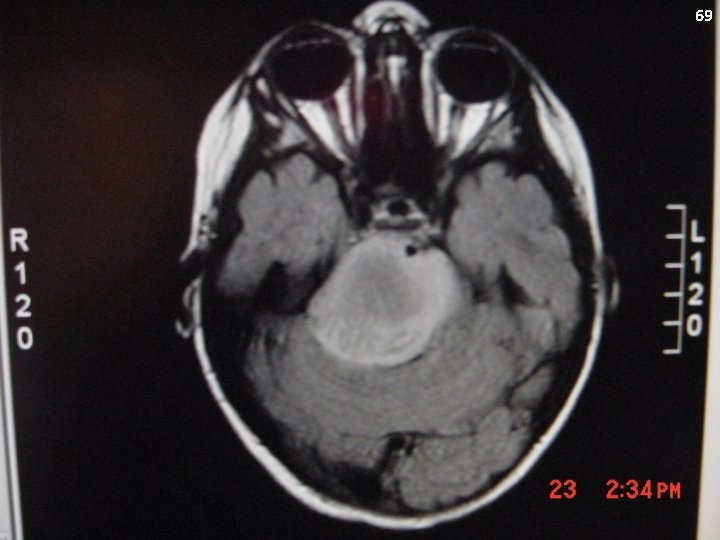
69
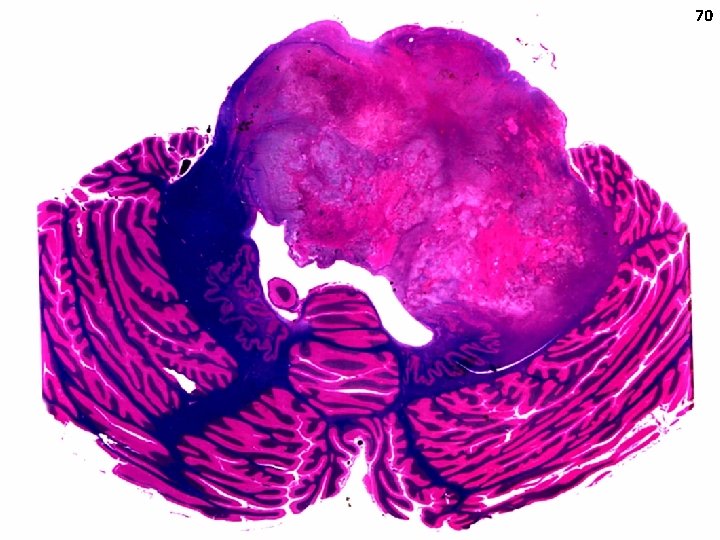
70

71 Diffuse midline glioma, H 3 K 27 M-mutant (DIPG) Focal XRT (54 Gy) is routinely recommended XRT is not curative but often provides disease stabilization for 3 -12 months No chemotherapeutic approach has been shown to alter EFS when compared to XRT alone

Diffuse midline glioma, H 3 K 27 M-mutant (DIPG): Prognosis While rare survivors are reported, in general all children with a DIPG will die between 6 mos and 2 years from the time of diagnosis 72

73 Brain Stem Gliomas pointers There’s a difference between an “intrinsic pontine glioma” and “exophytic” brain stem tumors. Exophytic BSGs are usually pilocytic astrocytomas. PA in the brain stem has a much better prognosis. PAs in the brain stem brightly enhance on postgad MRI and are often centered in other areas of the brain stem besides the pons

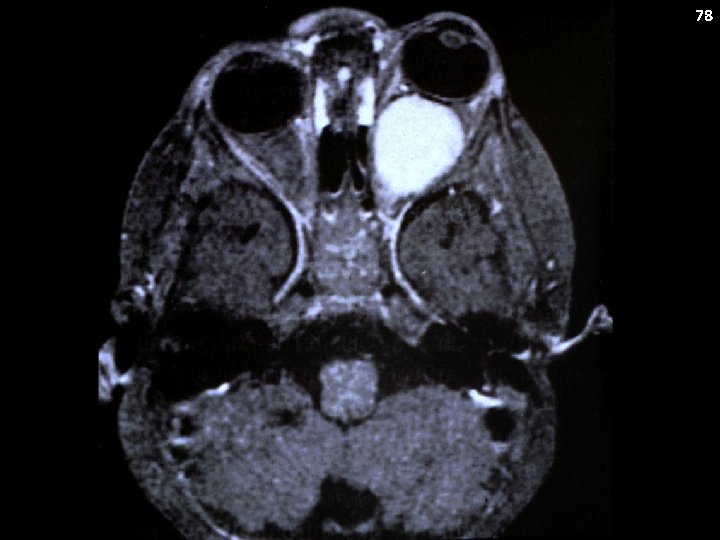
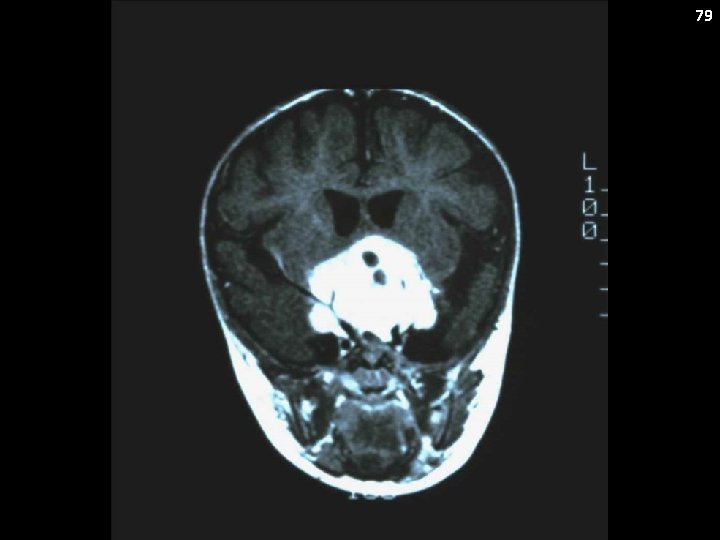
74 Pilocytic Astrocytoma: Case History The patient developed bitemporal headaches which were occurring every other day. She was treated for allergies, but with no improvement. An x-ray of her sinuses was normal. She was advised to reduce her caffeine intake and was referred for a visual screen which was normal. She had persistent headaches and was prescribed a bite block. Recently, she complained of a lack of coordination in her left hand. An MRI revealed a heterogenous cystic mass in the cerebellar hemisphere.

75 Pilocytic Astrocytomas Can occur virtually anywhere in the CNS Common locations include: Cerebellum (cerebellar astrocytoma) Optic pathway (optic pathway glioma) Hypothalamus Brain stem Spinal cord

76 Pilocytic Astrocytoma Synonyms: Astrocytoma (WHO 1), cerebellar astrocytoma, “low-grade” glioma Clinical presentation: Location dependent Metastatic Potential: No (for the most part) Staging: MRI of brain ± spine Laboratory Features: None Genetics: BRAF truncated fusion (BRAF-KIAA 1549 gene fusion) or BRAF V 600 E mutation
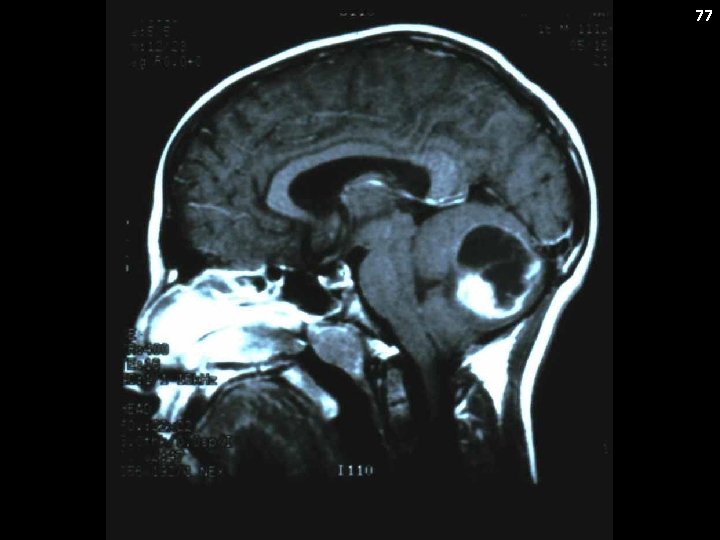
77
78
79

Pilocytic Astrocytoma Treatment Dependent on Location GTR is curative (e. g. cerebellar primaries) and no further treatment is necessary If GTR cannot be achieved (typically the case for optic pathway and hypothalamic tumors): Observation in selected cases Best safe resection and then consider chemotherapy for progression or bulky residual disease XRT is generally considered therapy of last resort 80

Pilocytic Astrocytoma Treatment Commonly used regimens: Carboplatin/Vincristine TPCV Carboplatin alone Vinblastine Cisplatin/etoposide Targeted therapies being studied BRAF fusions – MEK inhibitors V 600 E mutation – BRAF inhibitors 81

Pilocytic Astrocytoma Prognosis Dependent on Location Excellent prognosis in the setting of a GTR Midline tumors (e. g. hypothalamic, brain stem) have a more variable prognosis because GTR is rarely feasible 82

83 Optic Pathway Glioma (OPG) Most commonly Pilocytic Astrocytomas that arise in the region of the optic nerves, optic chiasm or the optic radiations Most occur in young children High incidence (~ 20%) in children with NF-1 Management varies: Observation (more common in NF-1 pts) Chemotherapy (see pilocytic astrocytoma)

84 Pilocytic Astrocytoma pointers PA can occur virtually anywhere in the brain If you can cut it out entirely, you can usually cure the patient Optic pathway PA is often associated with NF-1 and can have a pretty benign course

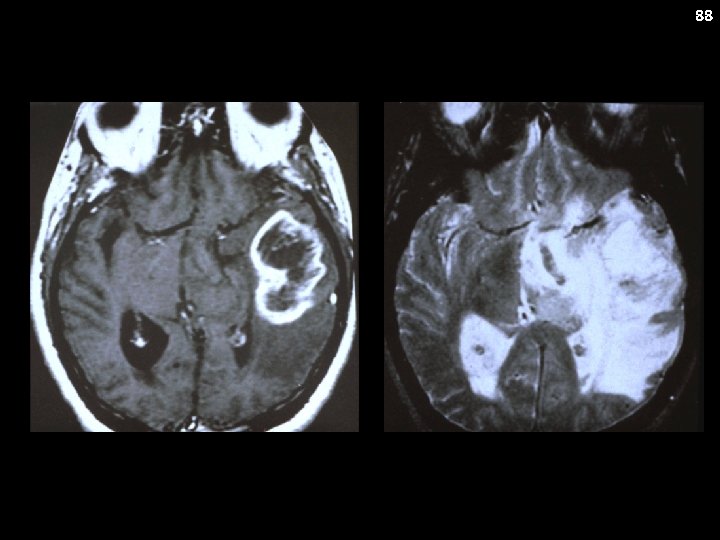
85 AA and GBM: Case History The patient is an 8 yo girl who was diagnosed with a large mass in the right frontoparietal region of her cerebral cortex after presenting to the ER following a seizure. Her parents indicate that she had a two-month history of progressive nausea and vomiting accompanied by headache

86 AA and GBM Synonyms: Astrocytoma (WHO 3 and 4); highgrade gliomas Clinical Presentation: Variable, headaches, seizures (due to supratentorial predilection) Metastatic Potential: No (for the most part) Staging: MRI of brain ± spine Laboratory Features: MGMT promoter methylation increasingly assayed
87 Astrocytoma: Genetics Tumor type Location Genetics Drug being tested Pilocytic Astrocytoma; High grade glioma (10%) Anywhere BRAF V 600 E Dabrafenib (never use with fusion) Pilocytic Astrocytoma Anywhere BRAF-KIAA 1549 fusion MEK inhibitors DIPG/HGG Midline H 3. 3 K 27 M H 3. 1 K 27 M ? HDAC inhibitors HGG Cortical H 3. 3 G 34 R H 3. 3 G 34 V ? HDAC inhibitors Note: In contrast to adults, IDH mutant diffuse astrocytomas (WHO 2, 3 and 4) rarely occur in the pediatric population with the exception of some older adolescents
88

89 AA and GBM: Treatment Maximal surgical resection Focal XRT generally with low-dose TMZ ? role of adjuvant chemotherapy and currently no standard in pediatrics Possible role for a nitrosourea (e. g. TMZ and/or CCNU) for most patients

90 AA and GBM: Prognosis You have a fighting chance of curing AA with a GTR and adjuvant therapy (~ 31% EFS) The same is not true for GBM (~ 18% EFS)

91 AA and GBM pointers No chemotherapy, excepting alkylating agents (e. g. nitrosoureas and TMZ), has really shown much benefit for these tumors in children. The jury is still out on the utility of temozolomide for children with AA and GBM Remember, AA and GBM occur on a continuum with each other and diffuse astrocytoma (WHO 2) tumors. The distinction is somewhat in the “eyes of the pathologist”

92 Germinoma: Case History This 19 -year-old male experienced dizziness over the last 3 months and has been bumping into objects. He was evaluated in July in the emergency room because of his staggering and nausea and vomiting. He had a sodium of 158 with a urine specific gravity of less than 1. 005. He also admitted at that point that he had been drinking a lot and also had polyuria.

Germinoma Synonyms: Non-secreting GCT Clinical Presentation: Two most common sites: 1. Suprasellar – DI, panhypituitarism 2. Pineal region – Parinauds, raised ICP Metastatic Potential: Yes Staging: Biopsy required for diagnosis MRI brain/spine CSF cytopathology Laboratory features: CSF markers (AFP/β-HCG) should be negative (AFP) or low (β-HCG) 93
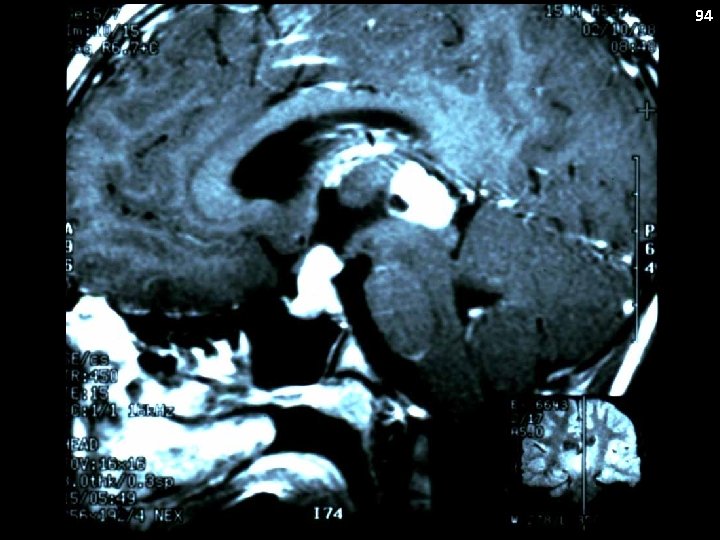
94

95 Germinoma: Treatment Two options: • Neoadjuvant chemotherapy followed by response based XRT – Typical chemotherapy: Carbo/Etoposide x 4 cycles – Dose of XRT is dependent on response but ranges between 30 -36 Gy and always includes whole ventricle • XRT alone – Dose in range of 40 -45 Gy and includes whole ventricle

96 Germinoma: Prognosis Excellent prognosis with > 90% OS at 5 years with either XRT alone of chemotherapy/XRT Endocrine abnormalities will not resolve even upon successful treatment

97 NG-GCT: Case History This 7 yo was well until his mother noted his voice deepening. Shortly thereafter, she noted the appearance of pubic hair, and increased fullness of the hair on his eyebrows and increased growth. She notes that for the past three months he has gained approximately 12 pounds. In addition, she has noticed enlargement of the penis.

98 NG-GCT Synonyms: Secreting GCT Clinical Presentation: Precocious puberty, DI, panhypopituitarism, raised ICP Metastatic Potential: Yes Diagnostic Evaluation: No biopsy if markers + MRI brain/spine CSF cytopathology Laboratory Findings: CSF tumor markers (AFP/β-HCG) elevated (one or both)
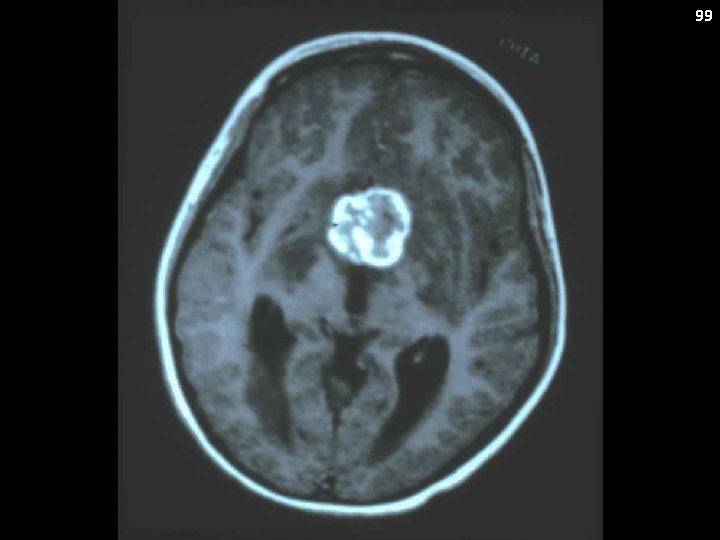
99

100 NG-GCT: Treatment Neoadjuvant chemotherapy: A–B–A–B A cycles: Carboplatin/etoposide B cycles: Ifosfamide/etoposide Response based radiation therapy XRT includes CSI “Second look” surgery is often recommended following neoadjuvant chemotherapy if markers have not normalized or mass disease remains. If markers are better but mass is growing, consider growing teratoma syndrome

101 NG-GCT: Prognosis > 80% EFS with current therapies

102 Germ cell tumor pointers CSF tumor marker (AFP) is not elevated in germinoma. Mild elevation in β-HCG can be seen Occult DI in a teenager is often germinoma waiting to declare itself (or LCH) NG-GCT reqruies more intensive therapy than germinoma 10% of germinomas are multifocal in the pineal and suprasellar region
103 Spinal cord tumors Primary intramedullary tumors include: Low-grade astrocytomas (often pilocytic) Ependymomas (often myxopapillary variant) AA/GBM (much less commonly) Clinical Presentation: Back pain, upper or lower extremity weakness, change in bowel/bladder function

104 Spinal cord tumors CNS tumors that metastasize to spine: Embryonal tumors Germ cell tumors Ependymoma Astrocytomas (much less commonly)

105 Stem cell transplantation The role of SCT continues to evolve in the treatment of children with brain tumors Major role has been in the treatment of very young children with embryonal tumors in an effort to delay/avoid XRT Unclear benefit in the treatment of astrocytic tumors Role in salvage is a growing debate, particularly for children who have been previously radiated

106 Genetic associations NF-1 think optic pathway gliomas and nonneoplastic hamartomatous lesions. Often very benign course. Tuberous sclerosis think subependymal giant cell astrocytoma (SEGA) presenting at the Foramen of Monro. Treatment considerations now include surgery vs. an m. Tor inhibitor (e. g. everolimus)

Genetic Associations 107 Atypical Teratoid/Rhaboid Tumor (AT/RT) INI 1/h. SNF 5/SMARCB 1 – no longer uniformly fatal with multimodality therapy including XRT. Therapy is more “sarcoma-like” than other brain tumors. Testing recommended at diagnosis to distinguish between AT/RT and other tumors (e. g. medulloblastoma, choroid plexus tumors) Genetic testing is recommended in all cases to distinguish germline vs. sporadic mutations

108 Genetic associations Retinoblastoma (heritable) are at risk for trilateral disease which is a small round blue cell tumor in the region of the pineal gland (pineoblastoma)

109 Complications/Late Effects Surgery: Depends what you cut/burn/suction Cerebellum: Cerebellar mutism syndrome Radiation Therapy: Neurodevelopmental morbidity Endocrine (e. g. hypothyroidism, GH deficiency) XRT associated second malignancy - GBM - Meningioma

110 Complications/Late Effects Chemotherapy: Hearing loss (platinum-based therapy) Second malignancy ? neurodevelopmental morbidity Endocrine: You don’t recover what you’ve lost
111
- Slides: 111