HAZARDS IDENTIFICATION KHADIJAH HANIM ABDUL RAHMAN DEPARTMENT OF

HAZARDS IDENTIFICATION KHADIJAH HANIM ABDUL RAHMAN DEPARTMENT OF CHEMICAL ENGINEERING TECHNOLOGY, FETECH, UNIMAP

A SCENARIO. . You and your family are on a road trip by a car in the middle of the night. You were replying a text message while driving at 100 km/h and it was raining heavily. The car hits a deep hole and one of your tire blows. You hit the brake, but due to slippery road and your car tire thread was thin, the car skidded and was thrown off the road.

POINTS TO PONDER What was the cause of the accident? What was the consequence of the event? What can we do to prevent all those things to happen in the first place? What other possible accidents might happen on the road trip? Can we be prepared before the accident occurs?
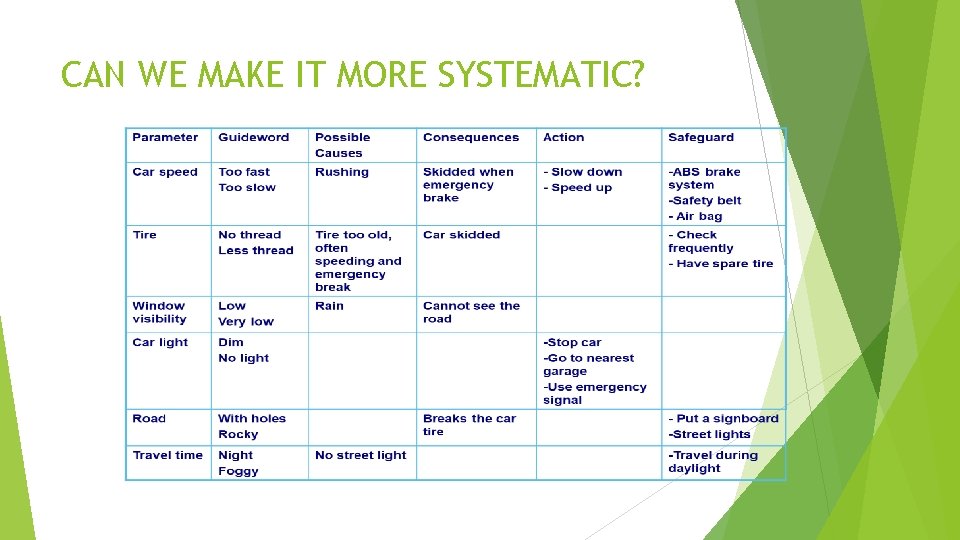
CAN WE MAKE IT MORE SYSTEMATIC?

HAZARD Hazards are everywhere. It is essential to identify hazards and reduce the risk well in advance of accident. For each process in a chemical plant the following questions must be asked: - WHAT ARE THE HAZARDS? - WHAT CAN GO WRONG AND HOW? - WHAT ARE THE CHANCES? - WHAT ARE THE CONSEQUENCES?
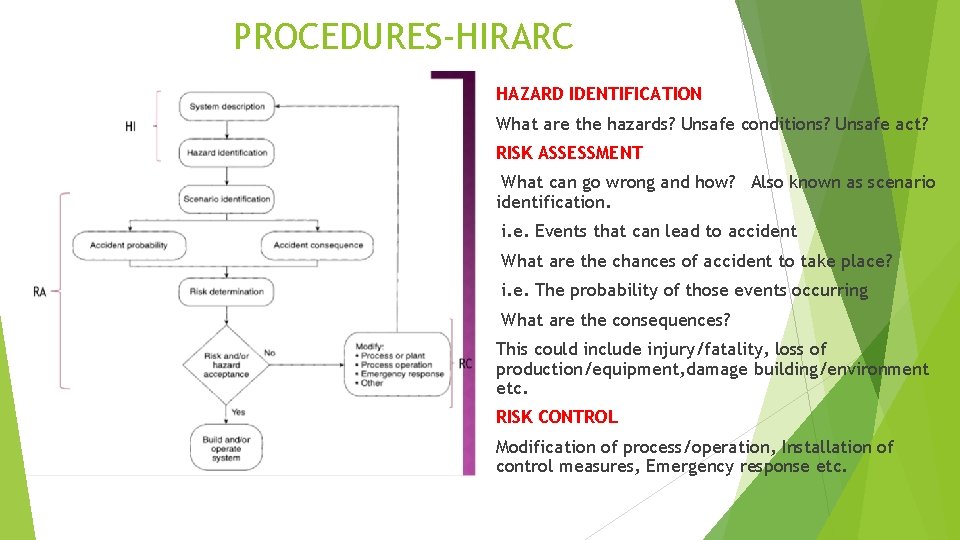
PROCEDURES-HIRARC HAZARD IDENTIFICATION What are the hazards? Unsafe conditions? Unsafe act? RISK ASSESSMENT What can go wrong and how? Also known as scenario identification. i. e. Events that can lead to accident What are the chances of accident to take place? i. e. The probability of those events occurring What are the consequences? This could include injury/fatality, loss of production/equipment, damage building/environment etc. RISK CONTROL Modification of process/operation, Installation of control measures, Emergency response etc.

HAZARD IDENTIFICATION Observing unsafe condition and unsafe acts that could potentially cause accidents. Conducting walk-through surveys, audits, checklist and inspections. Job Safety Analysis/Review. Examining documents and data

WHEN TO PERFORM HAZARD IDENTIFICATION & RISK ASSESSMENT? 1. At the initial design stage. ASAP to enable any modifications to be easily incorporated into the final design. 2. During ongoing operation. Note: *Best result is obtained if hazard identification is performed together with risk assessment. This could also prevented “gold-plated” i. e. implementing unnecessary /expensive safety equipment and procedures.

METHODS OF HAZARD IDENTIFICATION 1. Process Hazards Checklists 2. Hazard Surveys a. Inventory of hazardous material b. Dow indexes Dow Fire and Explosion Index Dow Chemical Exposure Index 3. HAZard and OPerability studies (HAZOP) 4. Safety Review

PROCESS HAZARDS CHECKLISTS A list of possible problems and areas to be checked. Reminder to reviewer or operator for potential problem areas. It can be used during design of process to identify design hazards or before process operation. A typical checklist covers: • • General layout Buildings Process Piping Equipment Instrument and Electrical Safety Equipment Raw Materials





HAZARDS SURVEYS Hazards survey can be: - simple inventory of hazardous materials in a facility, or - complex like rigorous procedure such as Dow Fire & Explosion Index (F&EI) and Dow-Chemical Exposure Index (CEI) – most popular forms of survey. - The Dow indexes are a formal rating system, much like an income tax form, that provide penalties for hazards and credits for safety equipment and procedures. Dow Fire and Explosion Index A ranking system that gives a relative index to the risk of individual process unit due to potential fires and explosions Dow Chemical Exposure Index Simple method of rating the relative acute health hazard potential for people in neighboring plants or communities arising from possible chemical release incidents



HAZARDS AND OPERABILITY STUDIES (HAZOP) HAZOP study- formal procedure to identify hazards in chemical process facility. It is effective in identifying hazards and well accepted by the industry. Basic idea - consider all the possible ways that process and operational failures can occur. Detailed information on the process must be available before HAZOP study which are: v process flow diagrams (PFDs) v process and instrumentation diagrams (P&IDs) v detailed equipment specifications v materials of construction v mass and energy balances

HAZOP PROCEDURES 1. Begin with a detailed flow sheet (PFD and P&ID). Break the flow sheet into a number of process units ( reactor system, boiler house, storage area). Thus the reactor area might be one unit, and the storage tank another. Select a unit for study. 2. Choose a study node (location of study)- (vessel, cooling coil, pipe line, tank operating instruction). 3. Describe the design intent of the study node (how it is expected to operate). For example, vessel V-1 is designed to store the benzene feedstock and provide it on demand to the reactor. 4. Pick a process parameter: flow, level, temperature, pressure, concentration, p. H, viscosity, state (solid, liquid, or gas), agitation, volume, reaction, sample, component, start, stop, stability, power, inert

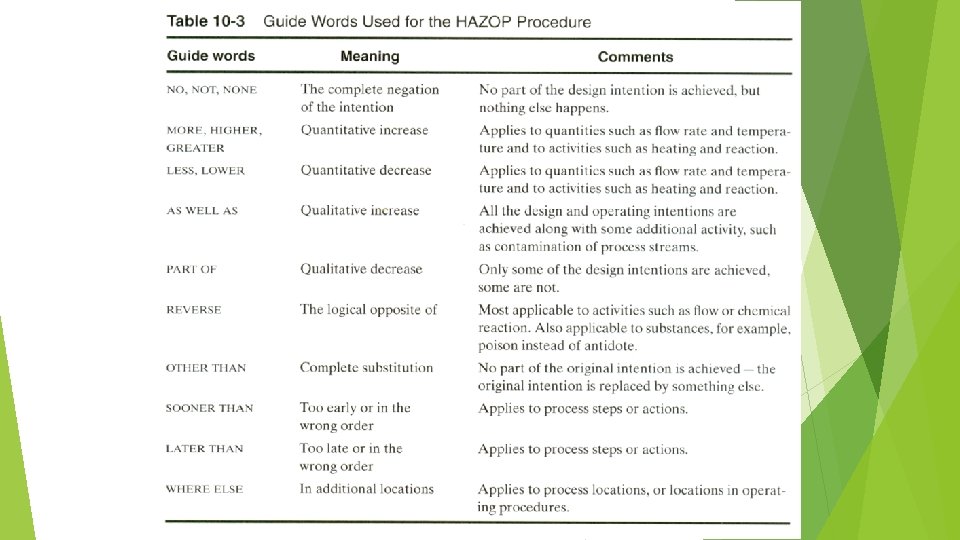
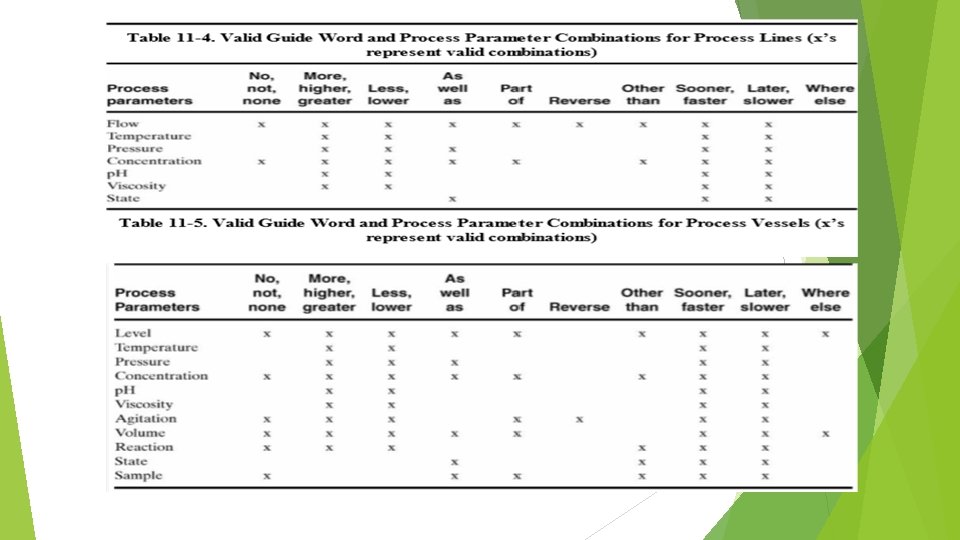
HAZOP PROCEDURES …CONTINUE 5. Apply a guide word to the process parameter to suggest possible deviations (a way that the process conditions may depart from their design/process intent). A list of guide words is shown in Table 10 -3. Some of the guide word process parameter combinations are meaningless, as shown in Tables 10 -4 and 10 -5 for process lines and vessels. 6. If the deviation is applicable, determine possible causes and note any protective systems. 7. Evaluate the consequences of the deviation (if any). 8. Recommend action (what? by whom? by when? ) 9. Record all information.

HAZOP PROCEDURES …CONTINUE 10. Repeat steps 5 through 9 until all applicable guide words have been applied to the chosen process parameter 11. Repeat steps 4 through 10 until all applicable process parameters have been considered for the given study node. 12. Repeat steps 2 through 11 until all study nodes have been considered for the given section and proceed to the next section on the flow sheet



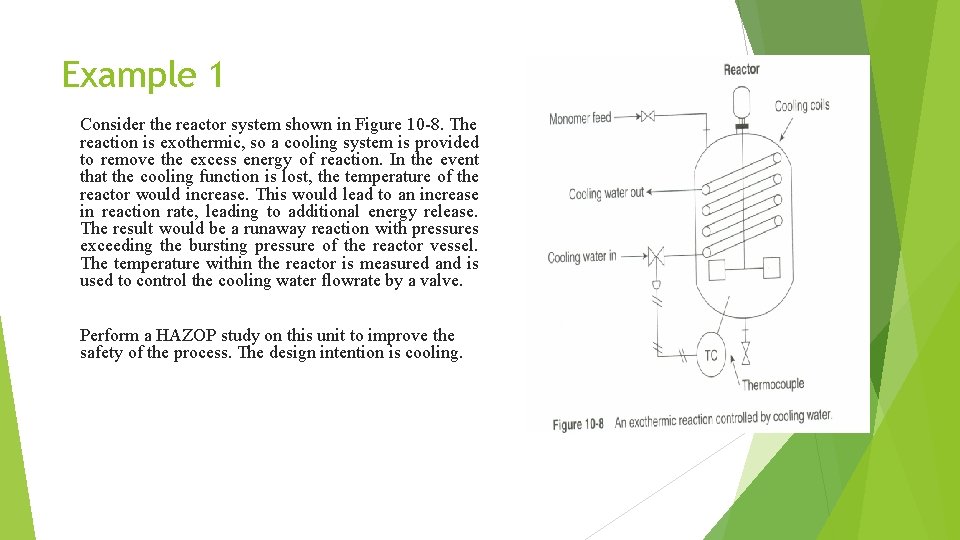
Example 1 Consider the reactor system shown in Figure 10 -8. The reaction is exothermic, so a cooling system is provided to remove the excess energy of reaction. In the event that the cooling function is lost, the temperature of the reactor would increase. This would lead to an increase in reaction rate, leading to additional energy release. The result would be a runaway reaction with pressures exceeding the bursting pressure of the reactor vessel. The temperature within the reactor is measured and is used to control the cooling water flowrate by a valve. Perform a HAZOP study on this unit to improve the safety of the process. The design intention is cooling.


The potential process modification resulting from this HAZOP study. . Install a high-temperature alarm in the event of cooling water loss. Install a high-temperature shutdown system, the shut-down temperature would be higher than the alarm temperature to alert the operator with the opportunity to restore the cooling. auto shut-down in the event of high reactor temperature. Install a check valve in the cooling line to prevent reverse flow. Periodically inspect the cooling coil to ensure its integrity. Study the cooling water source to consider possible contamination and interruption of supply. Install cooling water flow meter and low-flow alarm (to provide immediate indication of cooling loses). All coolant water failures be properly reported and proper action taken if occurred repeatedly. Monomer feed valve must be fail closed on power failure
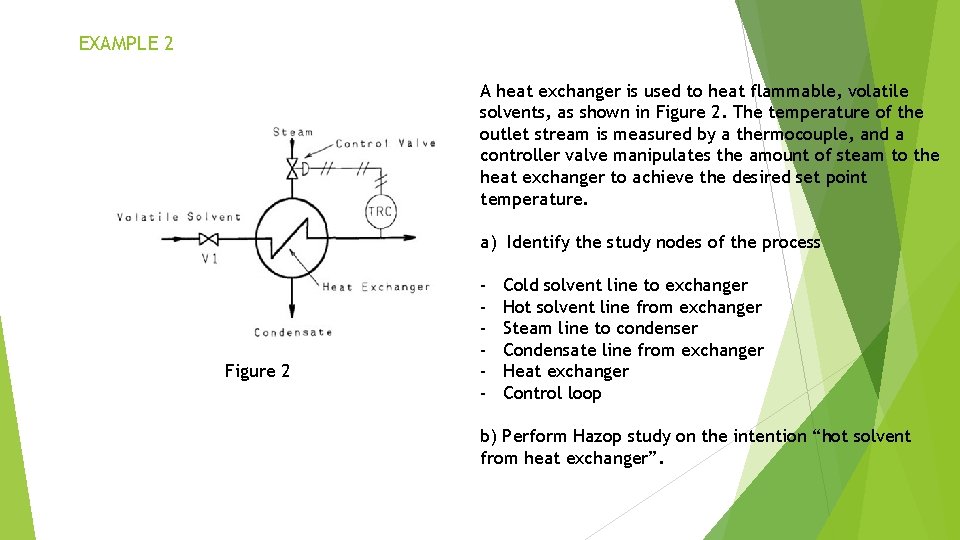
EXAMPLE 2 A heat exchanger is used to heat flammable, volatile solvents, as shown in Figure 2. The temperature of the outlet stream is measured by a thermocouple, and a controller valve manipulates the amount of steam to the heat exchanger to achieve the desired set point temperature. a) Identify the study nodes of the process Figure 2 - Cold solvent line to exchanger Hot solvent line from exchanger Steam line to condenser Condensate line from exchanger Heat exchanger Control loop b) Perform Hazop study on the intention “hot solvent from heat exchanger”.


SAFETY REVIEWS Another method that is commonly used to identify safety problems in laboratory and process areas and to develop solutions is the safety review. Purposes of Safety Reviews v Finding initiating events that can cause accidents. v Develop recommendations including new, modified and improved equipment, control and procedures. v Review the previous accident/incident in similar plant/process to prevent repeating the past mistakes. Review should be conducted periodically during the entire life of a project. There are two types of safety reviews: -informal safety review -formal safety review



FORMAL SAFETY REVIEW REPORT 1. Introduction Executive summary Process overview/description Reaction & Stoichiometry Engineering data -operating temperatures, pressures, and relevant physical property data for the materials used. 2. Raw materials and products Hazard and handling problems- related to raw materials and products- how to minimize these hazards. 3. Equipment setup Equipment description/configuration Equipment specifications 4. Procedures Normal operating procedures Safety procedure Emergency shutdown Fail-safe procedures Major release procedures Waste disposal procedure Cleanup procedures 5. Startup Safety Checklist 6. MSDS for each hazardous material

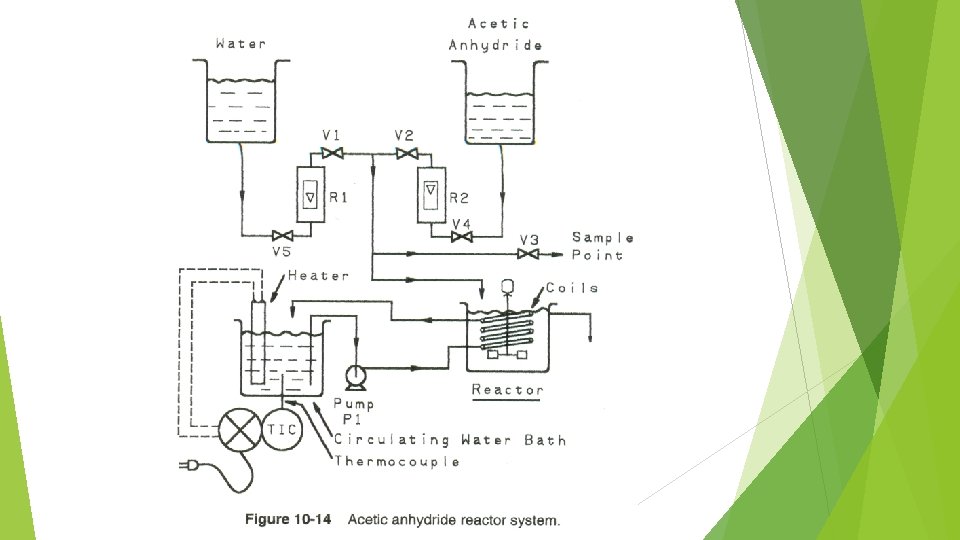
ASSIGNMENT- In groups (3 to 4 members) Submission deadline: 11 October 2018 The hydrolysis of acetic anhydride is being studied in a laboratory-scale continuously stirred tank reactor (CSTR). In this reaction acetic anhydride [(CH 3 CO)2 O reacts with water to produce acetic acid (CH 3 COOH). The concentration of acetic anhydride at any time in the CSTR is determined by titration with sodium hydroxide. Because the titration procedure requires time (relative to the hydrolysis reaction time), it is necessary to quench the hydrolysis reaction as soon as the sample is taken. The quenching is achieved by adding an excess of aniline to the sample. The quench reaction is The quenching reaction also forms acetic acid, but in a different stoichiometric ratio than the hydrolysis reaction. Thus it is possible to determine the acetic anhydride concentration at the time the sample was taken.

The initial experimental design is shown in Figure 10 -14. Water and acetic anhydride are gravity-fed from reservoirs and through a set of rotameters. The water is mixed with the acetic anhydride just before it enters the reactor. Water is also circulated by a centrifugal pump from the temperature bath through coils in the reactor vessel. This maintains the reactor temperature at a fixed value. A temperature controller in the water bath maintains the temperature to within 1°F of the desired temperature. a. Develop a safety checklist for use before operation of this experiment. b. Perform a HAZOP study on the intention “reactant flow to reactor” (process paremeter: flow) for your analysis. c. Suggest specific recommendations to improve the safety of this experiment?
- Slides: 38