HCV Retreatment Options and Adherence to Therapy Bruce

HCV Retreatment Options and Adherence to Therapy • Bruce R. Bacon, MD –James F. King MD Endowed Chair in Gastroenterology –Professor of Internal Medicine –Division of Gastroenterology and Hepatology –Saint Louis University Liver Center –Saint Louis University School of Medicine, St. Louis, Missouri 1

Agenda I. Program Overview and Opening Remarks II. Retreatment Approaches for Nonresponder and Relapsed HCV-Infected Patients – Case Studies/Q&A III. Importance of Adherence to Ribavirin (RBV) in HCV Treatment – Case Studies/Q&A IV. Closing Remarks 2

PROGRAM OVERVIEW AND OPENING REMARKS 3

Learning Objectives • Discuss the risks and benefits associated with a watchful waiting approach to nonresponders and relapsed patients • Define retreatment options for nonresponders and relapsed patients • Review the importance of RBV in optimizing combination therapy for HCV infection • Discuss ways to improve adherence to RBV in order to improve virologic outcomes 4

Housekeeping Information • This session is being recorded; please mute the ringer on your cell phone • If you would like to submit a question, please fill out a Q & A card • On your way out, please complete and return the evaluation form to receive credit 5

HCV Retreatment Options and Adherence to Therapy • Increasing numbers of patients have failed initial treatment • Several options are available – Retreat with the other pegylated interferon (Peg. IFN) – Retreat with consensus interferon (CIFN) – Watch and wait for the direct-acting antivirals (DAAs) • Necessity of RBV in combination therapy • Adherence to RBV is critical 6
Retreatment Approaches for Nonresponder and Relapsed HCV -Infected Patients • Brian Pearlman, MD, FACP • Center for Hepatitis C, Atlanta Medical Center, Atlanta, Georgia • Associate Clinical Professor Medicine, Emory School of Medicine, Atlanta, Georgia • Clinical Professor of Medicine, Medical College of Georgia, Augusta, Georgia 7

Patients Who Fail to Respond to Combination Treatment • 20 -50% of patients treated with Peg. IFN/RBV will not achieve a sustained virologic response (SVR) • Failure to achieve an SVR can be categorized as: – – Null response Partial response Relapse Breakthrough • Poor adherence to combination therapy and inappropriate dose reductions can contribute to Ghany MG, et al. Hepatology. 2009; 49: 1335 -1374. 8

Risks of Nonresponse and Relapse • Patients who do not achieve SVR remain at risk for progressive liver disease • Comparison of the 5 -yr occurrence of clinical events in patients with and without SVR: – Liver failure: 0% vs 13. 3%, respectively (P =. 001) – Liver-related death: 4. 4% vs 12. 9%, respectively (P =. 024) Veldt BJ et al. Ann Intern Med 2007; 147: 677 -684. 9
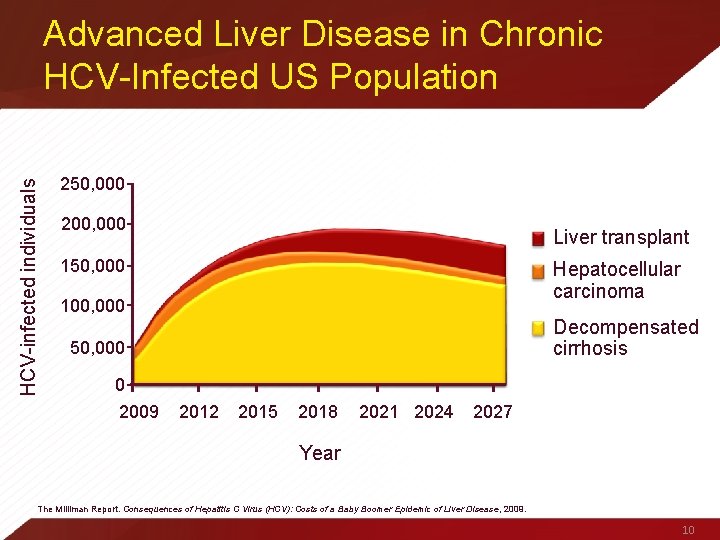
HCV-infected individuals Advanced Liver Disease in Chronic HCV-Infected US Population 250, 000 200, 000 Liver transplant 150, 000 Hepatocellular carcinoma 100, 000 Decompensated cirrhosis 50, 000 0 2009 2012 2015 2018 2021 2024 2027 Year The Milliman Report. Consequences of Hepatitis C Virus (HCV): Costs of a Baby Boomer Epidemic of Liver Disease, 2009. 10

Question Which of the following HCV nonresponders would you plan to retreat? a) 72 -yr-old female with advanced liver disease b) 24 -yr-old male with minimal liver disease c) 34 -yr-old pregnant woman with moderate liver disease d) 54 -yr-old male with stable bipolar disorder and advanced liver disease 11
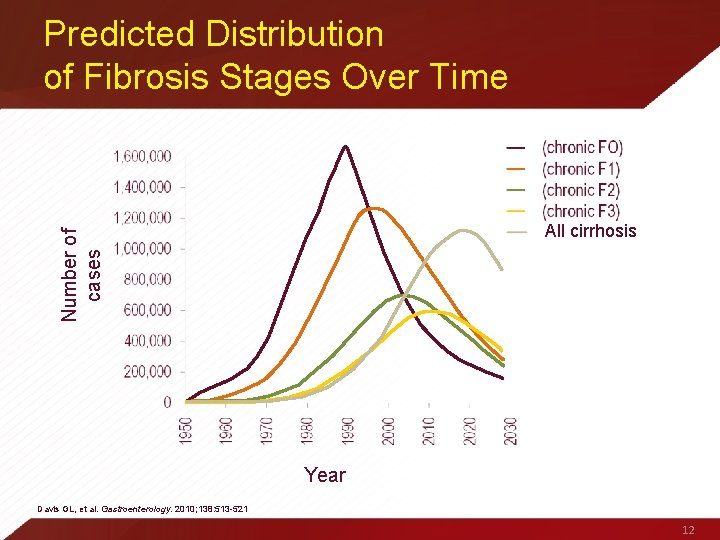
Predicted Distribution of Fibrosis Stages Over Time Number of cases All cirrhosis Year Davis GL, et al. Gastroenterology. 2010; 138: 513 -521 12
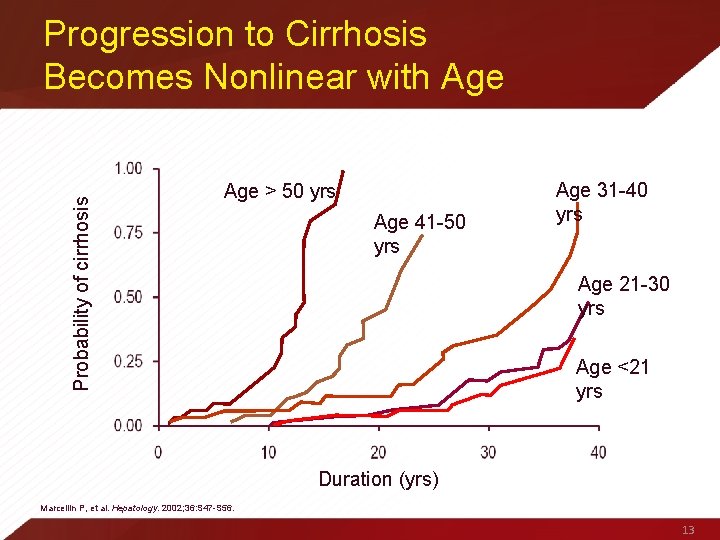
Probability of cirrhosis Progression to Cirrhosis Becomes Nonlinear with Age > 50 yrs Age 41 -50 yrs Age 31 -40 yrs Age 21 -30 yrs Age <21 yrs Duration (yrs) Marcellin P, et al. Hepatology. 2002; 36: S 47 -S 56. 13

Question How do you usually manage your patients that are Peg. IFN/RBV nonresponders? a) Maintenance Peg. IFN monotherapy b) Extended duration Peg. IFN/RBV combination therapy c) Induction Peg. IFN/RBV combination therapy followed by standard-dose Peg. IFN/RBV combination therapy d) CIFN/RBV combination therapy e) Watchful waiting 14

2009 AASLD Practice Guidelines* “Retreatment with peginterferon plus ribavirin in patients who did not achieve an SVR after a prior full course of peginterferon plus ribavirin is not recommended, even if a different type of peginterferon is administered” *Newer data, which will be discussed in this program (REPEAT, DIRECT, EPIC 3), were not available when the guidelines were published Ghany MG, et al. Hepatology. 2009; 49: 1335 -1374. 15

Options: Watchful Waiting vs Retreatment • Which patients should be monitored until the availability of different and newer medications? • Which patients should be retreated now with FDA-approved therapies? • Approach to patients who fail initial therapy depends on: – Nature of the initial response – Potency of initial treatment – Host–viral factors Ghany MG, et al. Hepatology. 2009; 49: 1335 -1374. 16

Factors Influencing Treatment Response Viral Factors Host Factors Genotype Quasispecies Viral escape mutants Pretreatment viral load Subtype Age Gender Ethnicity Body mass index Hepatic fibrosis Hepatic steatosis Insulin resistance LDL level Coinfection status Adherence IL-28 B genotype LDL, low-density lipoprotein. IL-28 B, interleukin-28, isoform B.
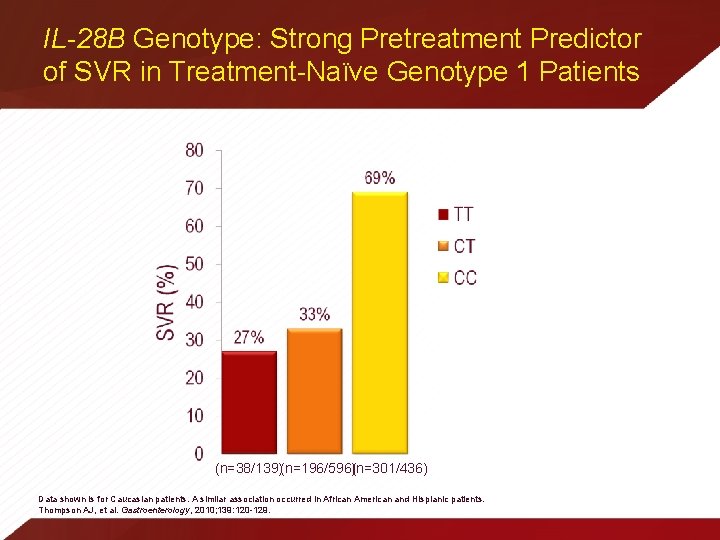
IL-28 B Genotype: Strong Pretreatment Predictor of SVR in Treatment-Naïve Genotype 1 Patients (n=38/139)(n=196/596)(n=301/436) Data shown is for Caucasian patients. A similar association occurred in African American and Hisplanic patients. Thompson AJ, et al. Gastroenterology, 2010; 139: 120 -129.

IL-28 B Genotype: New Information at 2010 AASLD • Sarrazin et al. , Abstract 1301 – SVR was achieved in 85%, 58%, and 46% of treatment-naïve, genotype 1 patients with CC, CT, and TT genotype, respectively – Overall relapse rates were 13%, 19%, and 21% in patients with CC, CT, and TT genotype, respectively • Asselah et al. , Abstract 9392 – Association of IL-28 B polymorphism with virologic response appears stronger in treatment-naïve patients compared with prior nonresponders – Suggests that other factors may mitigate the effect of IL 28 B polymorphism on outcome in nonresponder patients IL-28 B, Interleukin-28, isoform B 1. Sarrazin C, et al. Hepatology, 2010; 52: S 1, pages 320 A-421 A, Abstract 130. 2. Asselah T, et al. Hepatology, 2010; 52: S 1, pages 725 A-817 A, Abstract 939.
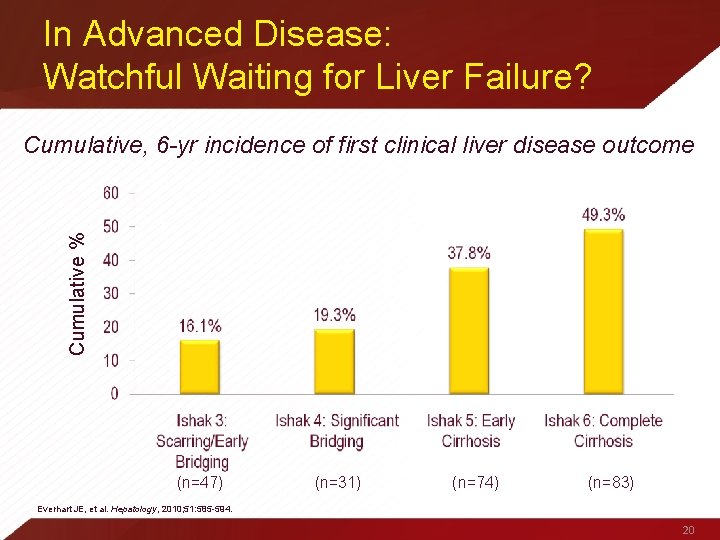
In Advanced Disease: Watchful Waiting for Liver Failure? Cumulative % Cumulative, 6 -yr incidence of first clinical liver disease outcome (n=47) (n=31) (n=74) (n=83) Everhart JE, et al. Hepatology, 2010; 51: 585 -594. 20

Take-Home Points • Morbidity and mortality associated with chronic HCV is projected to increase dramatically • Potential risks of watchful waiting should be discussed with the patient 21

RETREATMENT OPTIONS FOR NONRESPONDERS AND RELAPSERS 22

Case 1 • 54 -yr-old man with chronic HCV infection is sent to you for consideration for retreatment • He has HCV genotype 1 a • His HCV RNA level is 2, 350, 000 IU/m. L • His prior treatment was Peg. IFNα-2 a 180 µg qwk + RBV for 12 wks 23

Case 1 Question What additional information do you want to have? a) Findings on his pre-treatment biopsy b) His prior RBV dose and compliance history c) The type of virological response to prior therapy d) a, b, and c e) a and b f) b and c 24

Key Questions for HCV Patients Facing Retreatment 1. What were you treated with, at what dosage, and for how long? 2. What type of response did you have? 3. What is your genotype? 4. Did you require dose reductions or treatment interruptions during previous therapy? What caused these interruptions/dose reductions? 5. Did you have adherence issues? 6. Were you overweight during previous courses of therapy? 7. Do you have any significant comorbidities/other conditions that may affect your response to treatment? 8. Did you have a good support system during previous courses of treatment? 25 Do you have one now?

Case 2 • 42 -yr-old white female with chronic HCV referred to you for retreatment • Patient achieved a 2 -log 10 HCV RNA decline on Peg. IFN -2 b/RBV at wk 12, but had detectable HCV RNA at wk 24 • BMI: 26 • Labs: genotype 1 b, ALT 169 U/m. L • Biopsy: grade 3, stage 3 (A 3 F 3, METAVIR) 26

Case 2 Question How would you manage this patient? a) Start Peg. IFN maintenance monotherapy b) Retreat with the other Peg. IFN c) Retreat with CIFN/RBV d) Watch and wait for DAAs to be approved 27
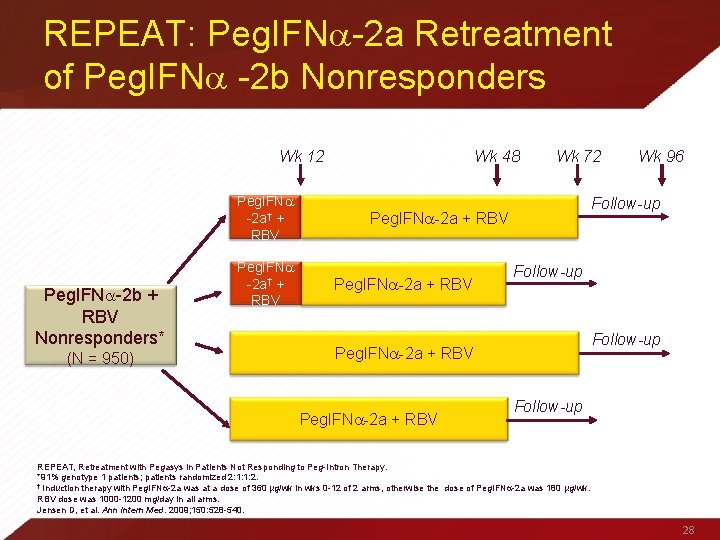
REPEAT: Peg. IFN -2 a Retreatment of Peg. IFN -2 b Nonresponders Wk 12 Peg. IFN -2 a† + RBV Peg. IFN -2 b + RBV Nonresponders* (N = 950) Peg. IFN -2 a† + RBV Wk 48 Wk 72 Follow-up Peg. IFN -2 a + RBV Wk 96 Follow-up REPEAT, Retreatment with Pegasys in Patients Not Responding to Peg-Intron Therapy. *91% genotype 1 patients; patients randomized 2: 1: 1: 2. † Induction therapy with Peg. IFN -2 a was at a dose of 360 µg/wk in wks 0 -12 of 2 arms, otherwise the dose of Peg. IFN -2 a was 180 µg/wk. RBV dose was 1000 -1200 mg/day in all arms. Jensen D, et al. Ann Intern Med. 2009; 150: 528 -540. 28
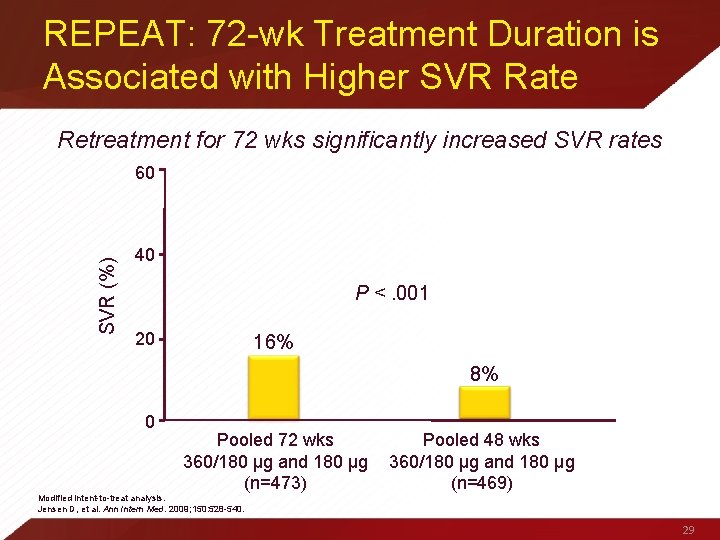
REPEAT: 72 -wk Treatment Duration is Associated with Higher SVR Rate Retreatment for 72 wks significantly increased SVR rates SVR (%) 60 40 P <. 001 20 16% 8% 0 Pooled 72 wks 360/180 µg and 180 µg (n=473) Pooled 48 wks 360/180 µg and 180 µg (n=469) Modified intent-to-treat analysis. Jensen D, et al. Ann Intern Med. 2009; 150: 528 -540. 29

EPIC 3: Peg. IFN -2 b Retreatment of Nonresponders and Relapsers Nonresponders and relapsers with significant fibrosis/cirrhosis (N = 2293) Peg. IFN -2 b + RBV Wk 12 Detectable HCV RNA Enter maintenance study Wk 18 Wk 48 Detectable HCV RNA Peg. IFN -2 b + RBV EPIC 3, Evaluation of Peg. Intron in Control of Hepatitis C Cirrhosis. EPIC 3 was a prospective, international, multicenter, open-label study. Peg. IFN -2 b dose was 1. 5 µg/kg/wk. RBVdose was 800 -1400 mg/day based on body weight. Poynard T, et al. Gastroenterology. 2009; 136: 1618 -1628. 30
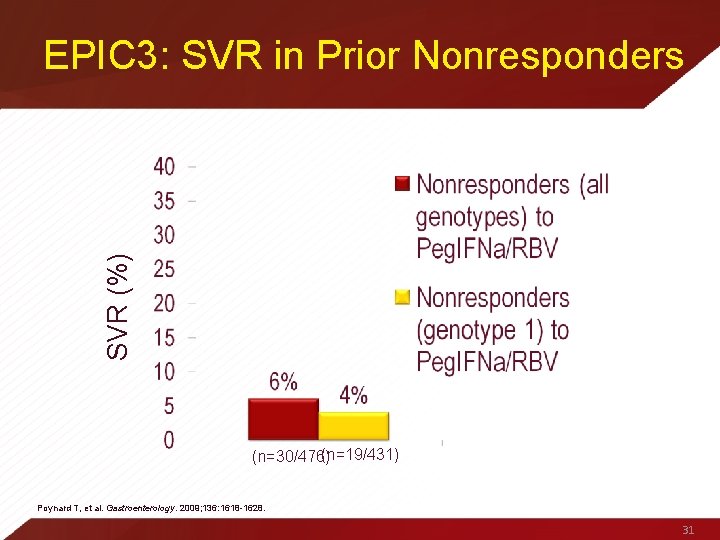
SVR (%) EPIC 3: SVR in Prior Nonresponders (n=19/431) (n=30/476) Poynard T, et al. Gastroenterology. 2009; 136: 1618 -1628. 31
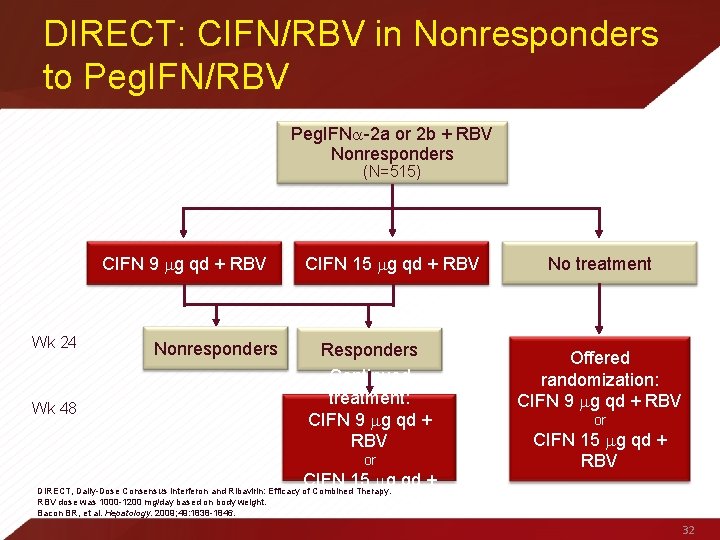
DIRECT: CIFN/RBV in Nonresponders to Peg. IFN/RBV Peg. IFN -2 a or 2 b + RBV Nonresponders (N=515) CIFN 9 g qd + RBV Wk 24 Wk 48 Nonresponders CIFN 15 g qd + RBV Responders Continued treatment: CIFN 9 g qd + RBV or CIFN 15 g qd + RBV No treatment Offered randomization: CIFN 9 g qd + RBV or CIFN 15 g qd + RBV DIRECT, Daily-Dose Consensus Interferon and Ribavirin: Efficacy of Combined Therapy. RBV dose was 1000 -1200 mg/day based on body weight. Bacon BR, et al. Hepatology. 2009; 49: 1838 -1846. 32
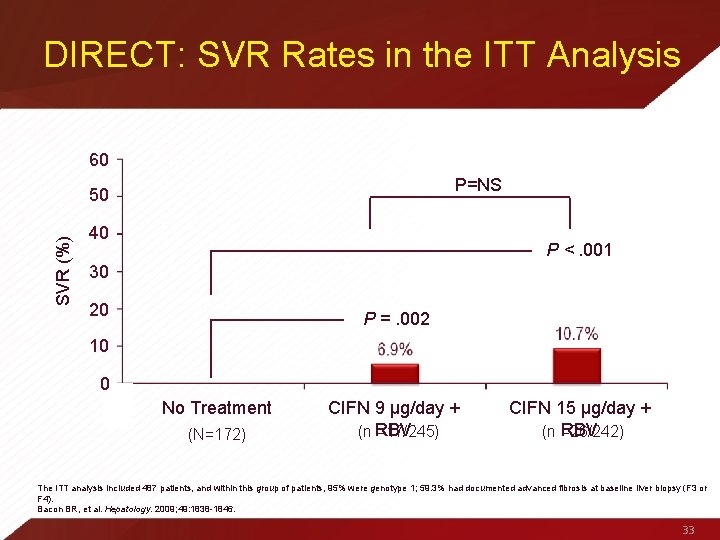
DIRECT: SVR Rates in the ITT Analysis 60 P=NS SVR (%) 50 40 P <. 001 30 20 P =. 002 10 0 No Treatment (N=172) CIFN 9 μg/day + (n RBV =17/245) CIFN 15 μg/day + RBV (n =26/242) The ITT analysis included 487 patients, and within this group of patients, 95% were genotype 1; 59. 3% had documented advanced fibrosis at baseline liver biopsy (F 3 or F 4). Bacon BR, et al. Hepatology. 2009; 49: 1838 -1846. 33
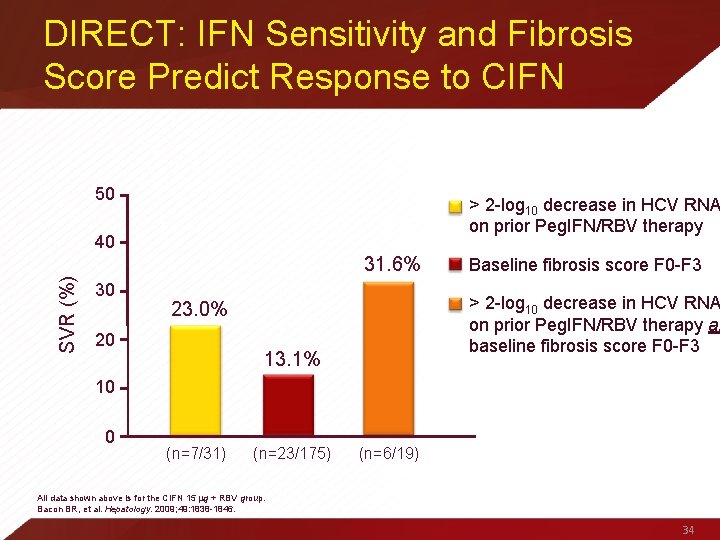
DIRECT: IFN Sensitivity and Fibrosis Score Predict Response to CIFN 50 > 2 -log 10 decrease in HCV RNA on prior Peg. IFN/RBV therapy 40 SVR (%) 31. 6% 30 > 2 -log 10 decrease in HCV RNA on prior Peg. IFN/RBV therapy an baseline fibrosis score F 0 -F 3 23. 0% 20 Baseline fibrosis score F 0 -F 3 13. 1% 10 0 (n=7/31) (n=23/175) (n=6/19) All data shown above is for the CIFN 15 g + RBV group. Bacon BR, et al. Hepatology. 2009; 49: 1838 -1846. 34

Case 3 • 36 -yr-old Hispanic female with high BMI (34) referred to you because she wants to be retreated for her HCV infection • Patient treated previously with 48 wks of Peg. IFN 2 b/RBV but never had undetectable viremia despite good adherence to therapy • Achieved a 2 -log 10 drop in HCV RNA at 12 wks, but had detectable HCV RNA by wk 24 • Labs: genotype 1 a, HCV RNA 2, 365, 879 IU/m. L • Biopsy: grade 2, stage 2 (A 2 F 2 METAVIR) • Patient begins retreatment with daily CIFN/RBV 35

Case 3 Question Based upon available data what is the most appropriate time to recheck HCV RNA to determine response in this prior nonresponder? a) Wk 4 b) Wk 8 c) Wk 12 d) Wk 24 36
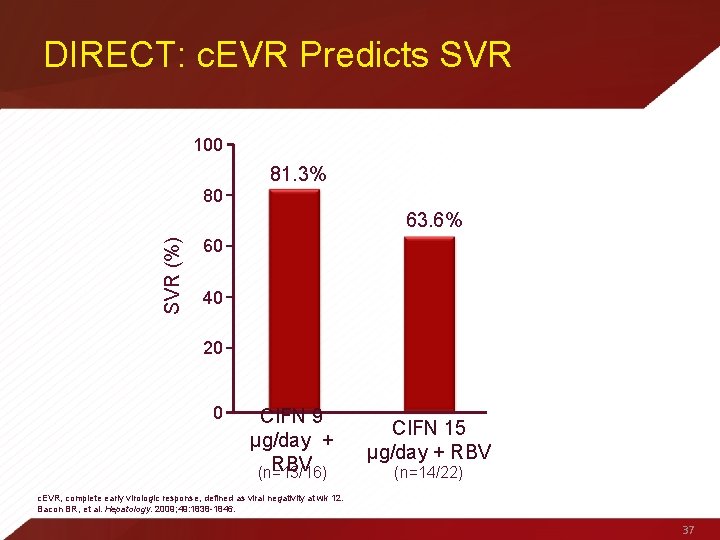
DIRECT: c. EVR Predicts SVR 100 81. 3% 80 SVR (%) 63. 6% 60 40 20 0 CIFN 9 μg/day + RBV (n=13/16) CIFN 15 μg/day + RBV (n=14/22) c. EVR, complete early virologic response, defined as viral negativity at wk 12. Bacon BR, et al. Hepatology. 2009; 49: 1838 -1846. 37

DIRECT: Safety and Tolerability • Most AEs were related to administration of both CIFN and RBV • AEs resulting from CIFN were typical of those with IFNbased therapy – Neutropenia • Most common – Fatigue – Leukopenia groups – Depression – Nausea AEs leading to dose modifications in CIFN – Myalgia – Lymphopenia – Anemia • Overall occurrence of discontinuations for AEs* – 14% in the 9 g group – 21% in the 15 g group AE, adverse event. *in the pooled ITT analysis. Bacon BR, et al. Hepatology. 2009; 49: 1838 -1846. 38

Relapse Is Common in HCV-Infected Patients Treated With Peg. IFN /RBV • Up to 18% of treatment-naïve patients of all genotypes will relapse 1 • ~1/3 of treatment-naïve genotype 1–infected patients will relapse 2 • Relapsers are good candidates for retreatment 3 – Better chance of response compared with prior nonresponders • Beware discordance between the more sensitive TMA HCV RNA qualitative assay and less sensitive PCR-based assays for determining end-of-treatment response 4, 5 TMA, transcription-mediated amplification. 1. Manns M, et al. Lancet. 2001; 358: 958 -965. 2. Mc. Hutchison JG, et al. N Engl J Med. 2009; 361: 580 -593. 3. Poynard T, et al. Gastroenterology. 2009; 136: 1618 -1628. 4. Gerotto M. J Hepatol. 2006; 44: 83 -87. 5. Roomer R, et al. Hepatology, 2010; 52: S 1, pages 725 A-817 A, Abstract 1015.
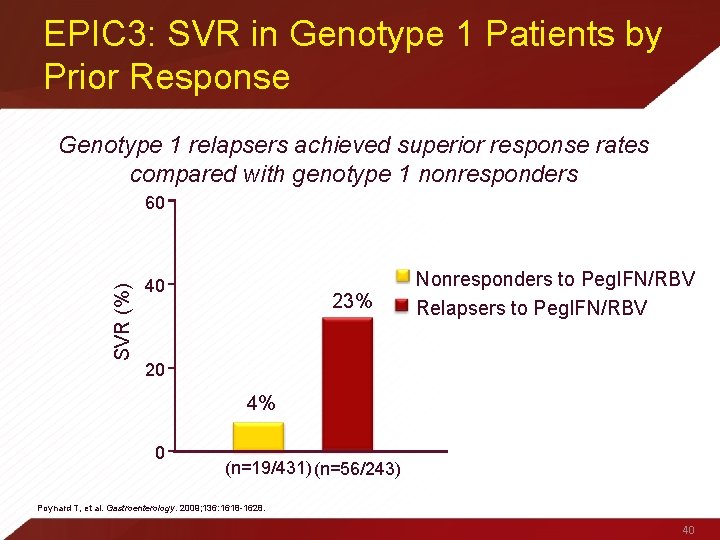
EPIC 3: SVR in Genotype 1 Patients by Prior Response Genotype 1 relapsers achieved superior response rates compared with genotype 1 nonresponders SVR (%) 60 40 23% Nonresponders to Peg. IFN/RBV Relapsers to Peg. IFN/RBV 20 4% 0 (n=19/431) (n=56/243) Poynard T, et al. Gastroenterology. 2009; 136: 1618 -1628. 40
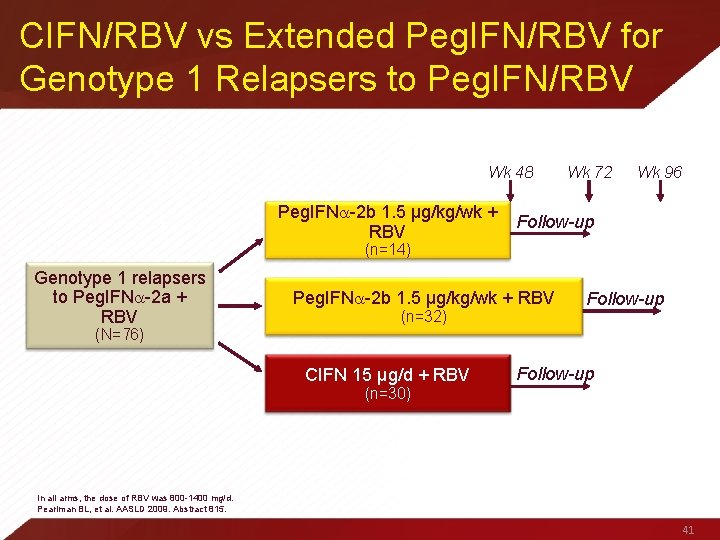
CIFN/RBV vs Extended Peg. IFN/RBV for Genotype 1 Relapsers to Peg. IFN/RBV Wk 48 Wk 72 Wk 96 Peg. IFN -2 b 1. 5 µg/kg/wk + Follow-up RBV (n=14) Genotype 1 relapsers to Peg. IFN -2 a + RBV Peg. IFN -2 b 1. 5 µg/kg/wk + RBV (n=32) Follow-up (N=76) CIFN 15 µg/d + RBV Follow-up (n=30) In all arms, the dose of RBV was 800 -1400 mg/d. Pearlman BL, et al. AASLD 2009. Abstract 815. 41
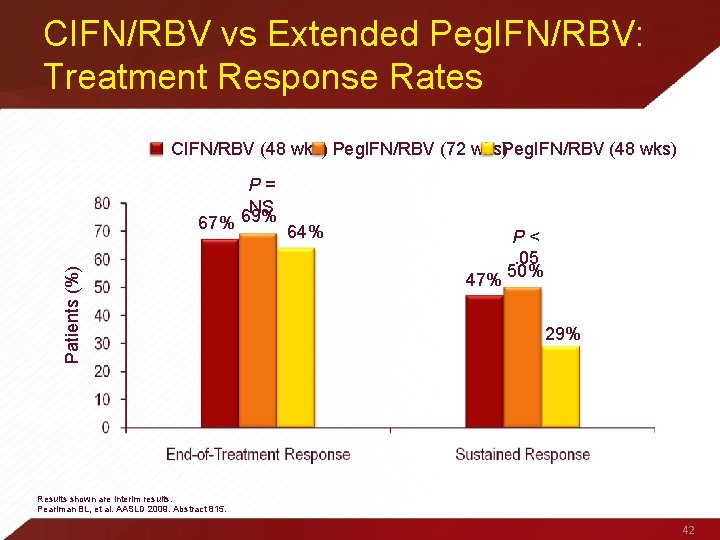
CIFN/RBV vs Extended Peg. IFN/RBV: Treatment Response Rates CIFN/RBV (48 wks) Peg. IFN/RBV (72 wks)Peg. IFN/RBV (48 wks) Patients (%) P= NS 69% 67% 64% 47% P<. 05 50% 29% Results shown are interim results. Pearlman BL, et al. AASLD 2009. Abstract 815. 42

Take-Home Points • When considering retreatment, several factors must be examined – HCV genotype – Previous therapy and degree of response • Consider in adherent, relapsing patients – Stage of liver disease – Issues of nonadherence, dose-reductions, and/or treatment interruptions – Comorbidities • Consider extended-duration Peg/RBV – Be aware of increased voluntary discontinuations and sideeffects • CIFN 15 g daily + RBV is FDA-approved as retreatment – Most beneficial for noncirrhotics (F 0 -F 3) with > 2 log decrease 43

FUTURE RETREATMENT OPTIONS FOR NONRESPONDERS AND RELAPSERS 44

Case 1 Revisited 54 -year-old man with HCV genotype 1 a infection is sent to you for retreatment, current HCV RNA is 2, 350, 000 IU/m. L Additional information is obtained: • Prior treatment was with Peg. IFNα-2 a 180 µg qwk + RBV for 12 wks – Pretreatment HCV RNA was 2, 800, 000 IU/m. L – 4 -wk HCV RNA was 1, 200, 000 IU/m. L – 12 -wk HCV RNA was 105, 000 IU/m. L – Received weight-based RBV, was not dose-reduced, and was compliant • Liver biopsy 2 yrs ago showed mild-to-moderate inflammatory activity with bridging fibrosis and nodularity 45

Case 1 Question How would you proceed at this point? a) Retreat with Peg. IFN -2 b + weight-based RBV b) Trial of CIFN at 15 µg/day + weight-based RBV c) Wait for DAAs to be approved d) Repeat liver biopsy 46

PROVE 3: Telaprevir (TVR) in Nonresponders and Relapsers to Peg. IFN/RBV Wk 12 T 12 PR 24 Peg. IFN/RBV/T VR (n=115) T 24 PR 48 (n=113) Wk 24 Wk 48 Peg. IFN/RBV/TVR Peg. IFN/RBV Genotype 1 nonresponders and relapsers to Peg. IFN/RBV T 24 P 24 Peg. IFN/TVR (no RBV) (n=111) PR 48 Peg. IFN/RBV (n=114) PROVE, Protease Inhibition for Viral Evaluation. Peg. IFN -2 a dose was 180 μg/wk. RBV dose was 1000 or 1200 mg/d according to body weight. TVR dose was 1125 -mg loading dose, then 750 mg every 8 hrs. Mc. Hutchison JM, et al. N Engl J Med. 2010; 362: 1292 -1303. 47
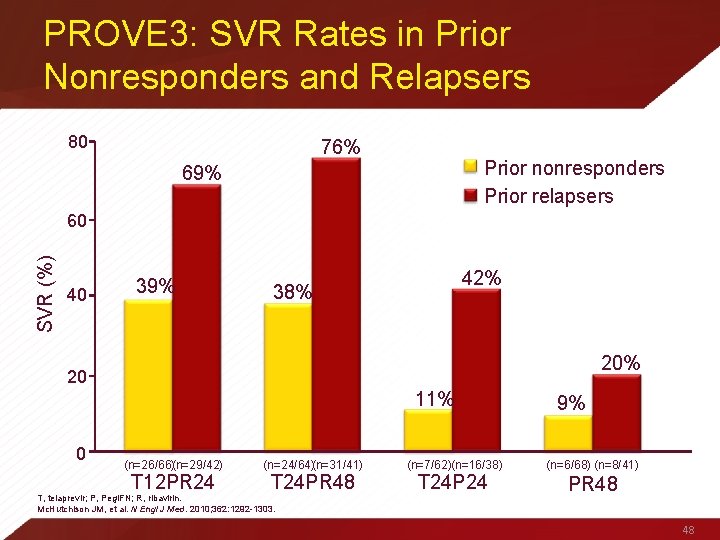
PROVE 3: SVR Rates in Prior Nonresponders and Relapsers 80 76% Prior nonresponders Prior relapsers 69% SVR (%) 60 40 39% 42% 38% 20 11% 0 (n=26/66)(n=29/42) T 12 PR 24 (n=24/64)(n=31/41) T 24 PR 48 T, telaprevir; P, Peg. IFN; R, ribavirin. Mc. Hutchison JM, et al. N Engl J Med. 2010; 362: 1292 -1303. (n=7/62)(n=16/38) T 24 P 24 9% (n=6/68) (n=8/41) PR 48 48

PROVE 3: Safety • AEs for which 1 or more of the TVR groups had incidence 10% higher than the PR 48 group: – – Rash Pruritus Nausea Diarrhea – Hemorrhoi – Alopecia ds – Insomnia – Pyrexia – Anemia – Fatigue • Severe AEs requiring therapy discontinuation occurred in 24% of patients in T 24 PR 48 arm • Patients treated with TVR were more likely to discontinue treatment because of an AE than were patients treated with Peg. IFN/RBV – 15% vs 4% Mc. Hutchison JM, et al. N Engl J Med. 2010; 362: 1292 -1303. 49
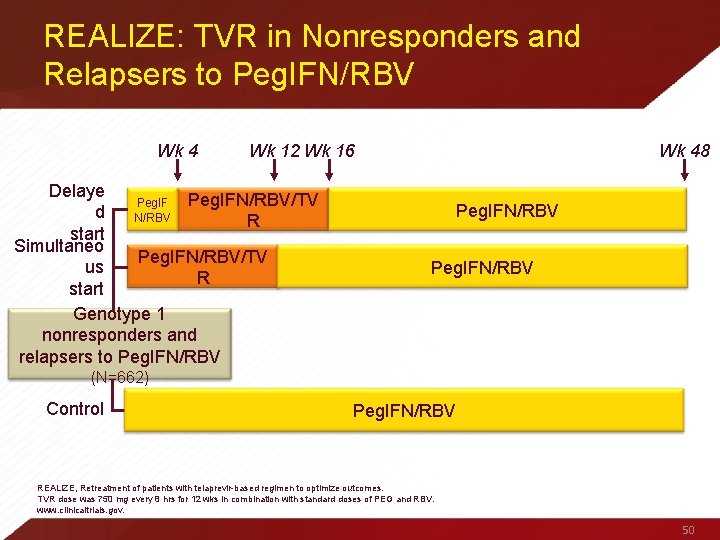
REALIZE: TVR in Nonresponders and Relapsers to Peg. IFN/RBV Wk 4 Wk 12 Wk 16 Delaye Peg. IFN/RBV/TV Peg. IF d N/RBV R start Simultaneo Peg. IFN/RBV/TV us R start Genotype 1 nonresponders and relapsers to Peg. IFN/RBV Wk 48 Peg. IFN/RBV (N=662) Control Peg. IFN/RBV REALIZE, Retreatment of patients with telaprevir-based regimen to optimize outcomes. TVR dose was 750 mg every 8 hrs for 12 wks in combination with standard doses of PEG and RBV. www. clinicaltrials. gov. 50
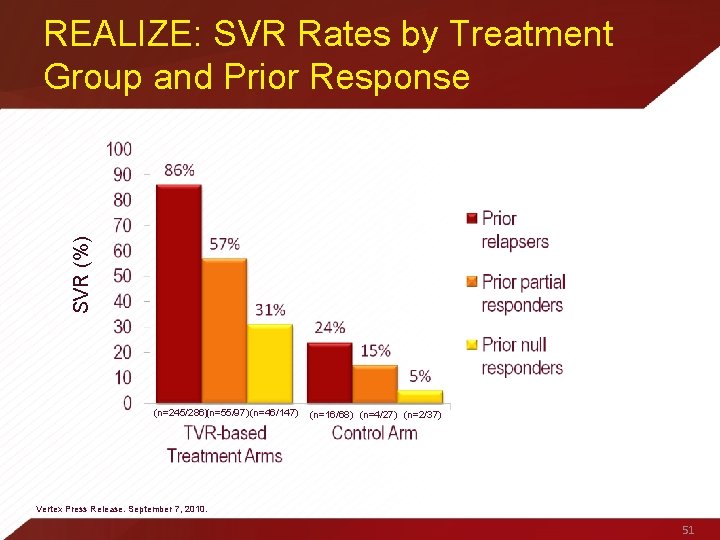
SVR (%) REALIZE: SVR Rates by Treatment Group and Prior Response (n=245/286)(n=55/97) (n=46/147) (n=16/68) (n=4/27) (n=2/37) Vertex Press Release. September 7, 2010. 51

RESPOND-2: Boceprevir (BOC) in Nonresponders and Relapsers to Peg. IFN/RBV Wk 4 Arm 1 (Contr ol) Genotype 1 nonresponders and relapsers to Peg. IFN/RBV Wk 48 Peg. IFN/RBV (N=403) Arm 2 (RGT*) Peg. IF NRBV Peg. IFN/RBV/BOC for 32 -44 wks Arm 3 Peg. IF NRBV Peg. IFN/RBV/BOC for 44 wks *RGT, Response-guided therapy, based on HCV negativity at wk 8: a) HCV-negative patients received 28 more wks of Peg. IFN/RBV/BOC; b) HCV-positive patients received 28 more wks of Peg. IFN/RBV/BOC followed by 12 wks of Peg. IFN/RBV. Peg. IFN dose in all arms was 1. 5 g/kg. RBV dose in all arms was 600 -1400 mg. BOC dose in Arms 2 and 3 was 800 mg TID. Bacon B, et al. Hepatology, 2010; 52: S 1, pages 422 A-522 A, Abstract 216.

RESPOND-2: Safety and Tolerability • Discontinuation due to AEs was reported in 3%, 8% and 12% of patients in Arms 1, 2, and 3, respectively • No discontinuations were related to rash • Most common reason for discontinuation was lack of response at wk 12 Bacon B, et al. Hepatology, 2010; 52: S 1, pages 422 A-522 A, Abstract 216.

Take-Home Points • Studies of DAAs have yielded promising results in terms of higher cure rates and shorter duration of therapy 1, 2 • These results are partially offset by 1, 2 – – New issues of resistance Increased side effects Increased pill burden Studies have been limited to patients infected with genotype 1 • RBV is required to maximize SVR with DAAs and limit resistance 1, 2 1. 2. Michaels AJ et al. Curr Opin Gastroenterol. 2010; 26: 196 -201. Naggie S et al. J Antimicrob Chemother. 2010; 65: 2063 -2069. 54
- Slides: 54