HCLPG 03 Phase II Trial of Vemurafenib Rituximab
HCL-PG 03: Phase II Trial of Vemurafenib + Rituximab in Patients With Relapsed/ Refractory Hairy Cell Leukemia Integrating New Hematology Findings Into Practice: Independent Conference Coverage of ASH 2017, * December 9 -12, Atlanta, Georgia *CCO is an independent medical education company that provides state-of-the-art medical information to healthcare professionals through conference coverage and other educational programs. This activity is supported by educational grants from Abb. Vie; Astra. Zeneca; Celgene Corporation; Genentech; Janssen Biotech, Inc administered by Janssen Scientific Affairs, LLC; Jazz Pharmaceuticals; Novartis Pharmaceuticals Corporation; Pharmacyclics Inc; Seattle Genetics; and Takeda Oncology.

Vemurafenib + Rituximab in R/R HCL (HCL-PG 03): Background § BRAF V 600 E mutation occurs in ~ 80% to 90% of HCL pts, leading to constitutive BRAF activation and promotion of cell survival and proliferation[1] § Up to 40% of HCL pts will relapse or become refractory to So. C first-line purine analogues (ie, cladribine or pentostatin)[1, 2] § Vemurafenib: oral BRAF inhibitor[3] – Phase II study of vemurafenib reported ORR of 96% (CR: 35%; PR: 61%) in pts with R/R HCL and BRAF V 600 E mutation; however, residual disease present even in CR[4] § Rituximab: CD 20 -directed cytolytic antibody[5] – Potential to eradicate BRAF inhibitor–resistant HCL cells[4] § Current study assessed efficacy and safety of chemotherapy-free combination of vemurafenib + rituximab in pts with R/R HCL[6] References in slidenotes Slide credit: clinicaloptions. com
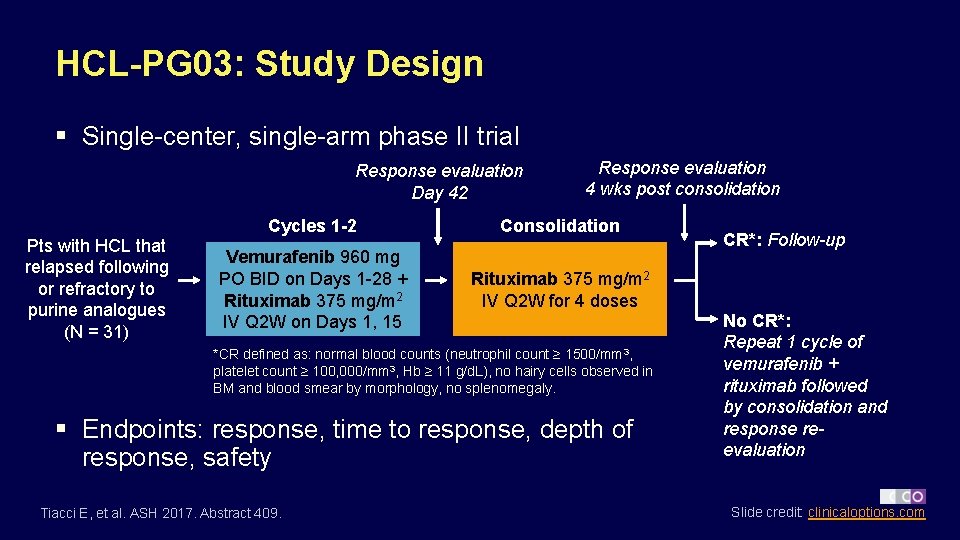
HCL-PG 03: Study Design § Single-center, single-arm phase II trial Response evaluation Day 42 Pts with HCL that relapsed following or refractory to purine analogues (N = 31) Response evaluation 4 wks post consolidation Cycles 1 -2 Consolidation Vemurafenib 960 mg PO BID on Days 1 -28 + Rituximab 375 mg/m 2 IV Q 2 W on Days 1, 15 Rituximab 375 mg/m 2 IV Q 2 W for 4 doses *CR defined as: normal blood counts (neutrophil count ≥ 1500/mm 3, platelet count ≥ 100, 000/mm 3, Hb ≥ 11 g/d. L), no hairy cells observed in BM and blood smear by morphology, no splenomegaly. § Endpoints: response, time to response, depth of response, safety Tiacci E, et al. ASH 2017. Abstract 409. CR*: Follow-up No CR*: Repeat 1 cycle of vemurafenib + rituximab followed by consolidation and response reevaluation Slide credit: clinicaloptions. com

HCL-PG 03: Baseline Pt Characteristics Characteristic Median age, yrs (range) Median prior therapies, n (range) Primary refractory to purine analogue, n (%) Tiacci E, et al. ASH 2017. Abstract 409. Pts (N = 31) 59 (35 -81) 3 (1 -14) 8 (26) Slide credit: clinicaloptions. com
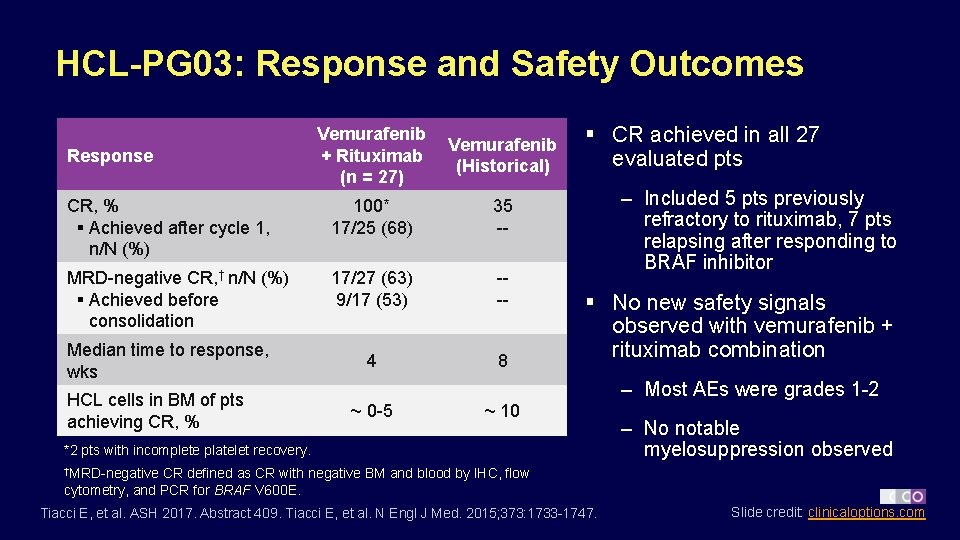
HCL-PG 03: Response and Safety Outcomes Vemurafenib + Rituximab (n = 27) Vemurafenib (Historical) CR, % § Achieved after cycle 1, n/N (%) 100* 17/25 (68) 35 -- MRD-negative CR, † n/N (%) § Achieved before consolidation 17/27 (63) 9/17 (53) --- 4 8 Response Median time to response, wks HCL cells in BM of pts achieving CR, % § CR achieved in all 27 evaluated pts – Included 5 pts previously refractory to rituximab, 7 pts relapsing after responding to BRAF inhibitor § No new safety signals observed with vemurafenib + rituximab combination – Most AEs were grades 1 -2 ~ 0 -5 ~ 10 *2 pts with incomplete platelet recovery. – No notable myelosuppression observed †MRD-negative CR defined as CR with negative BM and blood by IHC, flow cytometry, and PCR for BRAF V 600 E. Tiacci E, et al. ASH 2017. Abstract 409. Tiacci E, et al. N Engl J Med. 2015; 373: 1733 -1747. Slide credit: clinicaloptions. com

HCL-PG 03: Conclusions § Vemurafenib + rituximab associated with 100% CR rate in pts with R/R HCL – Effective in pts previously refractory to rituximab or who relapsed after treatment with BRAF inhibitors – CR rate and median time to response markedly improved from historical experience with vemurafenib monotherapy § AE profile consistent with previous experience with these agents – No significant myelotoxicity § Investigators conclude vemurafenib + rituximab is a safe, highly active chemotherapy-free option with a fixed, short duration for R/R HCL – Suggest evaluation in the frontline setting Tiacci E, et al. ASH 2017. Abstract 409. Slide credit: clinicaloptions. com

Go Online for More CCO Coverage of ASH 2017! Short slideset summaries of all the key data Additional CME-certified analyses with expert faculty commentary on all the key studies in: § § § Leukemias Lymphomas/CLL Myeloma Nonmalignant hematology Hot topics in hematology clinicaloptions. com/oncology
- Slides: 7