HARVONI sofosbuvir 400 mgledipasvir 100 mg QD Premio

® HARVONI sofosbuvir 400 mg/ledipasvir 100 mg QD Premio Galeno Circolo della Stampa, Corso Venezia 48 Milano 25 novembre 2015 Paolo Fedeli MD 1
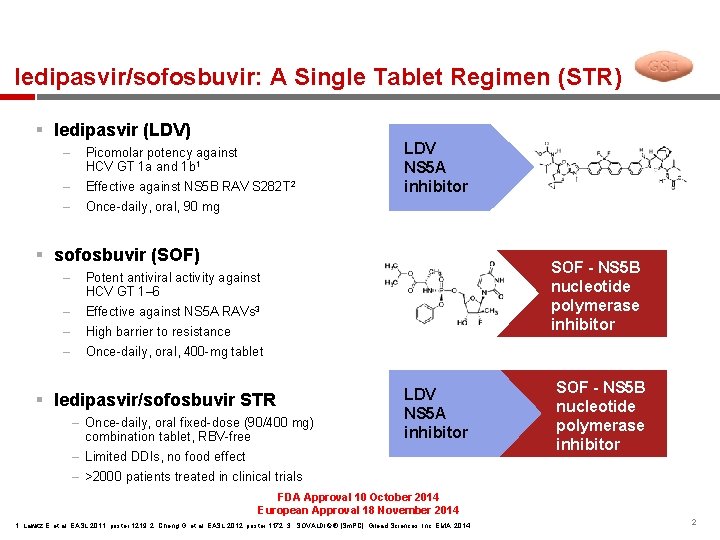
ledipasvir/sofosbuvir: A Single Tablet Regimen (STR) § ledipasvir (LDV) – Picomolar potency against HCV GT 1 a and 1 b 1 – Effective against NS 5 B RAV S 282 T 2 – Once-daily, oral, 90 mg LDV NS 5 A inhibitor § sofosbuvir (SOF) ‒ Potent antiviral activity against HCV GT 1– 6 ‒ Effective against NS 5 A RAVs 3 ‒ High barrier to resistance ‒ Once-daily, oral, 400 -mg tablet § ledipasvir/sofosbuvir STR – Once-daily, oral fixed-dose (90/400 mg) combination tablet, RBV-free SOF - NS 5 B nucleotide polymerase inhibitor LDV NS 5 A inhibitor – Limited DDIs, no food effect SOF - NS 5 B nucleotide polymerase inhibitor – >2000 patients treated in clinical trials FDA Approval 10 October 2014 European Approval 18 November 2014 1. Lawitz E, et al. EASL 2011, poster 1219; 2. Cheng G, et al. EASL 2012, poster 1172; 3. SOVALDI ® [Sm. PC]. Gilead Sciences, Inc. EMA, 2014 2

HARVONI® § First FDC, STR regimen to treat Gt 1 HCV chronic infection § Efficacy: high rates of SVR, confirmed in real world data § Tolerability: good safety profile, <1% D/C rate for SAEs § Opportunity to shorten treatment schedule § Used in several subpopulation: confirm results with high efficacy and good safety profile across all population studied § Efficacy and Effectiveness confirmed in all stages of hepatic disease (from F 0 up to decompensated cirrhosis) 3
Ledipasvir/Sofosbuvir as the First HCV Single Tablet Regimen (STR) 2011 -2013 2014 4
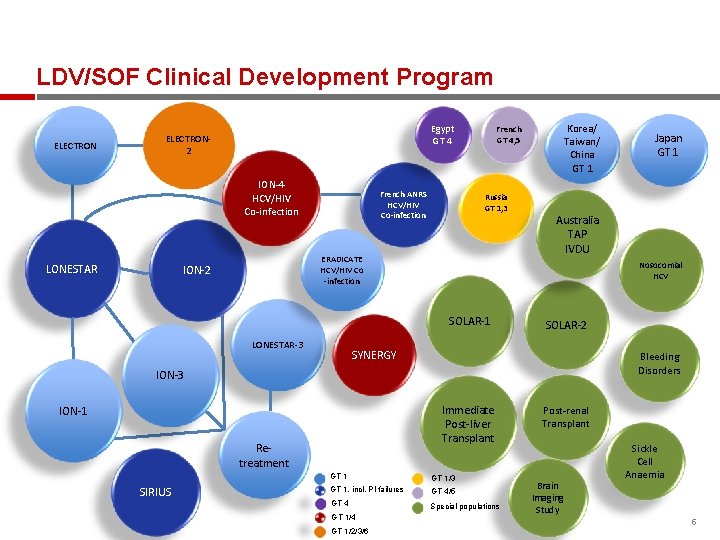
LDV/SOF Clinical Development Program ELECTRON Egypt GT 4 ELECTRON 2 ION-4 HCV/HIV Co-infection LONESTAR French ANRS HCV/HIV Co-infection Russia GT 1, 3 ERADICATE HCV/HIV Co -infection ION-2 Australia TAP IVDU SOLAR-2 SYNERGY Bleeding Disorders ION-3 Immediate Post-liver Transplant ION-1 Retreatment SIRIUS GT 1/3 GT 1, incl. PI failures GT 4/5 GT 4 Special populations GT 1/4 GT 1/2/3/6 Japan GT 1 Nosocomial HCV SOLAR-1 LONESTAR-3 Korea/ Taiwan/ China GT 1 French GT 4, 5 Post-renal Transplant Brain Imaging Study Sickle Cell Anaemia 5
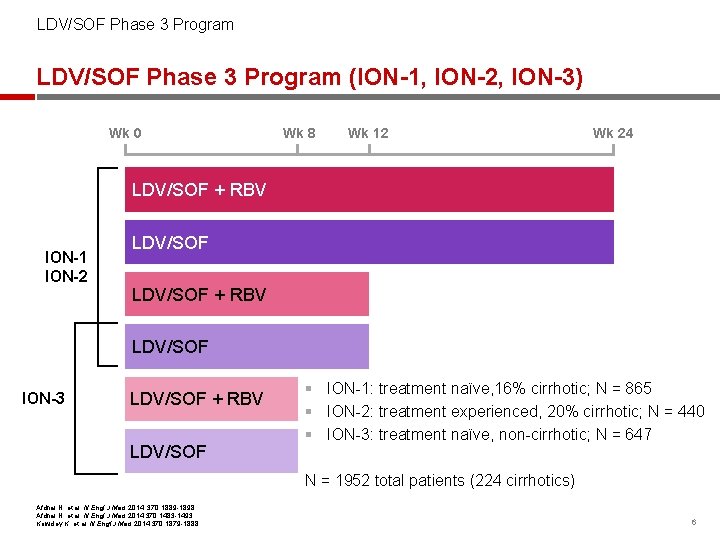
LDV/SOF Phase 3 Program (ION-1, ION-2, ION-3) Wk 0 Wk 8 Wk 12 Wk 24 LDV/SOF + RBV ION-1 ION-2 LDV/SOF + RBV LDV/SOF ION-3 LDV/SOF + RBV LDV/SOF § ION-1: treatment naïve, 16% cirrhotic; N = 865 § ION-2: treatment experienced, 20% cirrhotic; N = 440 § ION-3: treatment naïve, non-cirrhotic; N = 647 N = 1952 total patients (224 cirrhotics) Afdhal N, et al. N Engl J Med 2014; 370: 1889 -1898; Afdhal N, et al. N Engl J Med 2014; 370: 1483 -1493; Kowdley K, et al. N Engl J Med 2014; 370: 1879 -1888 6
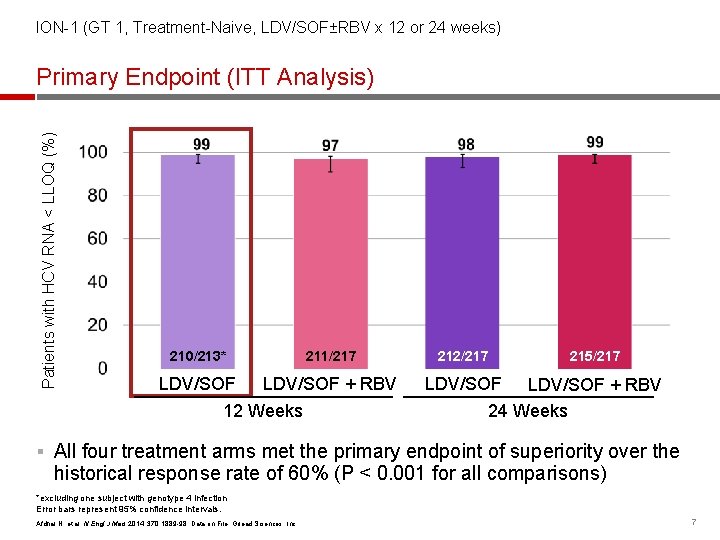
ION-1 (GT 1, Treatment-Naive, LDV/SOF±RBV x 12 or 24 weeks) Patients with HCV RNA < LLOQ (%) Primary Endpoint (ITT Analysis) 210/213* 211/217 LDV/SOF + RBV 12 Weeks 212/217 215/217 LDV/SOF + RBV 24 Weeks § All four treatment arms met the primary endpoint of superiority over the historical response rate of 60% (P < 0. 001 for all comparisons) *excluding one subject with genotype 4 infection Error bars represent 95% confidence intervals. Afdhal N, et al. N Engl J Med 2014; 370: 1889 -98. Data on File, Gilead Sciences, Inc. 7
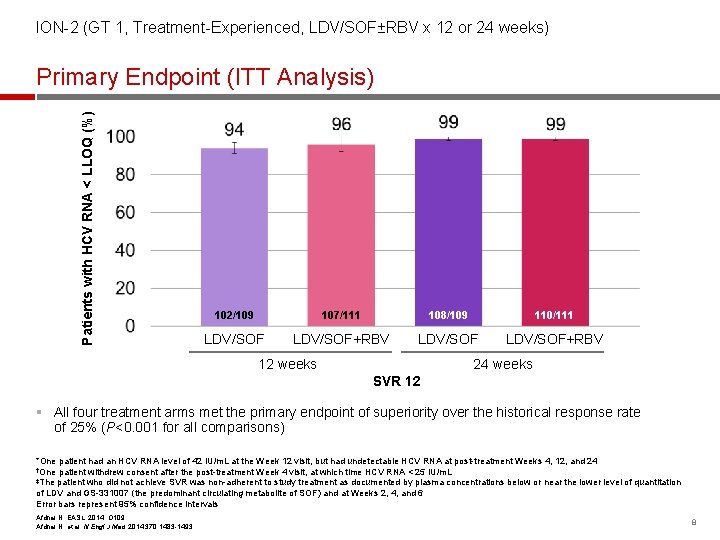
ION-2 (GT 1, Treatment-Experienced, LDV/SOF±RBV x 12 or 24 weeks) Patients with HCV RNA < LLOQ (%) Primary Endpoint (ITT Analysis) 102/109 107/111 108/109 110/111 LDV/SOF+RBV 12 weeks 24 weeks SVR 12 § All four treatment arms met the primary endpoint of superiority over the historical response rate of 25% (P<0. 001 for all comparisons) *One patient had an HCV RNA level of 42 IU/m. L at the Week 12 visit, but had undetectable HCV RNA at post-treatment Weeks 4, 12, and 24 †One patient withdrew consent after the post-treatment Week 4 visit, at which time HCV RNA < 25 IU/m. L ‡The patient who did not achieve SVR was non-adherent to study treatment as documented by plasma concentrations below or near the lower level of quantitation of LDV and GS-331007 (the predominant circulating metabolite of SOF) and at Weeks 2, 4, and 6 Error bars represent 95% confidence intervals Afdhal N, EASL, 2014, O 109 Afdhal N, et al. N Engl J Med 2014; 370: 1483 -1493 8
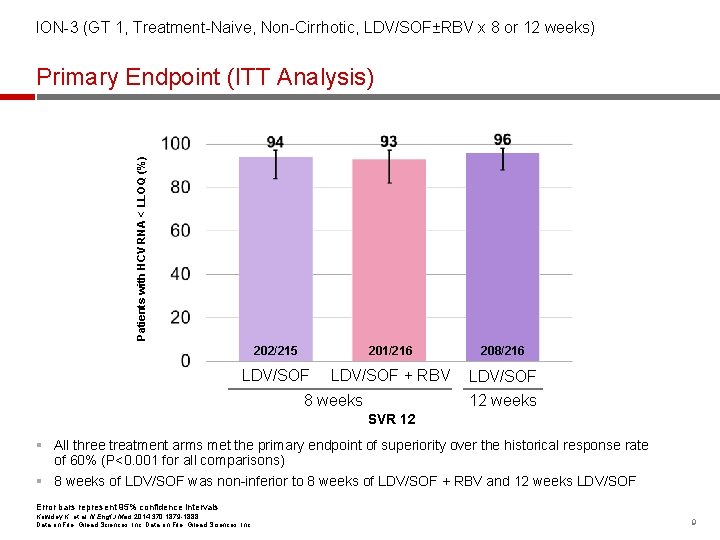
ION-3 (GT 1, Treatment-Naive, Non-Cirrhotic, LDV/SOF±RBV x 8 or 12 weeks) Patients with HCV RNA < LLOQ (%) Primary Endpoint (ITT Analysis) 202/215 201/216 208/216 LDV/SOF + RBV LDV/SOF 8 weeks 12 weeks SVR 12 § All three treatment arms met the primary endpoint of superiority over the historical response rate of 60% (P<0. 001 for all comparisons) § 8 weeks of LDV/SOF was non-inferior to 8 weeks of LDV/SOF + RBV and 12 weeks LDV/SOF Error bars represent 95% confidence intervals Kowdley K, et al. N Engl J Med 2014; 370: 1879 -1888 Data on File, Gilead Sciences, Inc. 9
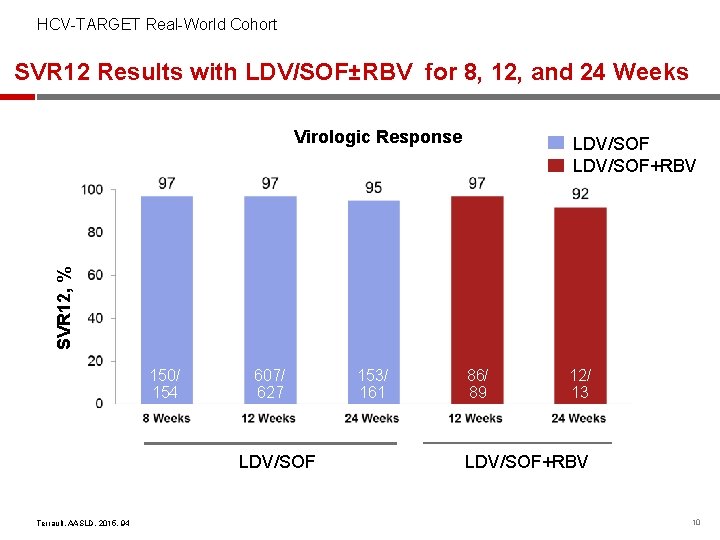
HCV-TARGET Real-World Cohort SVR 12 Results with LDV/SOF±RBV for 8, 12, and 24 Weeks Virologic Response SVR 12, % LDV/SOF+RBV 150/ 154 607/ 627 LDV/SOF Terrault, AASLD, 2015, 94 153/ 161 86/ 89 12/ 13 LDV/SOF+RBV 10
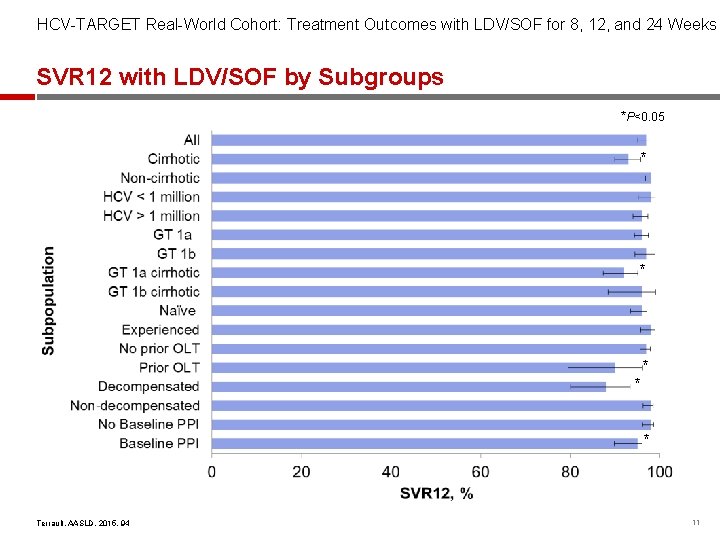
HCV-TARGET Real-World Cohort: Treatment Outcomes with LDV/SOF for 8, 12, and 24 Weeks SVR 12 with LDV/SOF by Subgroups *P<0. 05 * * * Terrault, AASLD, 2015, 94 11
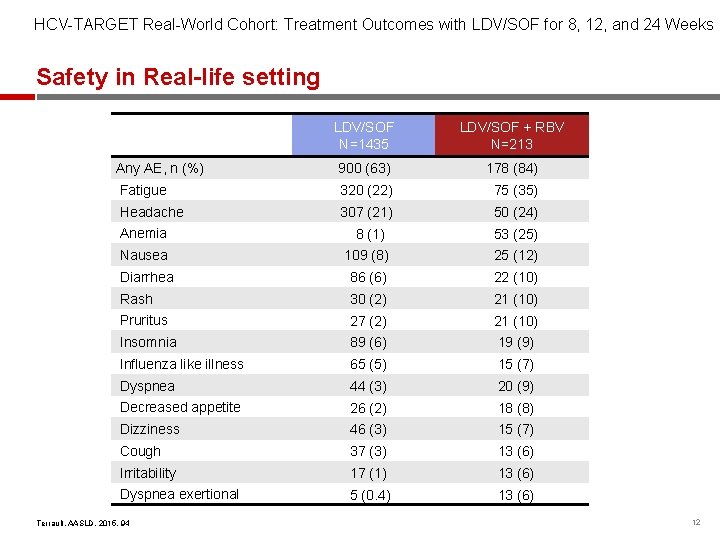
HCV-TARGET Real-World Cohort: Treatment Outcomes with LDV/SOF for 8, 12, and 24 Weeks Safety in Real-life setting LDV/SOF N=1435 LDV/SOF + RBV N=213 Any AE, n (%) 900 (63) 178 (84) Fatigue 320 (22) 75 (35) Headache 307 (21) 50 (24) Anemia 8 (1) 53 (25) Nausea 109 (8) 25 (12) Diarrhea 86 (6) 22 (10) Rash 30 (2) 21 (10) Pruritus 27 (2) 21 (10) Insomnia 89 (6) 19 (9) Influenza like illness 65 (5) 15 (7) Dyspnea 44 (3) 20 (9) Decreased appetite 26 (2) 18 (8) Dizziness 46 (3) 15 (7) Cough 37 (3) 13 (6) Irritability 17 (1) 13 (6) Dyspnea exertional 5 (0. 4) 13 (6) Terrault, AASLD, 2015, 94 12
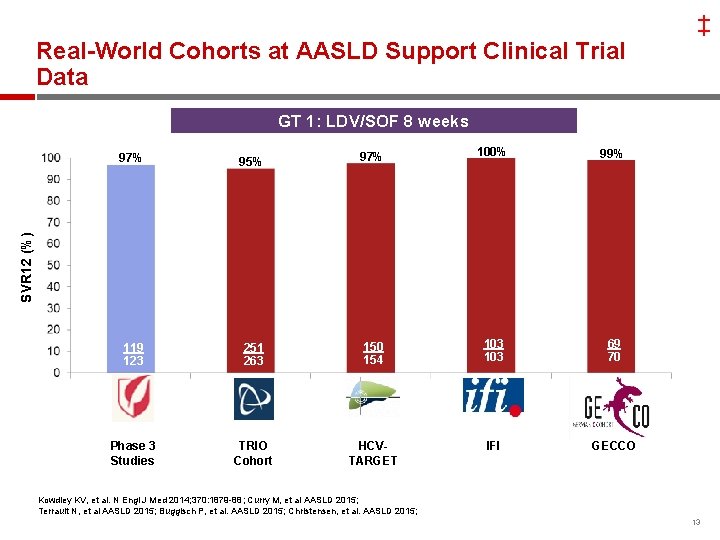
‡ Real-World Cohorts at AASLD Support Clinical Trial Data GT 1: LDV/SOF 8 weeks 95% 97% 119 123 251 263 150 154 Phase 3 Studies TRIO Cohort 100% 99% 103 69 70 IFI GECCO SVR 12 (%) 97% 43 44 HCV 172 TARGET 202 Kowdley KV, et al. N Engl J Med 2014; 370: 1879 -88; Curry M, et al AASLD 2015; Terrault N, et al AASLD 2015; Buggisch P, et al. AASLD 2015; Christensen, et al. AASLD 2015; 13

HARVONI® § First FDC, STR regimen to treat Gt 1 HCV chronic infection § Efficacy: high rates of SVR, confirmed in real world data § Tolerability: good safety profile, <1% D/C rate for SAEs § Opportunity to shorten treatment schedule § Used in several subpopulation: confirm results with high efficacy and good safety profile across all population studied § Efficacy and Effectiveness confirmed in all stages of hepatic disease (from F 0 up to decompensated cirrhosis) 14
- Slides: 14