HDL Biogenesis Functionality Thomas Dayspring MD FACP FNLA

HDL Biogenesis & Functionality Thomas Dayspring, MD, FACP, FNLA Diplomate of the American Board of Clinical Lipidology Fellow of the National Lipid Association North American Menopause Society Certified Menopause Practitioner: NCMP North Jersey Institute of Menopausal Lipidology New Jersey Wayne,

High Density Lipoproteins ► HDL is a mixture of lipoprotein particles with densities ranging from 1. 063 to 1. 21 g/ml. ► Depending on the lipid composition, HDL may have a discoidal or spherical shape. ► Mature spherical HDL contains 45– 55% (as mass %) apoproteins, 26– 32% phospholipids, 15– 20% esterified cholesterol, 3– 5% free cholesterol, and approximately 5% triglycerides. ► The main protein component of HDL is apolipoprotein AI (apo. A-I) which plays a key role in the biogenesis and functions of HDL Tsompanidi EM et al. Atherosclerosis 208 (2010) 3– 9
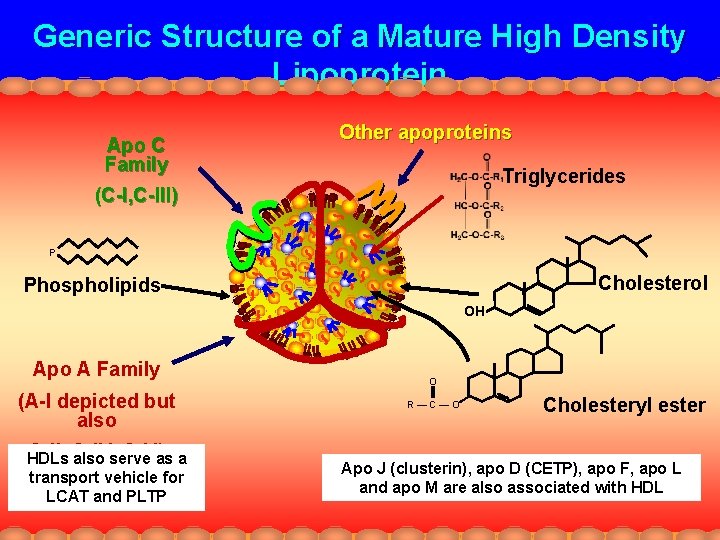
Generic Structure of a Mature High Density Lipoprotein Apo C Family Other apoproteins Triglycerides (C-I, C-III) P Cholesterol Phospholipids OH Apo A Family (A-I depicted but also A-II, A-IV, A-V)as a HDLs also serve transport vehicle for LCAT and PLTP O R—C—O Cholesteryl ester Apo J (clusterin), apo D (CETP), apo F, apo L and apo M are also associated with HDL

High Density Lipoproteins ► Work on phospholipid-containing HDL discoidal particles suggests that the apo. A-I molecules adopt a ‘double-belt’ orientation in which two apo. A-I molecules wrap around a patch of phospholipid bilayer in an antiparallel fashion. ► They maintain a registry, stabilized by intermolecular salt bridges, in which the fifth amphipathic helix of each molecule overlaps ► Most circulating HDL contains a cholesteryl-ester and triglyceride-rich core that results in a spherical shape. Huang R et al. Nat Struct Mol Biol. 2011 Apr; 18(4): 416 -22. Epub 2011 Mar 13

High Density Lipoproteins ► The cross-linking patterns in reconstituted spheres are highly similar to those in the discs, regardless of whether they contained three molecules of apo. A-I in each particle or only two. ► For three apo. A-I molecules in spherical HDL to adopt the same intermolecular contacts as only two in the discs the trefoil model is proposed ► This invokes a bending of the planar belts and the intercalation of a similarly bent third apo. A-I molecule so that all three adopt intermolecular contacts identical to the two in the discs. ► The resulting cage-like structure can stabilize surface phospholipids and contain the neutral lipid core. Apo. A-I thus adopts a common structural framework regardless of particle shape. Huang R et al. Nat Struct Mol Biol. 2011 Apr; 18(4): 416 -22. Epub 2011 Mar 13

High Density Lipoproteins Apo. A-I adopts a common general structural organization, characterized by distinct intermolecular contacts, in virtually all lipid-containing particles, regardless of size and shape or natural versus synthetic method of production. However, it is also clear that apo. A-I can undergo conformational changes within this framework, particularly in the smaller particles Huang R et al. Nat Struct Mol Biol. 2011 Apr; 18(4): 416 -22. Epub 2011 Mar 13
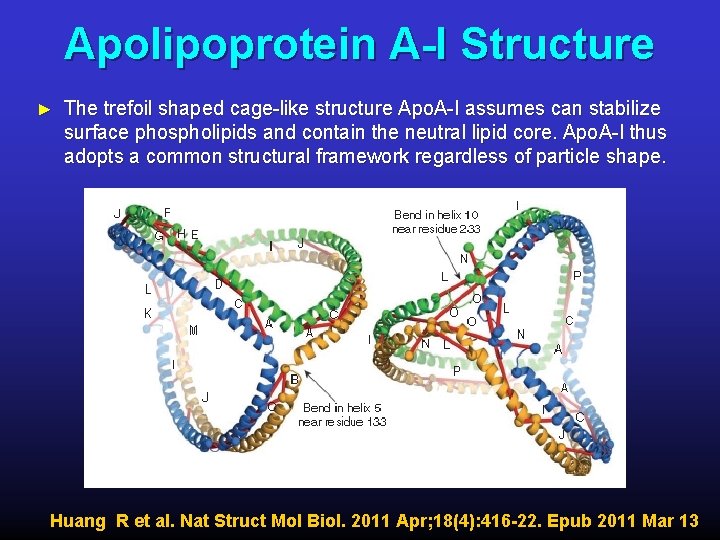
Apolipoprotein A-I Structure ► The trefoil shaped cage-like structure Apo. A-I assumes can stabilize surface phospholipids and contain the neutral lipid core. Apo. A-I thus adopts a common structural framework regardless of particle shape. Huang R et al. Nat Struct Mol Biol. 2011 Apr; 18(4): 416 -22. Epub 2011 Mar 13
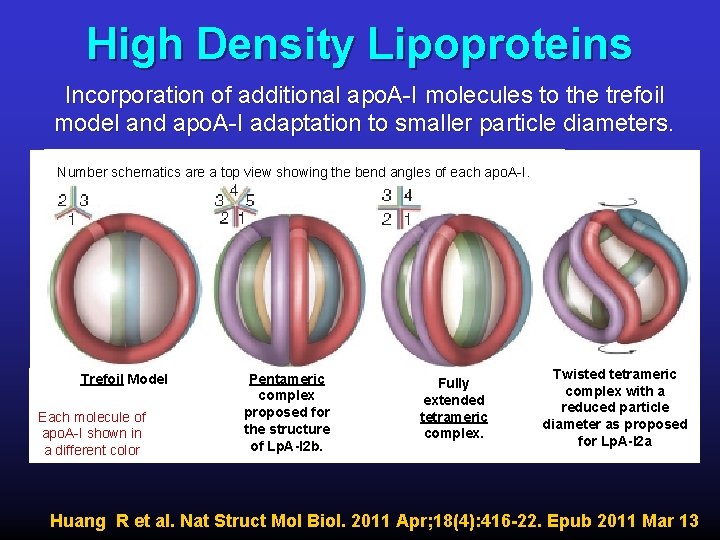
High Density Lipoproteins Incorporation of additional apo. A-I molecules to the trefoil model and apo. A-I adaptation to smaller particle diameters. Number schematics are a top view showing the bend angles of each apo. A-I. Trefoil Model Each molecule of apo. A-I shown in a different color Pentameric complex proposed for the structure of Lp. A-I 2 b. Fully extended tetrameric complex. Twisted tetrameric complex with a reduced particle diameter as proposed for Lp. A-I 2 a Huang R et al. Nat Struct Mol Biol. 2011 Apr; 18(4): 416 -22. Epub 2011 Mar 13

High Density Lipoproteins Molecular twist required to attain the experimentally Lp. A-I particle diameters. The helical domains of apo. A-I molecules are represented as tubes extending across the particle surface with each molecule in a different color. In Lp. A-I 2 b, apo. A-I helical domains from five molecules are all maximally extended. The smaller particles contain four apo. A-I molecules per particle and each pole has been twisted by an angle Θ such that the complex diameter (D) matches the experimentally determined values (average of the diameter values from gel filtration and native PAGE) Huang R et al. Nat Struct Mol Biol. 2011 Apr; 18(4): 416 -22. Epub 2011 Mar 13
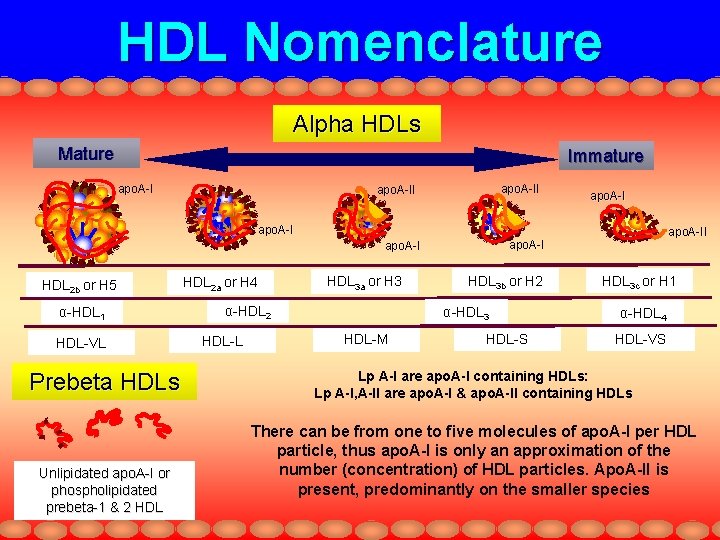
HDL Nomenclature Alpha HDLs Mature Immature apo. A-II apo. A-I HDL 2 b or H 5 α-HDL 1 HDL-VL Prebeta HDLs Unlipidated apo. A-I or phospholipidated prebeta-1 & 2 HDL 2 a or H 4 HDL 3 a or H 3 α-HDL 2 HDL-L HDL 3 b or H 2 α-HDL 3 HDL-M apo. A-II HDL-S HDL 3 c or H 1 α-HDL 4 HDL-VS Lp A-I are apo. A-I containing HDLs: Lp A-I, A-II are apo. A-I & apo. A-II containing HDLs There can be from one to five molecules of apo. A-I per HDL particle, thus apo. A-I is only an approximation of the number (concentration) of HDL particles. Apo. A-II is present, predominantly on the smaller species

Double Belt Model of Discoidal HDL Apo. A-I contains a globular (possibly lipid-binding) N-terminal domain (residues 1– 43) and a lipid-binding C-terminal domain (residues 44– 243) The apo A-I dimer is docked around the edge of an 85Å diameter space-filling patch of 1 -palmitoyl-2 -oleoylsnglycero-3 - phosphocholine (POPC) bilayer Segrest. JP, Harvey SC, & Zannis V. TCM 2000; 10: 246 -252
Acquiring Cholesterol Apo. A-I Mediated Cholesterol Transport Particle Lipidation
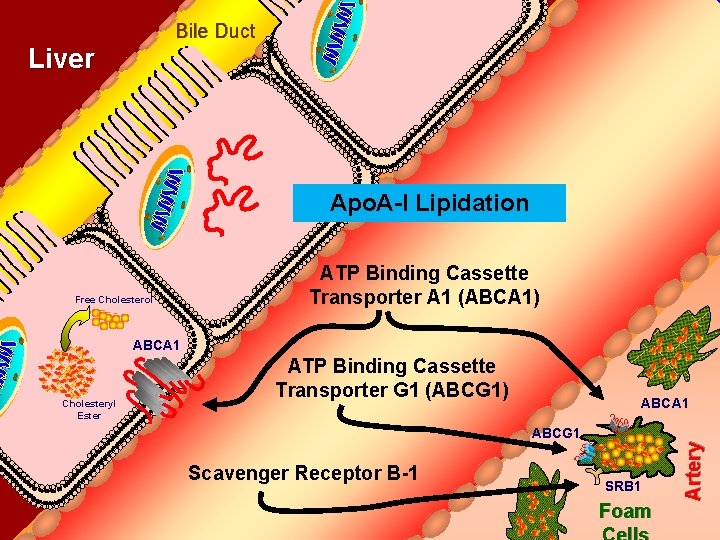
Bile Duct Liver Apo. A-I Lipidation Free Cholesterol ATP Binding Cassette Transporter A 1 (ABCA 1) ABCA 1 ABCG 1 Scavenger Receptor B-1 SRB 1 Foam Artery Cholesteryl Ester ATP Binding Cassette Transporter G 1 (ABCG 1)
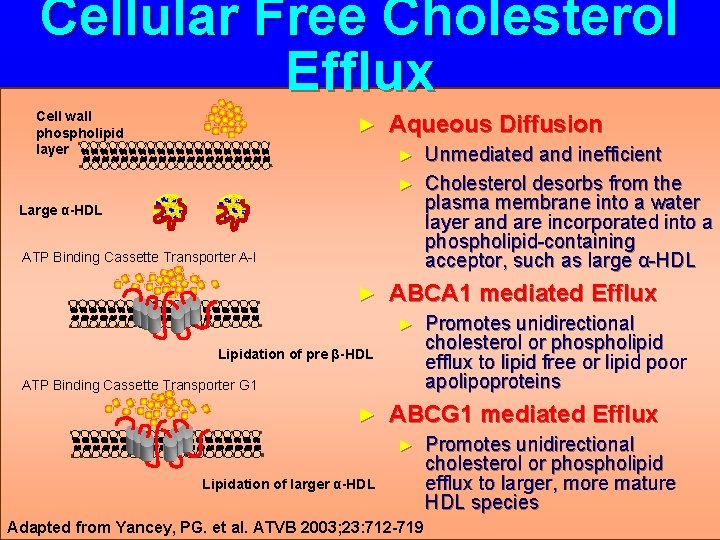
Cellular Free Cholesterol Efflux Cell wall phospholipid layer ► Aqueous Diffusion Unmediated and inefficient ► Cholesterol desorbs from the plasma membrane into a water layer and are incorporated into a phospholipid-containing acceptor, such as large α-HDL ► Large α-HDL ATP Binding Cassette Transporter A-I ► ABCA 1 mediated Efflux ► Lipidation of pre β-HDL ATP Binding Cassette Transporter G 1 ► Promotes unidirectional cholesterol or phospholipid efflux to lipid free or lipid poor apolipoproteins ABCG 1 mediated Efflux ► Lipidation of larger α-HDL Adapted from Yancey, PG. et al. ATVB 2003; 23: 712 -719 Promotes unidirectional cholesterol or phospholipid efflux to larger, more mature HDL species
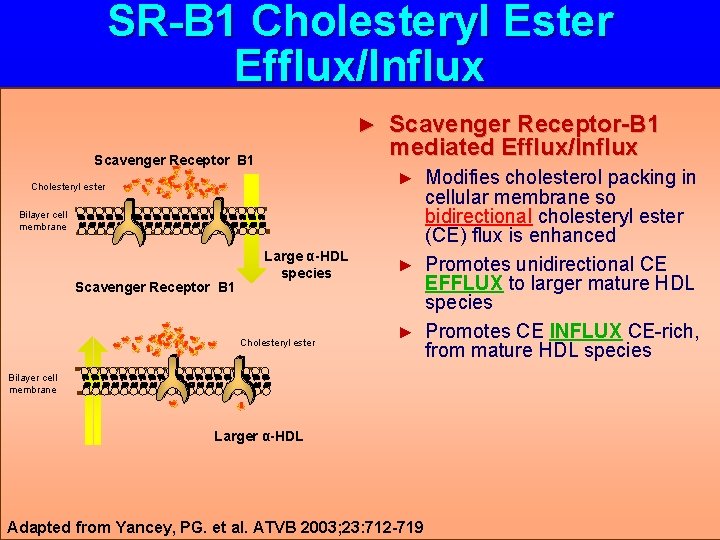
SR-B 1 Cholesteryl Ester Efflux/Influx ► Scavenger Receptor B 1 Scavenger Receptor-B 1 mediated Efflux/Influx Modifies cholesterol packing in cellular membrane so bidirectional cholesteryl ester (CE) flux is enhanced ► Promotes unidirectional CE EFFLUX to larger mature HDL species ► Promotes CE INFLUX CE-rich, from mature HDL species ► Cholesteryl ester Bilayer cell membrane Scavenger Receptor B 1 Large α-HDL species Cholesteryl ester Bilayer cell membrane Larger α-HDL Adapted from Yancey, PG. et al. ATVB 2003; 23: 712 -719
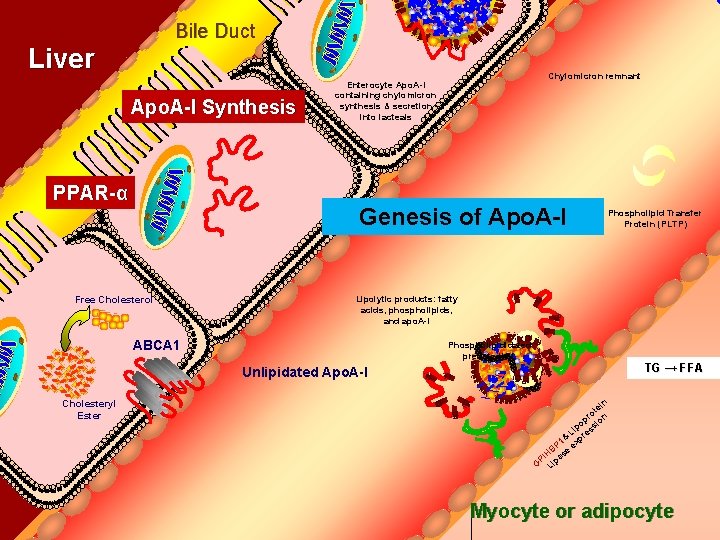
Bile Duct Liver Apo. A-I Synthesis PPAR-α Chylomicron remnant Enterocyte Apo. A-I containing chylomicron synthesis & secretion into lacteals Genesis of Apo. A-I Free Cholesterol Phospholipid Transfer Protein (PLTP) Lipolytic products: fatty acids, phospholipids, and apo. A-I ABCA 1 Phospholipididated prebeta HDL TG → FFA Unlipidated Apo. A-I Cholesteryl Ester n ei ot n r p io po s Li res & p P 1 ex B H se PI a G Lip Myocyte or adipocyte
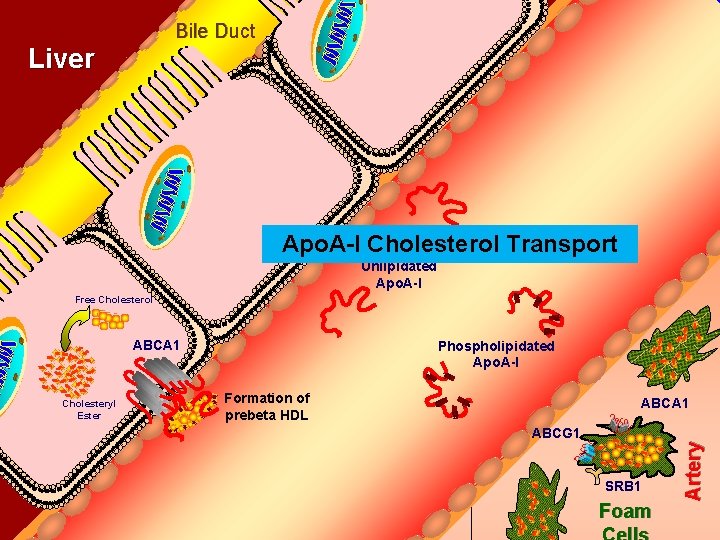
Bile Duct Liver Apo. A-I Cholesterol Transport Unlipidated Apo. A-I Free Cholesterol ABCA 1 Formation of prebeta HDL ABCA 1 ABCG 1 SRB 1 Foam Artery Cholesteryl Ester Phospholipidated Apo. A-I
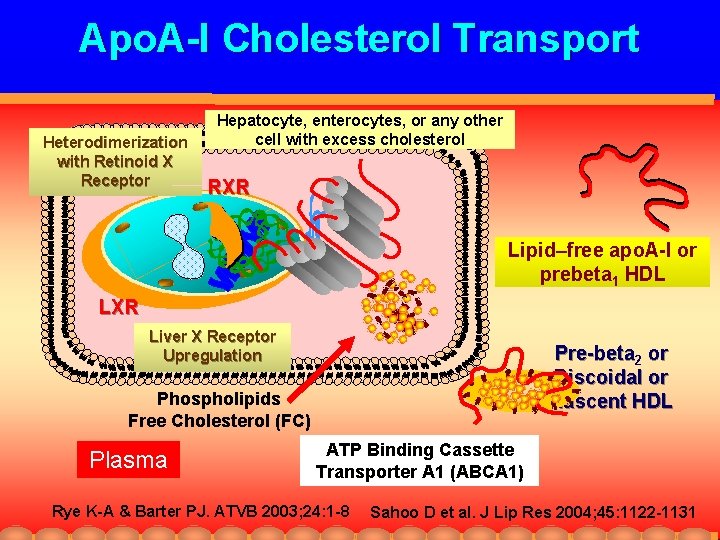
Apo. A-I Cholesterol Transport Heterodimerization with Retinoid X Receptor Hepatocyte, enterocytes, or any other cell with excess cholesterol RXR Lipid–free apo. A-I or prebeta 1 HDL LXR Liver X Receptor Upregulation Pre-beta 2 or Discoidal or Nascent HDL Phospholipids Free Cholesterol (FC) Plasma ATP Binding Cassette Transporter A 1 (ABCA 1) Rye K-A & Barter PJ. ATVB 2003; 24: 1 -8 Sahoo D et al. J Lip Res 2004; 45: 1122 -1131

ATP Binding Cassette Transporter A-1 Model for the apolipoprotein / ABCA 1 lipid-removal pathway Excess free cholesterol is incorporated into phospholipid domains of the inner leaflet of the plasma membrane. ABCA 1 transports free cholesterol and phospholipids into its transmembrane chamber where they prepare for exit In Free Cholesterol Phospholipid John Oram ATVB 2003; 23: 720 -727

ATP Binding Cassette Transporter A-1 Model for the apolipoprotein / ABCA 1 lipid-removal pathway Nascent or pre-β HDL Lipid-free apo. A 1 Out Lipid-poor apo. A 1 solubilizes these cell surface lipid domains, and is lipidated In Cholesterol Free cholesterol and phospholipids are flipped to the outer leaflet of the plasma membrane and extruded from the chamber. Phospholipid Discoid, nascent prebeta HDL particles are formed John Oram ATVB 2003; 23: 720 -727
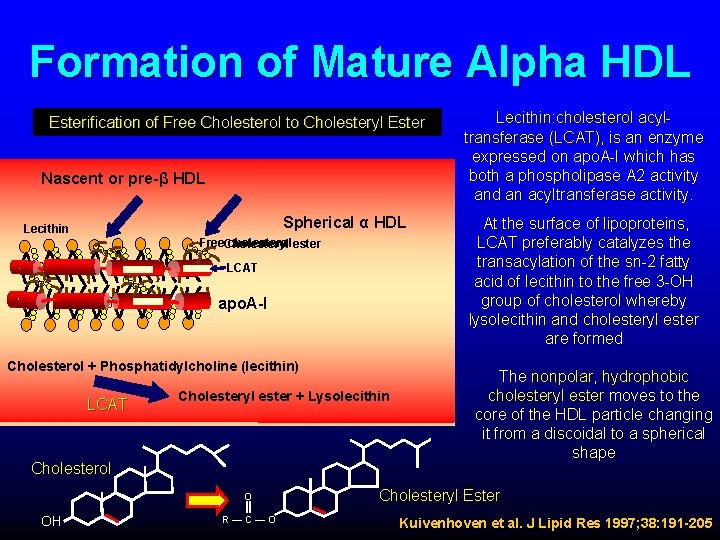
Formation of Mature Alpha HDL Esterification of Free Cholesterol to Cholesteryl Ester Nascent or pre-β HDL Spherical α HDL Lecithin Free. Cholesteryl cholesterol ester LCAT apo. A-I Cholesterol + Phosphatidylcholine (lecithin) LCAT Cholesteryl ester + Lysolecithin Cholesterol OH O R—C—O Lecithin: cholesterol acyltransferase (LCAT), is an enzyme expressed on apo. A-I which has both a phospholipase A 2 activity and an acyltransferase activity. At the surface of lipoproteins, LCAT preferably catalyzes the transacylation of the sn-2 fatty acid of lecithin to the free 3 -OH group of cholesterol whereby lysolecithin and cholesteryl ester are formed The nonpolar, hydrophobic cholesteryl ester moves to the core of the HDL particle changing it from a discoidal to a spherical shape Cholesteryl Ester Kuivenhoven et al. J Lipid Res 1997; 38: 191 -205
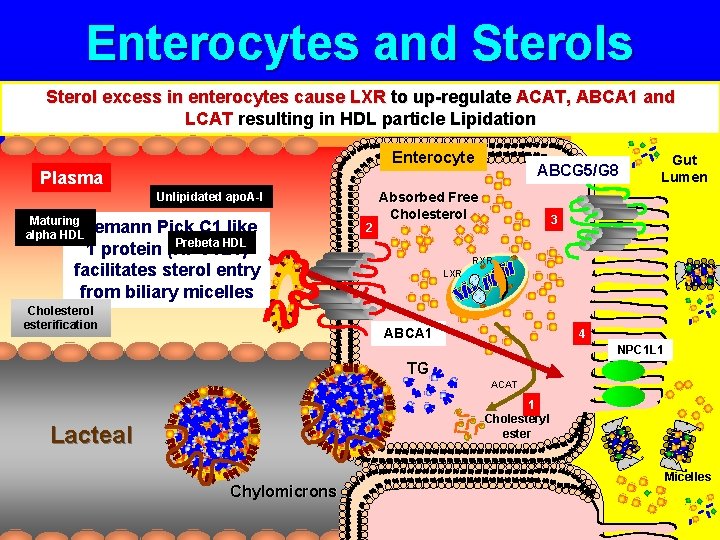
Enterocytes and Sterols Sterol excess in enterocytes cause LXR to up-regulate ACAT, ABCA 1 and LCAT resulting in HDL particle Lipidation Enterocyte ABCG 5/G 8 Plasma Unlipidated apo. A-I Maturing Niemann alpha HDL Pick C 1 like Prebeta HDL 1 protein (NPC 1 L 1) facilitates sterol entry from biliary micelles Cholesterol esterification 2 Absorbed Free Cholesterol Gut Lumen 3 RXR LXR ABCA 1 4 NPC 1 L 1 TG ACAT 1 Cholesteryl ester Lacteal Chylomicrons Micelles
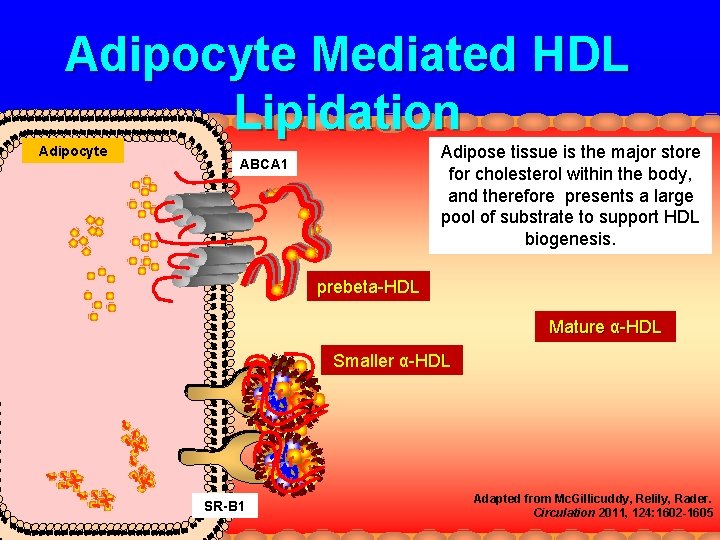
Adipocyte Mediated HDL Lipidation Adipocyte Adipose tissue is the major store for cholesterol within the body, and therefore presents a large pool of substrate to support HDL biogenesis. ABCA 1 prebeta-HDL Mature α-HDL Smaller α-HDL SR-B 1 Adapted from Mc. Gillicuddy, Relily, Rader. Circulation 2011, 124: 1602 -1605
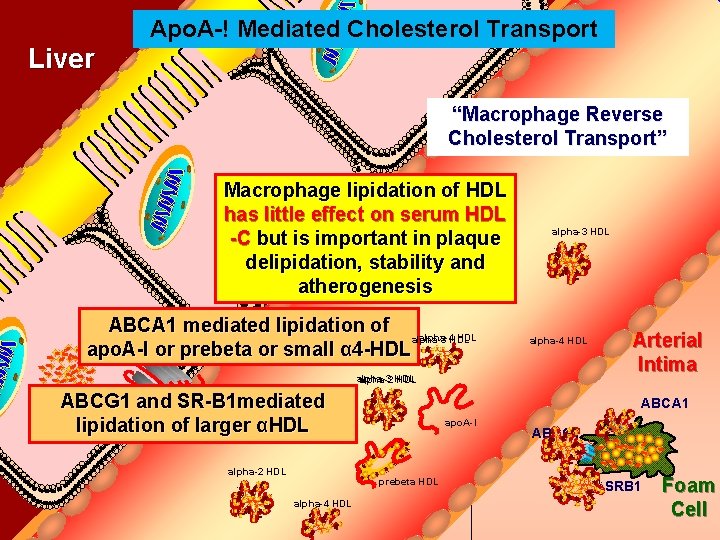
Apo. A-! Mediated Cholesterol Transport Bile Duct Liver “Macrophage Reverse Cholesterol Transport” Macrophage lipidation of HDL has little effect on serum HDL -C but is important in plaque delipidation, stability and atherogenesis ABCA 1 mediated lipidation of alpha-4 HDL alpha-3 HDL ABCA 1 apo. A-I or prebeta or small α 4 -HDL alpha-3 HDL alpha-4 HDL alpha-3 alpha-2 HDL Cholesteryl ABCG 1 and SR-B 1 mediated Ester lipidation of larger αHDL alpha-2 HDL ABCA 1 apo. A-I prebeta HDL alpha-4 HDL Arterial Intima ABCG 1 SRB 1 Foam Cell
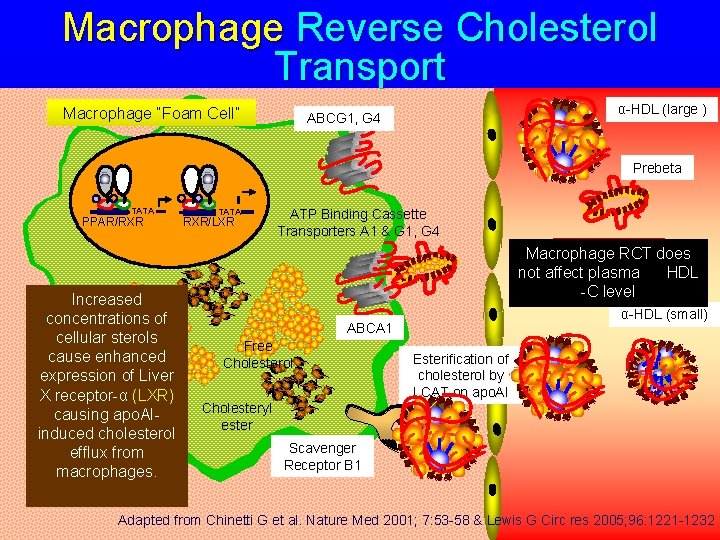
Macrophage Reverse Cholesterol Transport Macrophage “Foam Cell” α-HDL (large ) ABCG 1, G 4 Prebeta TATA PPAR/RXR Increased concentrations of cellular sterols cause enhanced expression of Liver X receptor-α (LXR) causing apo. AIinduced cholesterol efflux from macrophages. TATA RXR/LXR ATP Binding Cassette Transporters A 1 & G 1, G 4 More mature Macrophage RCTα-does HDL particles HDL not affect plasma -C level α-HDL (small) ABCA 1 Free Cholesterol Cholesteryl ester Esterification of cholesterol by LCAT on apo. AI Scavenger Receptor B 1 Adapted from Chinetti G et al. Nature Med 2001; 7: 53 -58 & Lewis G Circ res 2005; 96: 1221 -1232
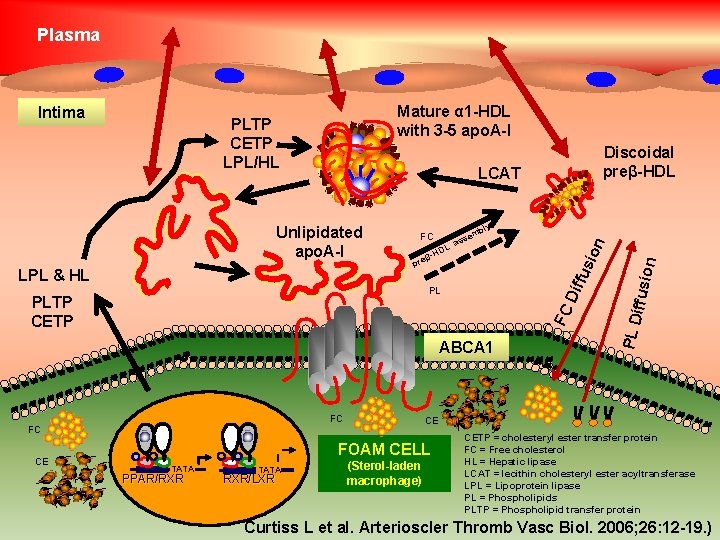
Plasma Mature α 1 -HDL with 3 -5 apo. A-I LCAT PL FC PLTP CETP ABCA 1 FC FC CE FOAM CELL TATA PPAR/RXR TATA RXR/LXR on L -HD bly ion β pre em ass iffusi LPL & HL FC fus Unlipidated apo. A-I CE Discoidal preβ-HDL PL D PLTP CETP LPL/HL Dif Intima (Sterol-laden macrophage) CETP = cholesteryl ester transfer protein FC = Free cholesterol HL = Hepatic lipase LCAT = lecithin cholesteryl ester acyltransferase LPL = Lipoprotein lipase PL = Phospholipids PLTP = Phospholipid transfer protein Curtiss L et al. Arterioscler Thromb Vasc Biol. 2006; 26: 12 -19. )
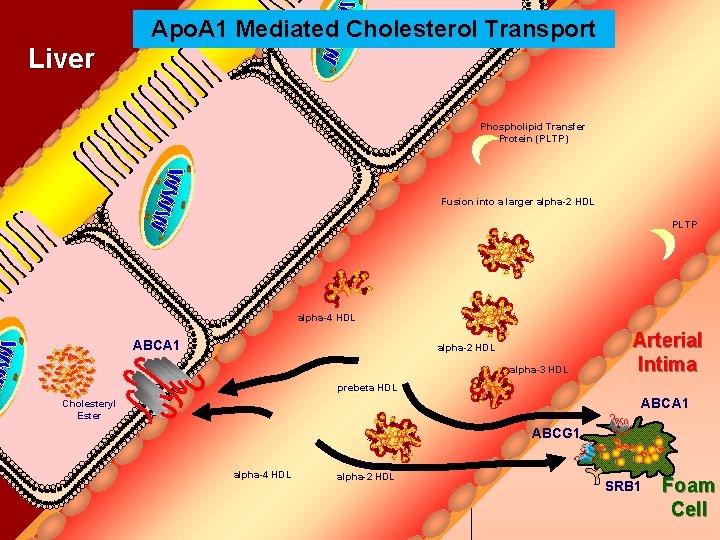
Apo. A 1 Mediated Cholesterol Transport Bile Duct Liver Phospholipid Transfer Protein (PLTP) Fusion into a larger alpha-2 HDL PLTP alpha-4 HDL ABCA 1 alpha-2 HDL alpha-3 HDL Arterial Intima prebeta HDL ABCA 1 Cholesteryl Ester ABCG 1 alpha-4 HDL alpha-2 HDL SRB 1 Foam Cell
Delipidation HDL (apo. A-I) Mediated Cholesterol Transport “Forward” Cholesterol Transport
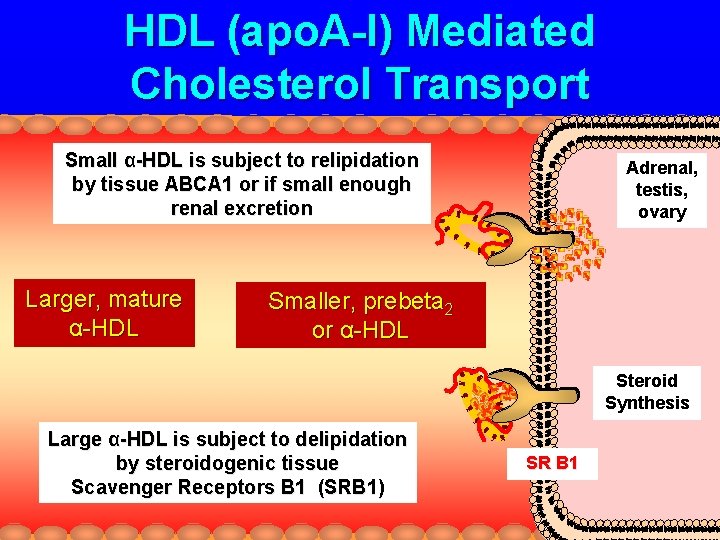
HDL (apo. A-I) Mediated Cholesterol Transport Small α-HDL is subject to relipidation by tissue ABCA 1 or if small enough renal excretion Larger, mature α-HDL Adrenal, testis, ovary Smaller, prebeta 2 or α-HDL Steroid Synthesis Large α-HDL is subject to delipidation by steroidogenic tissue Scavenger Receptors B 1 (SRB 1) SR B 1
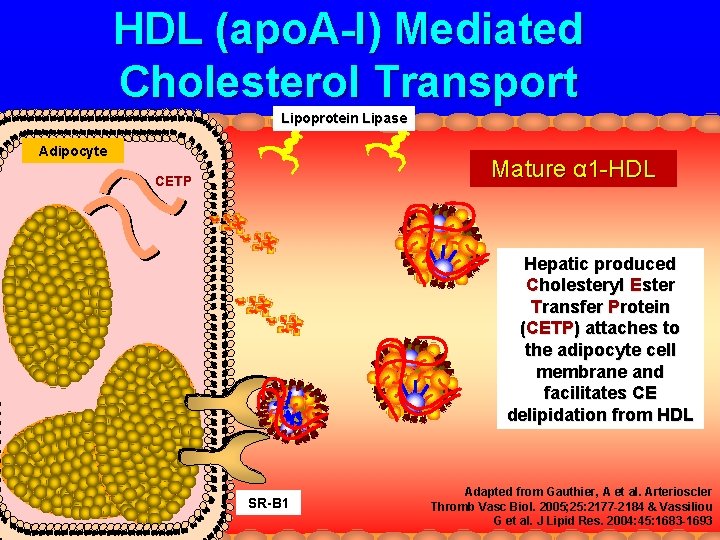
HDL (apo. A-I) Mediated Cholesterol Transport Lipoprotein Lipase Adipocyte Mature α 1 -HDL CETP Hepatic produced Cholesteryl Ester Transfer Protein (CETP) attaches to the adipocyte cell membrane and facilitates CE delipidation from HDL SR-B 1 Adapted from Gauthier, A et al. Arterioscler Thromb Vasc Biol. 2005; 25: 2177 -2184 & Vassiliou G et al. J Lipid Res. 2004: 45: 1683 -1693
Delipidation HDL (apo. A-I) Mediated Cholesterol Transport Direct Reverse Cholesterol Transport
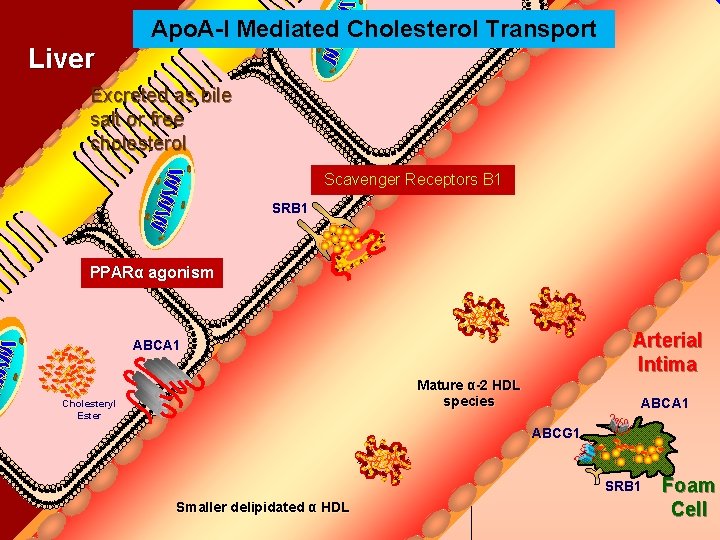
Apo. A-I Mediated Cholesterol Transport Bile Duct Liver Excreted as bile salt or free cholesterol Scavenger Receptors B 1 SRB 1 PPARα agonism Arterial Intima ABCA 1 Mature α-2 HDL species Cholesteryl Ester ABCA 1 ABCG 1 SRB 1 Smaller delipidated α HDL Foam Cell
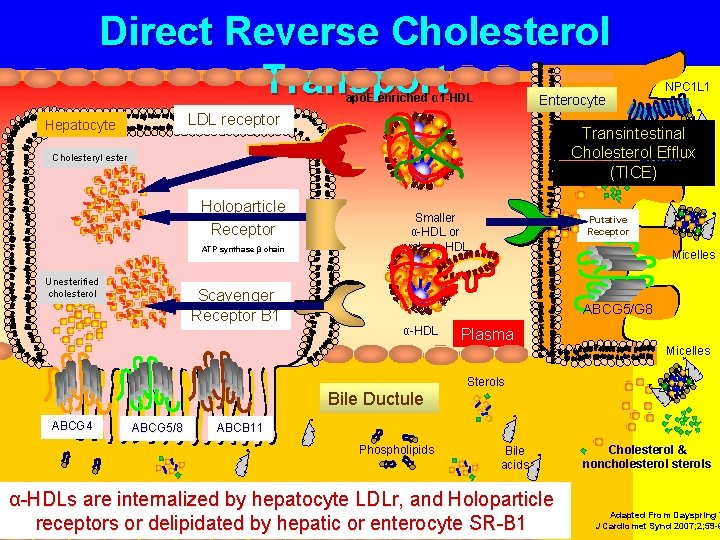
Direct Reverse Cholesterol Transport apo. E enriched α 1 -HDL Enterocyte LDL receptor Hepatocyte Transintestinal Cholesterol Efflux (TICE) Cholesteryl ester Holoparticle Receptor ATP synthase β chain Unesterified cholesterol Scavenger Receptor B 1 NPC 1 L 1 Smaller α-HDL or prebeta HDL Putative Receptor Micelles ABCG 5/G 8 α-HDL Plasma Micelles Bile Ductule ABCG 4 ABCG 5/8 Sterols ABCB 11 Phospholipids Bile acids α-HDLs are internalized by hepatocyte LDLr, and Holoparticle receptors or delipidated by hepatic or enterocyte SR-B 1 Cholesterol & noncholesterols Adapted From Dayspring T J Cardiomet Synd 2007; 2; 59 -6

Additional Delipidation Apo. A-I Mediated Cholesterol Transport Indirect Reverse Cholesterol Transport
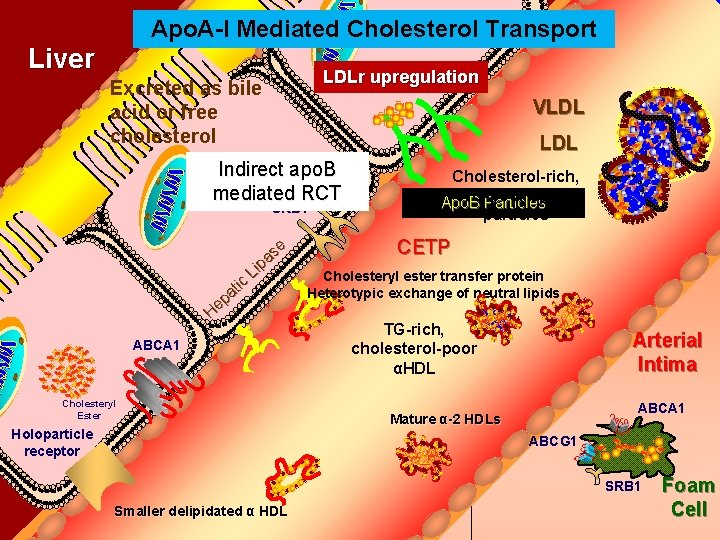
Apo. A-I Mediated Cholesterol Transport Bile Duct Liver LDLr upregulation Excreted as bile acid or free cholesterol VLDL Indirect apo. B mediated RCT SRB 1 tic a p He se a p Li ABCA 1 Cholesteryl Ester Holoparticle receptor Cholesterol-rich, TG-poor apo. B Apo. B Particles particles CETP Cholesteryl ester transfer protein Heterotypic exchange of neutral lipids TG-rich, cholesterol-poor αHDL Arterial Intima ABCA 1 Mature α-2 HDLs ABCG 1 SRB 1 Smaller delipidated α HDL Foam Cell
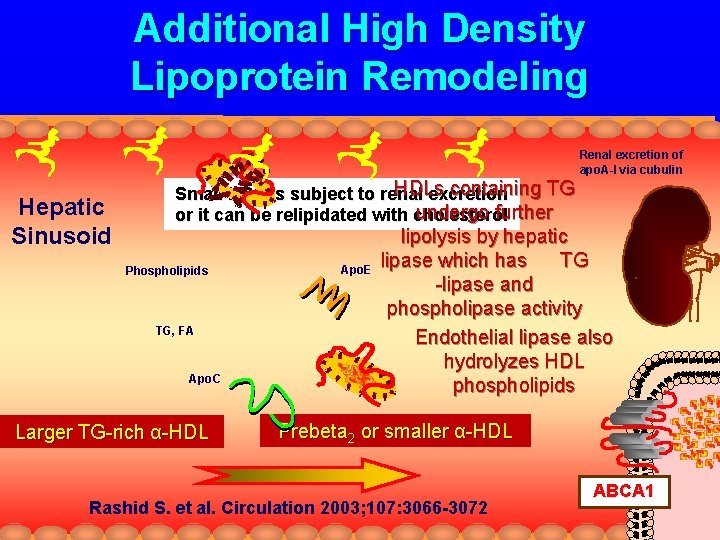
Additional High Density Lipoprotein Remodeling Renal excretion of apo. A-I via cubulin Hepatic Sinusoid HDLs containing TG Small HDL is subject to renal excretion undergo further or it can be relipidated with cholesterol Phospholipids TG, FA Apo. C Larger TG-rich α-HDL Apo. E lipolysis by hepatic lipase which has TG -lipase and phospholipase activity Endothelial lipase also hydrolyzes HDL phospholipids Prebeta 2 or smaller α-HDL Rashid S. et al. Circulation 2003; 107: 3066 -3072 ABCA 1
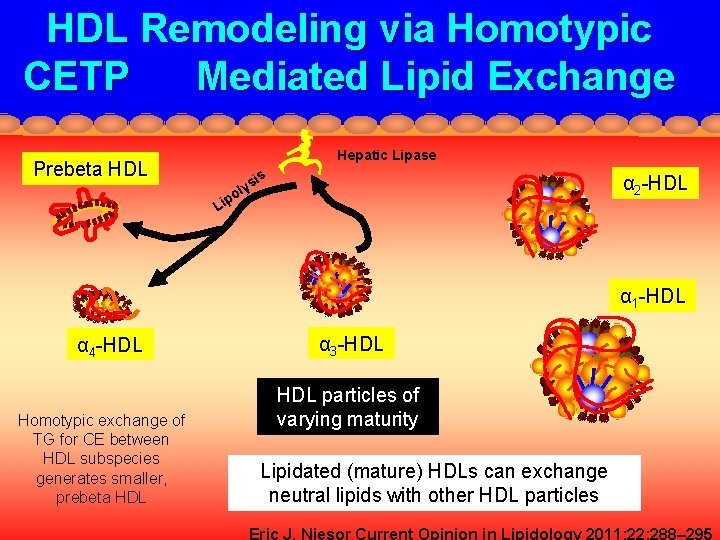
HDL Remodeling via Homotypic CETP Mediated Lipid Exchange Prebeta HDL Hepatic Lipase is ys l po Li α 2 -HDL α 1 -HDL α 4 -HDL Homotypic exchange of TG for CE between HDL subspecies generates smaller, prebeta HDL α 3 -HDL particles of varying maturity Lipidated (mature) HDLs can exchange neutral lipids with other HDL particles Eric J. Niesor Current Opinion in Lipidology 2011; 22: 288– 295
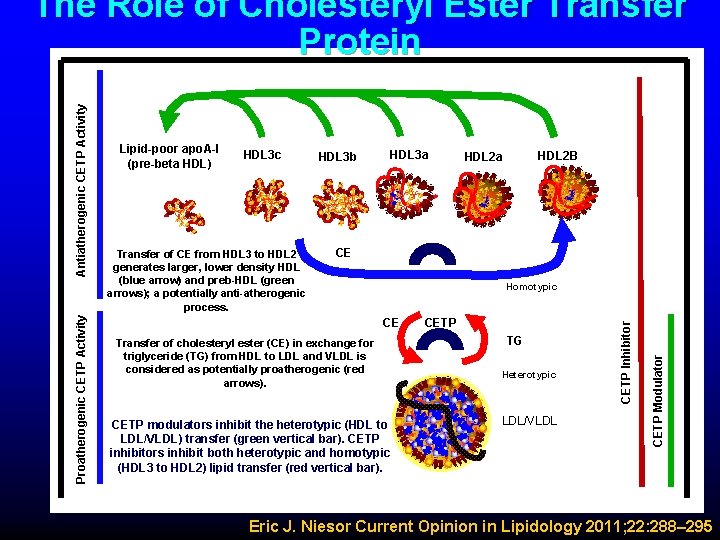
HDL 3 c Transfer of CE from HDL 3 to HDL 2 generates larger, lower density HDL (blue arrow) and preb-HDL (green arrows); a potentially anti-atherogenic process. HDL 3 b HDL 3 a HDL 2 B HDL 2 a CE Homotypic CE Transfer of cholesteryl ester (CE) in exchange for triglyceride (TG) from HDL to LDL and VLDL is considered as potentially proatherogenic (red arrows). CETP modulators inhibit the heterotypic (HDL to LDL/VLDL) transfer (green vertical bar). CETP inhibitors inhibit both heterotypic and homotypic (HDL 3 to HDL 2) lipid transfer (red vertical bar). CETP TG Heterotypic LDL/VLDL CETP Modulator Lipid-poor apo. A-I (pre-beta HDL) CETP Inhibitor Proatherogenic CETP Activity Antiatherogenic CETP Activity The Role of Cholesteryl Ester Transfer Protein Eric J. Niesor Current Opinion in Lipidology 2011; 22: 288– 295
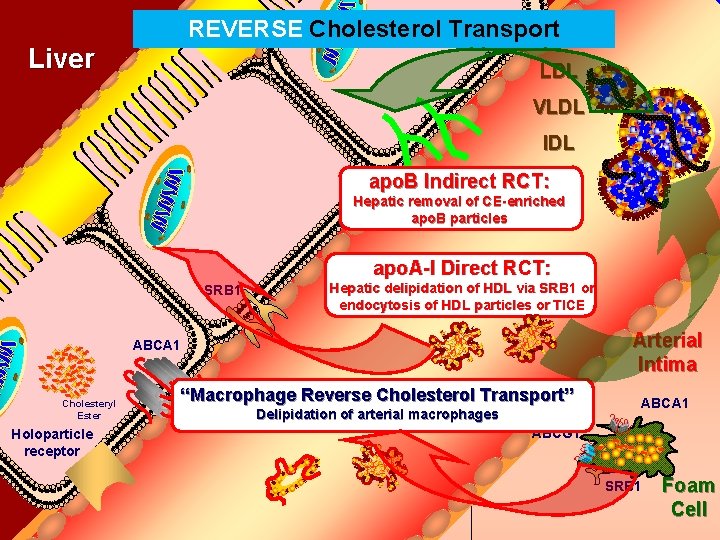
REVERSE Bile Duct Cholesterol Transport Liver LDL VLDL IDL apo. B Indirect RCT: Hepatic removal of CE-enriched apo. B particles apo. A-I Direct RCT: SRB 1 Hepatic delipidation of HDL via SRB 1 or endocytosis of HDL particles or TICE Arterial Intima ABCA 1 Cholesteryl Ester Holoparticle receptor “Macrophage Reverse Cholesterol Transport” Delipidation of arterial macrophages ABCA 1 ABCG 1 SRB 1 Foam Cell
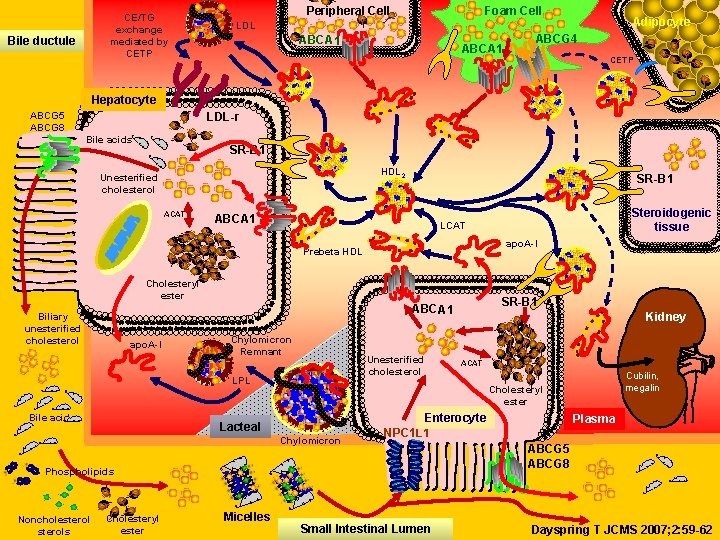
CE/TG exchange mediated by CETP Bile ductule Peripheral Cell Foam Cell Adipocyte LDL ABCA 1 ABCG 4 CETP Hepatocyte ABCG 5 ABCG 8 LDL-r Bile acids SR-B 1 HDL 2 Unesterified cholesterol ACAT SR-B 1 ABCA 1 apo. A-I Prebeta HDL Cholesteryl ester apo. A-I Chylomicron Remnant Unesterified cholesterol LPL Bile acids Lacteal Micelles ACAT Enterocyte NPC 1 L 1 Phospholipids Cholesteryl ester Kidney Cubilin, megalin Cholesteryl ester Chylomicron Noncholesterols SR-B 1 ABCA 1 Biliary unesterified cholesterol Steroidogenic tissue LCAT Small Intestinal Lumen Plasma ABCG 5 ABCG 8 Dayspring T JCMS 2007; 2: 59 -62
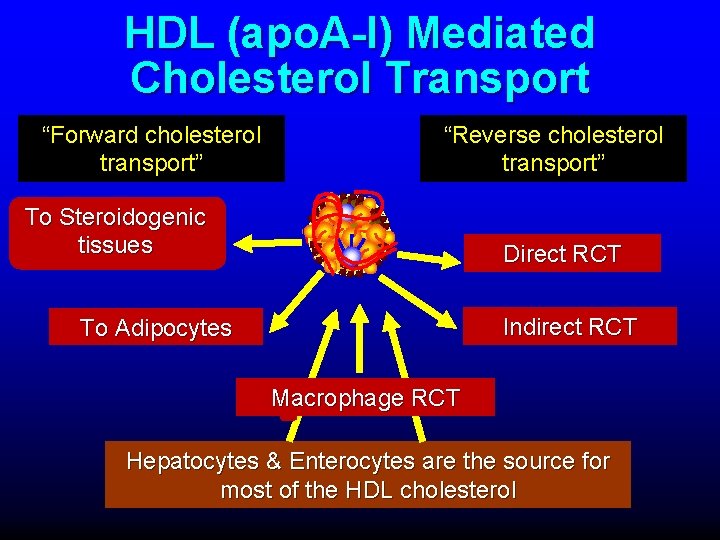
HDL (apo. A-I) Mediated Cholesterol Transport “Forward cholesterol transport” “Reverse cholesterol transport” To Steroidogenic tissues Direct RCT Indirect RCT To Adipocytes Macrophage RCT Hepatocytes & Enterocytes are the source for most of the HDL cholesterol

Plasma HDL-Cholesterol (HDL-C) ► The cholesterol content of HDL particles (HDL-C) depends on: ►apo. A-I production (hepatocyte, enterocyte) ►apo. A-I lipidation ► Hepatic, enterocytic, adipocyte, foam cell, peripheral cell via ABCA 1, SRB 1 ► PLTP and particle fusion ►apo. A-I delipidation ► Steroid gland SR-B 1 ► Hepatic SR-B 1, LDL receptor, holoparticle receptor ► CETP activity (heterotypic and homotypic) ► TICE ►apo. A-I removal ► Holoparticle or LDL receptor endocytosis ► Renal excretion ► Macrophage RCT does not contribute to significantly to HDL-C levels

Proteomic Analysis of HDL F F F To understand the role of HDL in biology it is essential to elucidate the make-up of the HDL particle, i. e. , its phospholipid and protein composition. Although apolipoproteins and other proteins involved in lipid metabolism may be the major constituents of HDL particles, there also a great number of known acute phase proteins on HDL. 56 proteins have been identified as associated with HDL. • Grouping of functionally related proteins present on HDL suggests the existence of clusters that perform specific tasks. F The role of these identified HDL-associated proteins in atherothrombosis awaits further investigation and may reveal new insights into the mechanism(s) of atherothrombotic diseases. Rezaee F et al. Proteomics 2006; 6: 721 -30

High Density Lipoproteins ► Apo. A-I overwhelmingly dominates the surface of the HDL particles which provide clues to how other HDL-associated proteins interact with these particles. ► HDL has been shown to contain some 35– 50 minor proteins in addition to apo. A-I and apo. A-II, and a majority of these are associated with the smallest or most dense HDL 3 c fractions. ► About 85% of the Lp. A-I 3 c particle surface is covered by apo. A-I. It is thus difficult to imagine how other proteins can find room to bind. ► One implication of this is that additional proteins may associate directly with apo. A-I and not the lipid surface, or ► They may form their own separate particles that happen to cofractionate in this density range. Huang R et al. Nat Struct Mol Biol. 2011 Apr; 18(4): 416 -22. Epub 2011 Mar 13
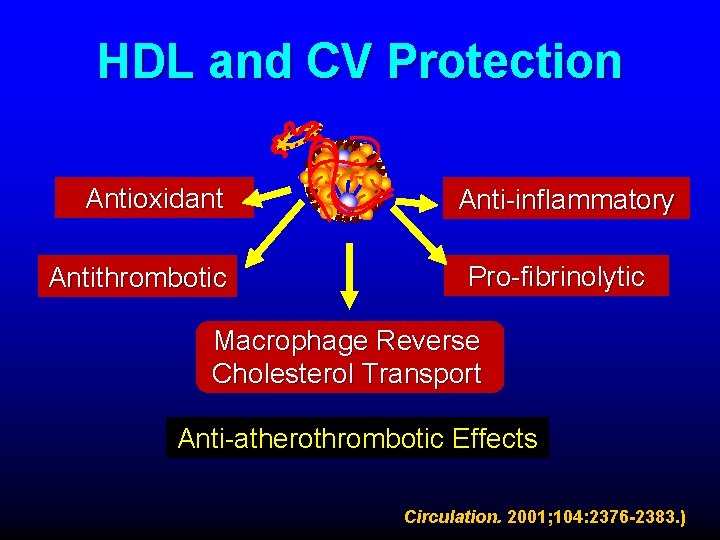
HDL and CV Protection Antioxidant Antithrombotic Anti-inflammatory Pro-fibrinolytic Macrophage Reverse Cholesterol Transport Anti-atherothrombotic Effects Circulation. 2001; 104: 2376 -2383. )
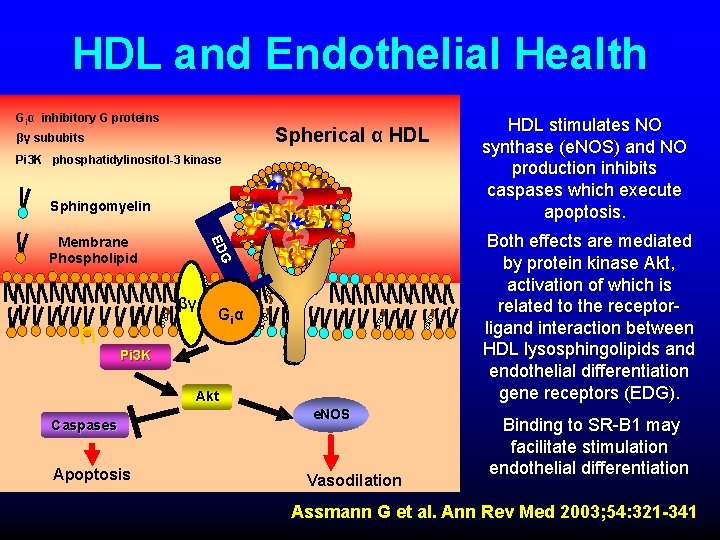
HDL and Endothelial Health Giα inhibitory G proteins Spherical α HDL βγ sububits Pi 3 K phosphatidylinositol-3 kinase Sphingomyelin βγ h Both effects are mediated by protein kinase Akt, activation of which is related to the receptorligand interaction between HDL lysosphingolipids and endothelial differentiation gene receptors (EDG). G ED Membrane Phospholipid Giα Pi 3 K Akt Caspases Apoptosis HDL stimulates NO synthase (e. NOS) and NO production inhibits caspases which execute apoptosis. e. NOS Vasodilation Binding to SR-B 1 may facilitate stimulation endothelial differentiation Assmann G et al. Ann Rev Med 2003; 54: 321 -341
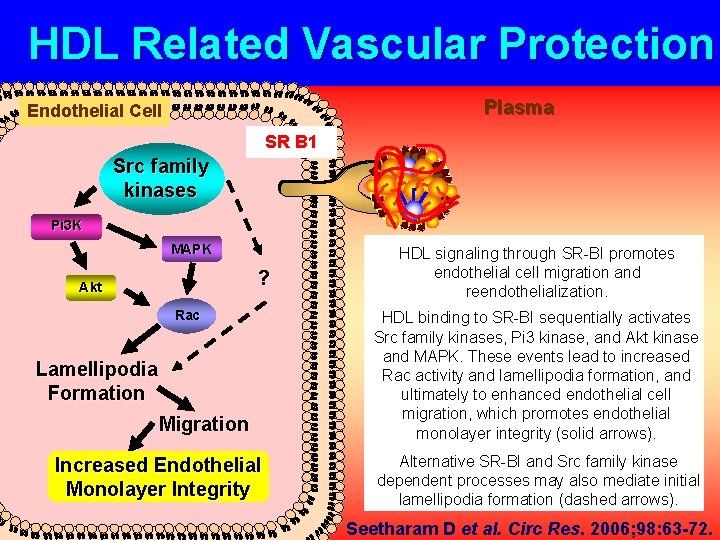
HDL Related Vascular Protection Plasma Endothelial Cell SR B 1 Src family kinases Pi 3 K MAPK ? Akt Rac Lamellipodia Formation Migration Increased Endothelial Monolayer Integrity HDL signaling through SR-BI promotes endothelial cell migration and reendothelialization. HDL binding to SR-BI sequentially activates Src family kinases, Pi 3 kinase, and Akt kinase and MAPK. These events lead to increased Rac activity and lamellipodia formation, and ultimately to enhanced endothelial cell migration, which promotes endothelial monolayer integrity (solid arrows). Alternative SR-BI and Src family kinase dependent processes may also mediate initial lamellipodia formation (dashed arrows). Seetharam D et al. Circ Res. 2006; 98: 63 -72.
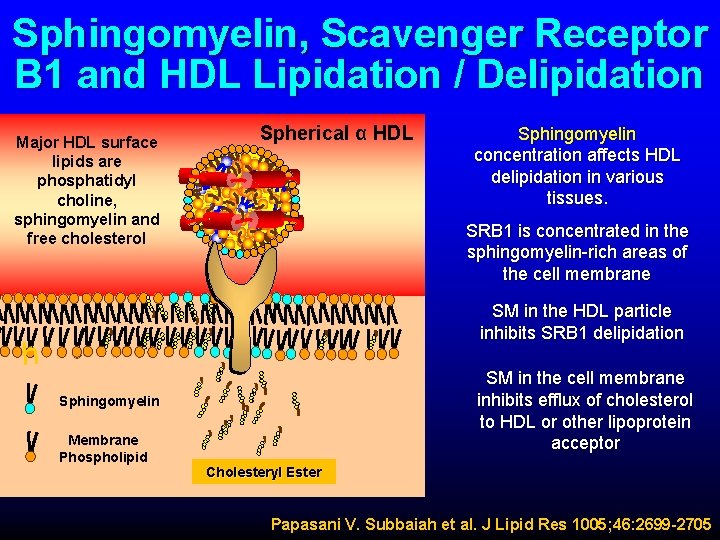
Sphingomyelin, Scavenger Receptor B 1 and HDL Lipidation / Delipidation Major HDL surface lipids are phosphatidyl choline, sphingomyelin and free cholesterol Spherical α HDL Sphingomyelin concentration affects HDL delipidation in various tissues. SRB 1 is concentrated in the sphingomyelin-rich areas of the cell membrane SM in the HDL particle inhibits SRB 1 delipidation h SM in the cell membrane inhibits efflux of cholesterol to HDL or other lipoprotein acceptor Sphingomyelin Membrane Phospholipid Cholesteryl Ester Papasani V. Subbaiah et al. J Lipid Res 1005; 46: 2699 -2705

HDL: Anti-inflammatory Activity LDL Phospholipids Fatty Acids O 2 - , OH- L CE OO -O X OX Oxidized phospholipids on LDL are deleterious. These are generated by seeding molecules like HPODE and HPETE as catalysts generated by 12 -liopoxygenase and lipid peroxides (LOOX) and oxidized CE (CE-OOX). LDL-ox HP HP OD ET E E A-I, J α-TOC HDL PON, PAF-AH GPX Oxidized Phospholipids (POVPC, PGPC) 12 -lipooxygenase HODE HETE The antioxidative effects of HDL are related to presence of α-tocopherol and other antioxidants including apo. A-I and apo. J. These scavenge ROS and prevent lipid peroxides (LOOX) and oxidized CE (CE-OOX). HDL overtakes seeding molecules and degrades them using paraoxonase (PON), PAF-AH or glutathione peroxidase (GPX) HPODE = Hydroperoxyoctadecadienoic acid HPETE = Hydroperoxycicosatetraenoic acid Assmann G et al. Ann Rev Med 2003; 54: 321 -341
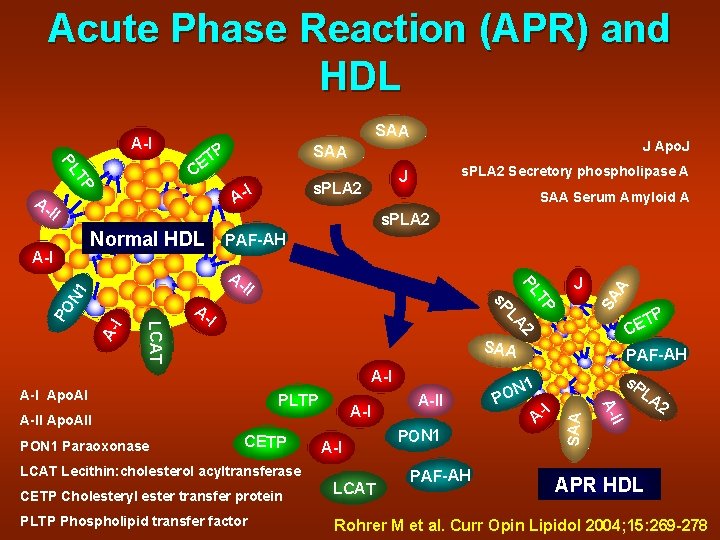
Acute Phase Reaction (APR) and HDL SAA C s. PLA 2 I A- SAA Serum Amyloid A s. PLA 2 Normal HDL PAF-AH II I PO A- 2 LA I SAA A-I PLTP A-I CETP A-I LCAT Lecithin: cholesterol acyltransferase PON 1 PAF-AH I A- P T CE PAF-AH s. P LA 2 I A-II Apo. AII A-II 1 N PO A-I Apo. AI PON 1 Paraoxonase P s. P LCAT A- J T PL A- N 1 A-I s. PLA 2 Secretory phospholipase A J SA A TP PL AII J Apo. J SAA P ET SAA A-I APR HDL CETP Cholesteryl ester transfer protein LCAT PLTP Phospholipid transfer factor Rohrer M et al. Curr Opin Lipidol 2004; 15: 269 -278
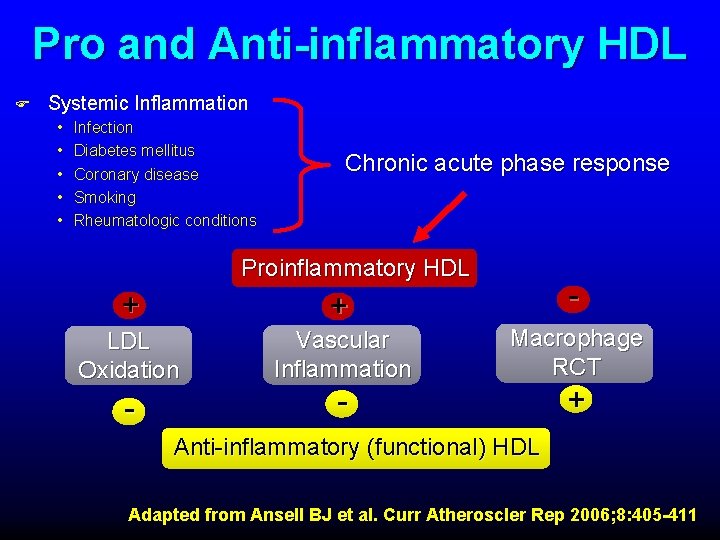
Pro and Anti-inflammatory HDL F Systemic Inflammation • • • Infection Diabetes mellitus Coronary disease Smoking Rheumatologic conditions Chronic acute phase response Proinflammatory HDL + + LDL Oxidation Vascular Inflammation - Macrophage RCT - + Anti-inflammatory (functional) HDL Adapted from Ansell BJ et al. Curr Atheroscler Rep 2006; 8: 405 -411

References ► Lagor WR & Rader DJ. Phospholipidation of HDL—How Much Is Too Much? Clinical Chemistry 2011; 57: 14– 6 ► Rothblat GH & Phillips MC. High-density lipoprotein heterogeneity and function in reverse cholesterol transport. Current Opinion in Lipidology 2010; 21: 229– 238 ► Eirini M. Tsompanidi et al. HDL biogenesis and functions: Role of HDL quality and quantity in atherosclerosis. Atherosclerosis 2010; 208: 3– 9 ► Cuchel M & Rader DJ. Macrophage Reverse Cholesterol Transport Key to the Regression of Atherosclerosis? Circulation. 2006; 113: 2548 -2555. ► van der Velde AE et al. Transintestinal cholesterol efflux Current Opinion in Lipidology 2010; 21: 167– 171 ► Rosenson RS et al, HDL Measures, Particle Heterogeneity, Proposed Nomenclature, and Relation to Atherosclerotic Cardiovascular Events. Clinical Chemistry 2011; 57: 3: 392– 410.
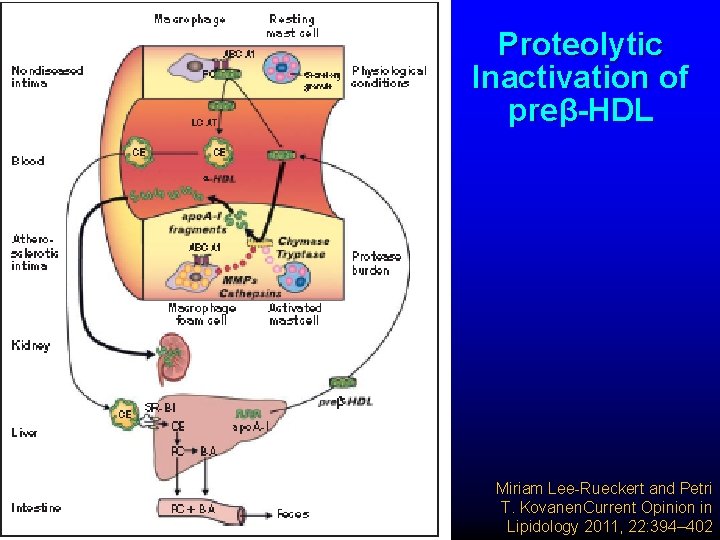
Proteolytic Inactivation of preβ-HDL Miriam Lee-Rueckert and Petri T. Kovanen. Current Opinion in Lipidology 2011, 22: 394– 402
- Slides: 53