Harris Chapter 7 Atomic Structure 7 1 Orbital

Harris Chapter 7 - Atomic Structure • 7. 1 – Orbital Magnetic Moments, discovery of intrinsic spin • 7. 2 & 7. 3 – Identical Particles (warning: examples in book all inf-squ well) – Exclusion Principle • 7. 4 & 7. 5 – Multielectron Atoms, effective charges – Hartree Treatment • 7. 6 – Spin-Orbit Effect • 7. 7 – Adding QM Angular Momenta • 7. 9 & 7. 8 – Multielectron Spectroscopic Notation – Zeeman Effect

Summary So Far http: //asd-www. larc. nasa. gov/cgi-bin/SCOOL_Clouds/Cumulus/list. cgi

7. 1 Orbital Magnetic Moments and Discovery of Intrinsic Spin

Two kinds of Angular Momentum • Classical Angular Momentum – – L=rxp r vector, p vector L vector L obeys vector math Any L possible, no contraints on Lx Ly Lz • Quantum – – – Quantum Mechanical Angular Momentum L=rxp r vector, p vector operator L 3 component operator L obeys …… got to be careful L described by two labels l , m L and Lz can be known, Lx and Ly cannot
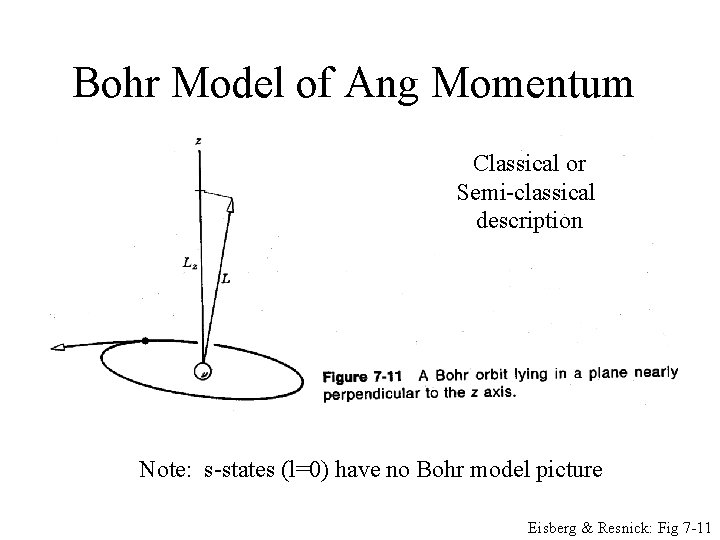
Bohr Model of Ang Momentum Classical or Semi-classical description Note: s-states (l=0) have no Bohr model picture Eisberg & Resnick: Fig 7 -11
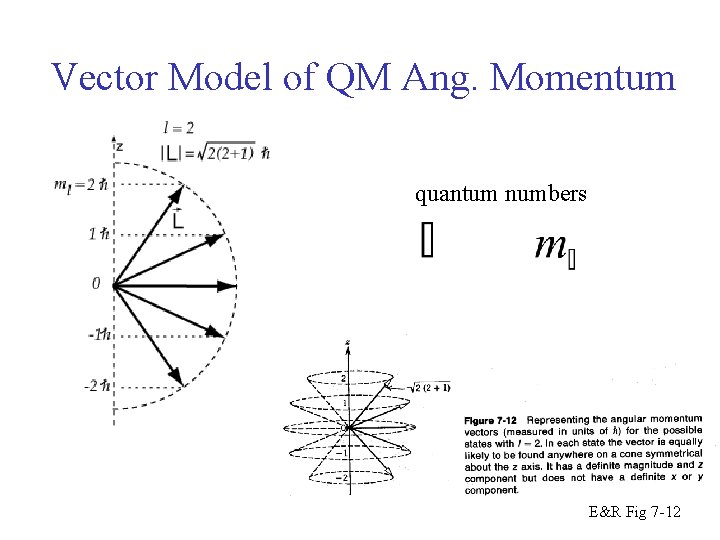
Vector Model of QM Ang. Momentum quantum numbers E&R Fig 7 -12

Edmonds “A. M. in QM” pg 19: “We might imagine the vector moving in an unobservable way about the z-axis. . . ” pg 29: “The QM probability density, not being time dependent, gives us no information about the motion of the particle in it’s orbit. ” Y*(r, t) Y(r, t)=Y(r) e-iwt

Morrison, Estle, Lane “Understanding More QM”, Prentice-Hall, 1991

Otto Stern & Walther Gerlach ~1922 Bohr’s Q hypothesis Sommerfeld’s Q hypothesis 3 2 1 Assigned by advisor Max Born to demonstrate existence of the l, ml quantum numbers
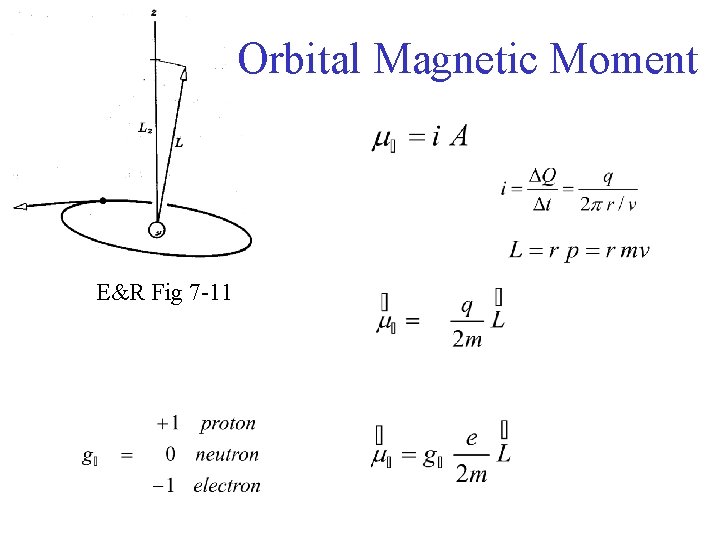
Orbital Magnetic Moment E&R Fig 7 -11

E&R Fig 7 -11 Orbital Magnetic Moment
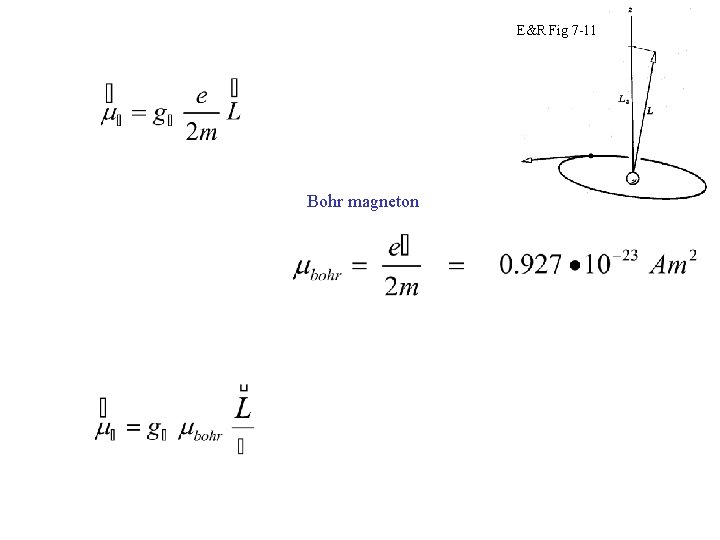
E&R Fig 7 -11 Bohr magneton

B m

B m Different ml states experience different forces

Use B as z-axis. Different ml states experience different forces

Stern & Gerlach ~1922 Harris Fig 7. 3, 7. 4

Stern & Gerlach ~1922 Intended to demonstrate space quantization (l), & therefore expected odd number of spots, but observed an even number. http: //upload. wikimedia. org/wikipedia/en/2/29/Stern-Gerlach_experiment. PNG

Despite Stern's careful design and feasibility calculations, the experiment took more than a year to accomplish. In the final form of the apparatus, a beam of silver atoms (produced by effusion of metallic vapor from an oven heated to 1000°C) was collimated by two narrow slits (0. 03 mm wide) and traversed a deflecting magnet 3. 5 cm long with field strength about 0. 1 tesla and gradient 10 tesla/cm. The splitting of the silver beam achieved was only 0. 2 mm. Accordingly, misalignments of collimating slits or the magnet by more than 0. 01 mm were enough to spoil an experimental run. The attainable operating time was usually only a few hours between breakdowns of the apparatus. Thus, only a meager film of silver atoms, too thin to be visible to an unaided eye, was deposited on the collector plate. Stern described an early episode: http: //www. physicstoday. org/pt/vol-56/iss-12/p 53. html

Stern described an early episode: After venting to release the vacuum, Gerlach removed the detector flange. But he could see no trace of the silver atom beam and handed the flange to me. With Gerlach looking over my shoulder as I peered closely at the plate, we were surprised to see gradually emerge the trace of the beam. . Finally we realized what [had happened]. I was then the equivalent of an assistant professor. My salary was too low to afford good cigars, so I smoked bad cigars. These had a lot of sulfur in them, so my breath on the plate turned the silver into silver sulfide, which is jet black, so easily visible. It was like developing a photographic film. 7 http: //www. physicstoday. org/pt/vol-56/iss-12/p 53. html
Wolfgang Pauli ~ 1924 • Pauli Exclusion Principle • No two electrons can have the same quantum number • Postulated an additional quantum number (i. e. label) • Believed it came from the interaction between electrons.

Ralph Kronig ~1925 • Spinning Electron Idea

Goudsmit & Ulhenbeck ~ 1925 • Studied high resolution spectra of alkali elements
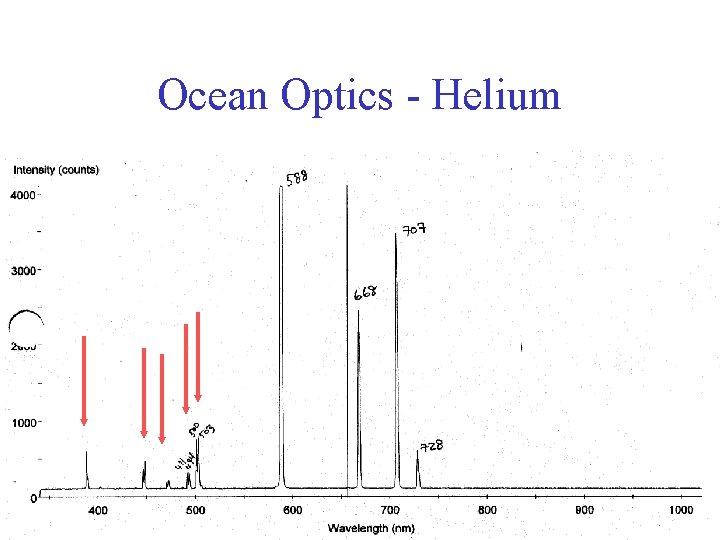
Ocean Optics - Helium
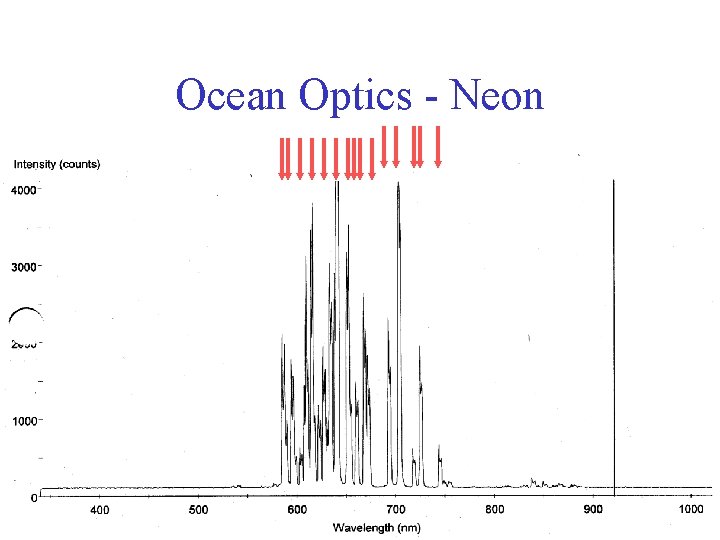
Ocean Optics - Neon

Giancoli – fig 36. 21
![The old and the new term scheme of hydrogen [5]. The scheme shows the The old and the new term scheme of hydrogen [5]. The scheme shows the](http://slidetodoc.com/presentation_image/27e01b1af759b22fc8f65e812f19e8f4/image-26.jpg)
The old and the new term scheme of hydrogen [5]. The scheme shows the multiplet splitting of the excited states of the hydrogen atom with principal quantum number n=3, presented by Goudsmit in the form in which it appeared in the original publications of 1926. The assignment in the current notation has been added at the right. With the development of quantum mechanics the notation changed. The quantum numbers L and J now usedfor the orbital and total angular momentum, respectively, correspond to K-1/2 and J-1/2 in the figure. The "forbidden component" referred to by Goudsmit is of the type 3 2 P 2 1/2 --> 2 S in which the total angular momentum is conserved and L changes by plus or minus 1. [5] S. Goudsmit and G. E. Uhlenbeck, Physica 6 (1926) 273.

Uhlenbeck & Goudsmit ~ 1925 The discovery note in Naturwissenschaften is dated 17 October 1925. One day earlier Ehrenfest had written to Lorentz to make an appointment and discuss a "very witty idea" of two of his graduate students. When Lorentz pointed out that the idea of a spinning electron would be incompatible with classical electrodynamics, Uhlenbeck asked Ehrenfest not to submit the paper. Ehrenfest replied that he had already sent off their note, and he added: "You are both young enough to be able to afford a stupidity!" http: //www. lorentz. leidenuniv. nl/history/spin. html

Uhlenbeck & Goudsmit ~ 1925 Ehrenfest's encouraging response to his students ideas contrasted sharply with that of Wolfgang Pauli. As it turned out, Ralph Kronig, a young Columbia University Ph. D who had spent two years studying in Europe, had come up with the idea of electron spin several months before Uhlenbeck and Goudsmit. He had put it before Pauli for his reactions, who had ridiculed it, saying that "it is indeed very clever but of course has nothing to do with reality". Kronig did not publish his ideas on spin. No wonder that Uhlenbeck would later refer to the "luck and privilege to be students of Paul Ehrenfest". http: //www. lorentz. leidenuniv. nl/history/spin. html

“This isn't right. This isn't even wrong. ” http: //www-groups. dcs. st-and. ac. uk/~history/Mathematicians/Pauli. html His ability to make experiments self destruct simply by being in the same room was legendary, and has been dubbed the "Pauli effect" (Frisch 1991, p. 48; Gamow 1985). There were some people thinking about electron spin in those days, but there was a lot of basic opposition to such an idea. One of the first was Ralph de Laer Kronig. He got the idea that the electron should have a spin in addition to its orbital motion. He was working with Wolfgang Pauli at the time, and he told his idea to Pauli said, "No, it's quite impossible. " Pauli completely crushed Kronig. Then the idea occurred quite independently to two Young Dutch physicists, George Uhlenbeck and Samuel Goudsmit. They were working in Leiden with Professor Paul Ehrenfest, and they wrote up a little paper about it and took it to Ehrenfest liked the idea very much. He suggested to Uhlenbeck and Goudsmit that they should go and talk it over with Hendrik Lorentz, who lived close by in Haarlem. "The Birth of Particle Physics, " edited by Laurie M. Brown and Lillian Hoddeson. The essay by Paul A. M. Dirac is entitled "Origin of Quantum Field Theory. "

“This isn't right. This isn't even wrong. ” http: //www-groups. dcs. st-and. ac. uk/~history/Mathematicians/Pauli. html They did go and talk it over with Lorentz said, "No, it's quite impossible for the electron to have a spin. I have thought of that myself, and if the electron did have a spin, the speed of the surface of the electron would be greater than the velocity of light. So, it's quite impossible. " Uhlenbeck and Goudsmit went back to Ehrenfest and said they would like to withdraw the paper that they had given to him. Ehrenfest said, "No, it's too late; I have already sent it in for publication " His ability to make experiments self destruct simply by being in the same room was legendary, and has been dubbed the "Pauli effect" (Frisch 1991, p. 48; Gamow 1985). "The Birth of Particle Physics, " edited by Laurie M. Brown and Lillian Hoddeson. The essay by Paul A. M. Dirac is entitled "Origin of Quantum Field Theory. "

The calculation (using current values) r < 2. 8 E-19 m b > 3 * 10 + 6 value from Bhabha scattering at CERN

“This isn't right. This isn't even wrong. ” http: //www-groups. dcs. st-and. ac. uk/~history/Mathematicians/Pauli. html That is how the idea of electron spin got publicized to the world. We really owe it to Ehrenfest's impetuosity and to his not allowing the younger people to be put off by the older ones. The idea of the electron having two states of spin provided a perfect answer to the duplexity. His ability to make experiments self destruct simply by being in the same room was legendary, and has been dubbed the "Pauli effect" (Frisch 1991, p. 48; Gamow 1985). "The Birth of Particle Physics, " edited by Laurie M. Brown and Lillian Hoddeson. The essay by Paul A. M. Dirac is entitled "Origin of Quantum Field Theory. "

Letter fm Thomas to Goudsmit Part of a letter by L. H. Thomas to Goudsmit (25 March 1926). Reproduced from a transparency shown by Goudsmit during his 1971 lecture. The original is presumably in the Goudsmit archive kept by the AIP Center for History of Physics. http: //www. lorentz. leidenuniv. nl/history/spin/goudsmit. html

intrinsic spin • Fundamental objects – electron – neutrino – photon spin – ½ , but LH only spin – 1 • Composite objects – proton – neutron – D delta spin – ½ spin – 3/2

How to Denote Wavefunctions (version 1) the spinor has no ‘functional form’ because spin is not a spatial feature
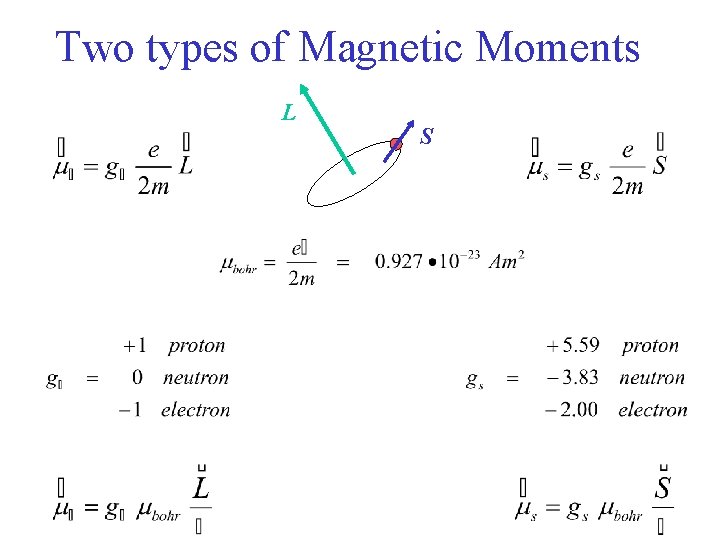
Two types of Magnetic Moments L S

interesting fundamental constants -2. 002 319 304 3622 (15) 1. 602 176 487 (40) x 10 -19 C

7. 2 & 7. 3 Complications from having Identical Particles
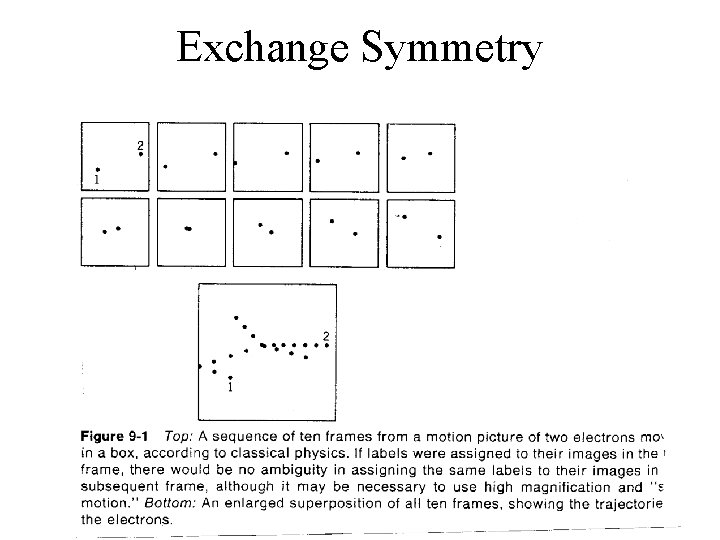
Exchange Symmetry

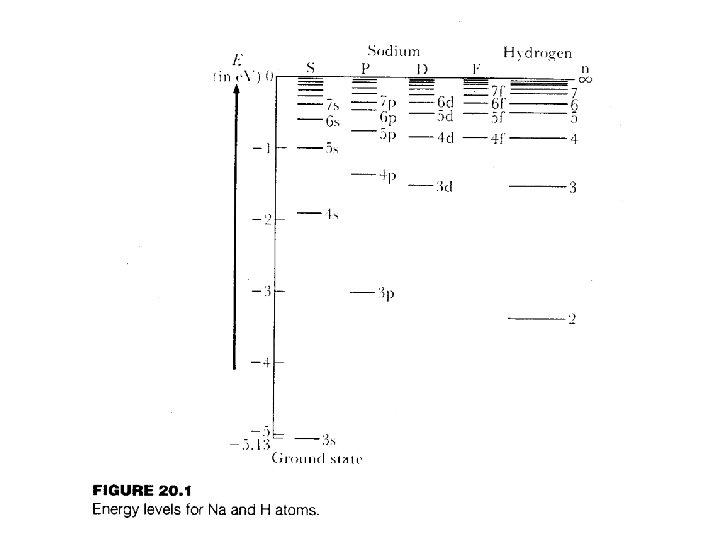
7. 4 & 7. 5 Multielectron Atoms

En = ( -13. 6 e. V ) (Z 2/n 2) r = n 2 ao / Z ao = 0. 529 Å
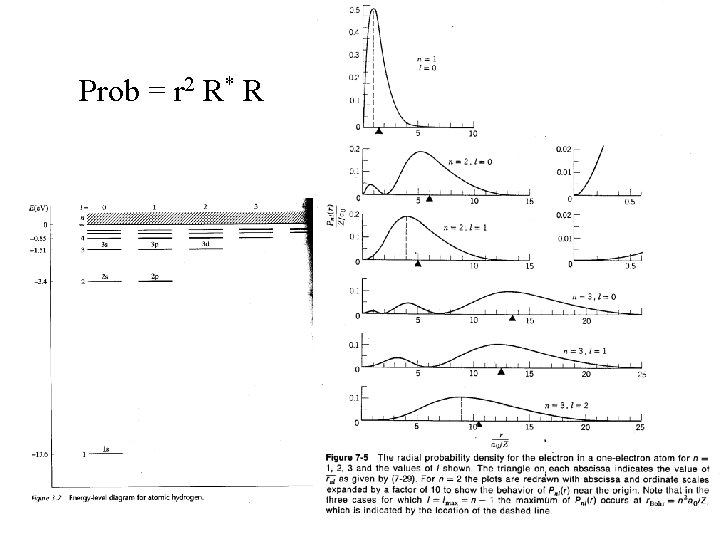
Prob = r 2 R* R
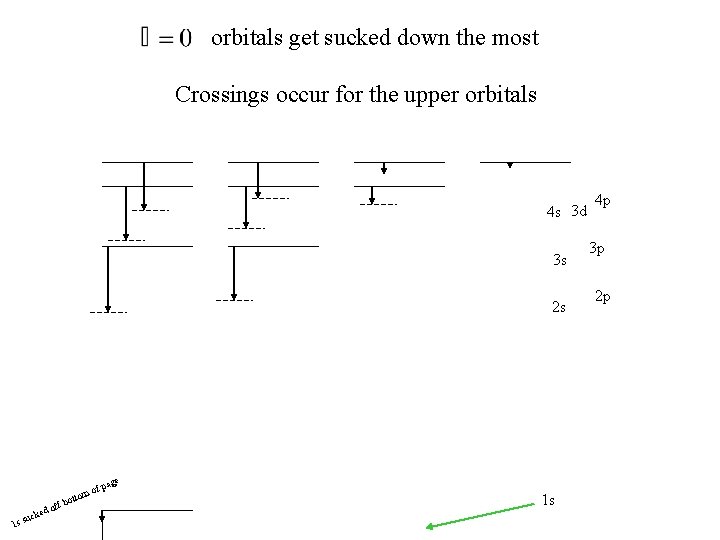
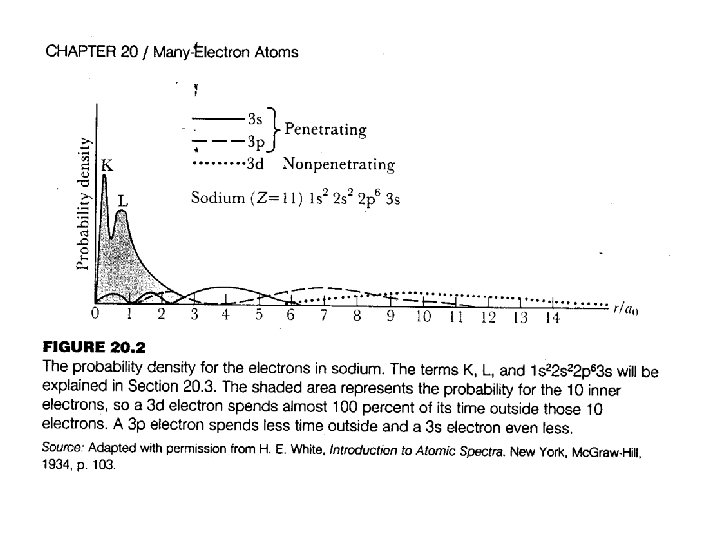
orbitals get sucked down the most Crossings occur for the upper orbitals 4 s 3 d 3 s 2 s age of p m otto 1 s ke suc f b d of 1 s 4 p 3 p 2 p
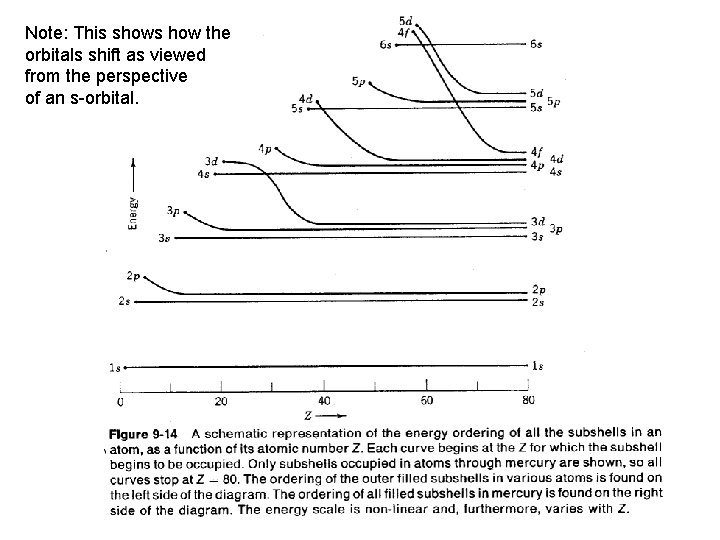
Note: This shows how the orbitals shift as viewed from the perspective of an s-orbital.
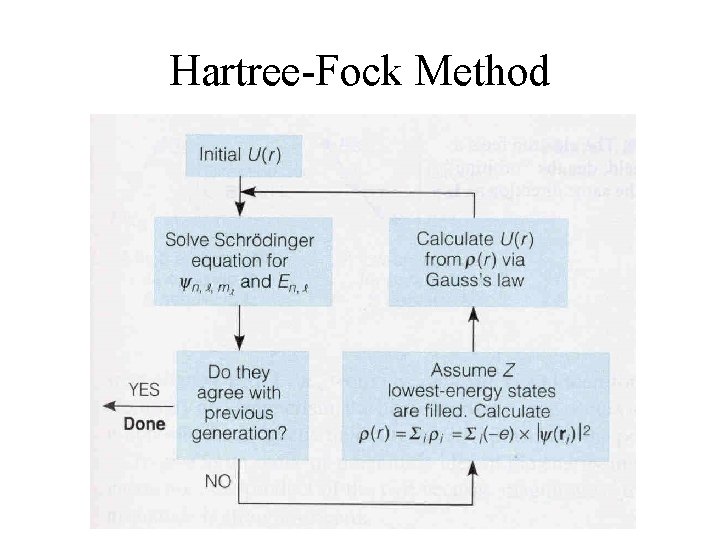
Hartree-Fock Method
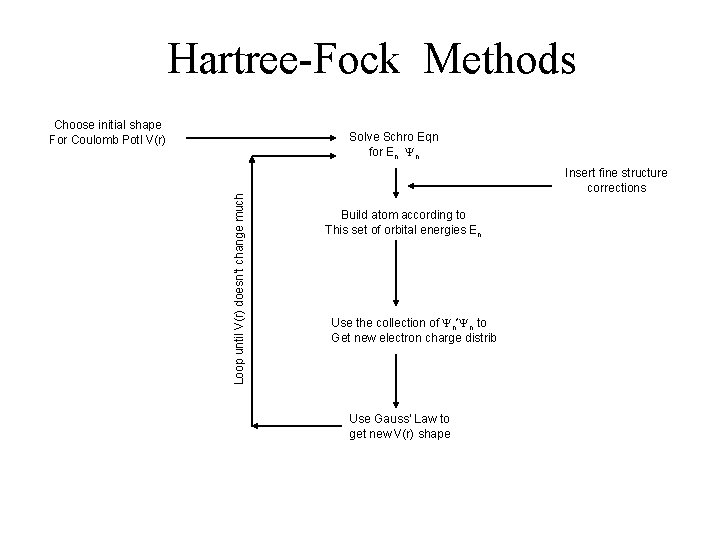
Hartree-Fock Methods Choose initial shape For Coulomb Potl V(r) Loop until V(r) doesn’t change much Solve Schro Eqn for En Yn Insert fine structure corrections Build atom according to This set of orbital energies En Use the collection of Yn*Yn to Get new electron charge distrib Use Gauss’ Law to get new V(r) shape
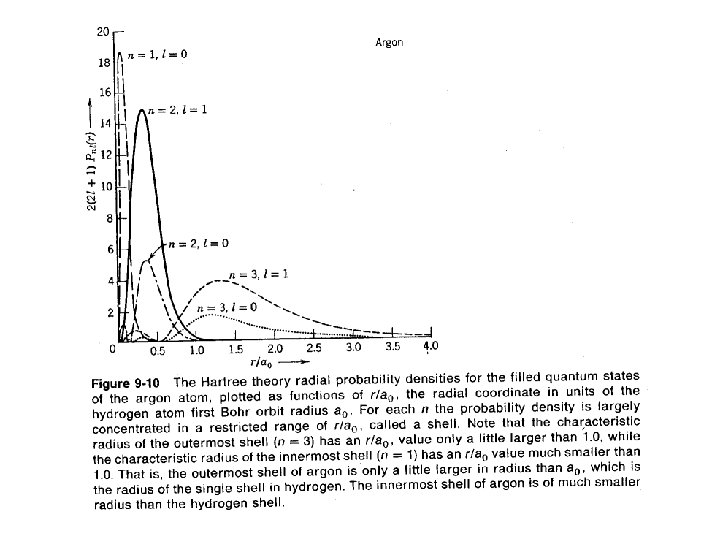
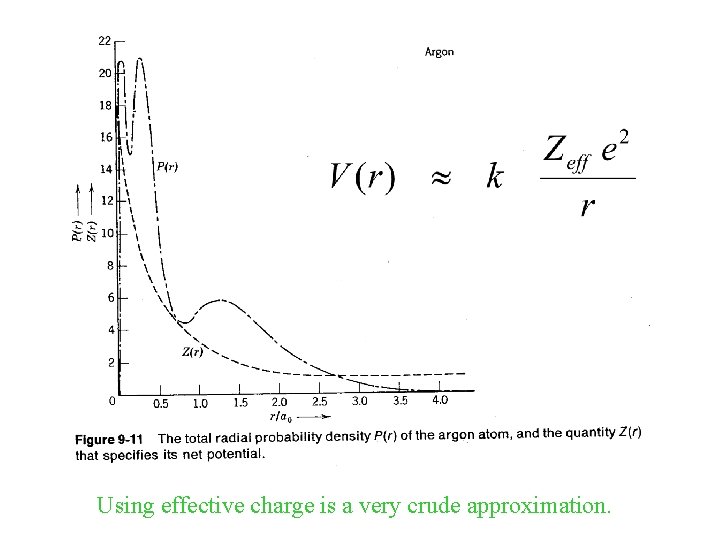
Using effective charge is a very crude approximation.
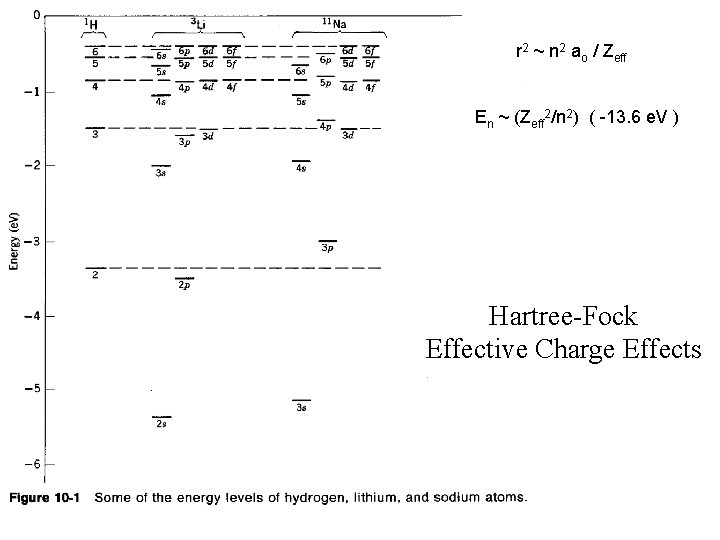
r 2 ~ n 2 ao / Zeff En ~ (Zeff 2/n 2) ( -13. 6 e. V ) Hartree-Fock Effective Charge Effects

7. 6 Spin-Orbit Effect

Corrections to the Coulomb Potl for H-atom • • Central Potential Spin-Orbit (electron viewpoint) Relativistic Spin (Thomas precession) Relativistic Kinetic Energy Spin-Orbit (nucleus viewpoint) Spin-Spin Impact of External Fields – Zeeman Effect (applied B-field) – Stark Effect (applied E-field)
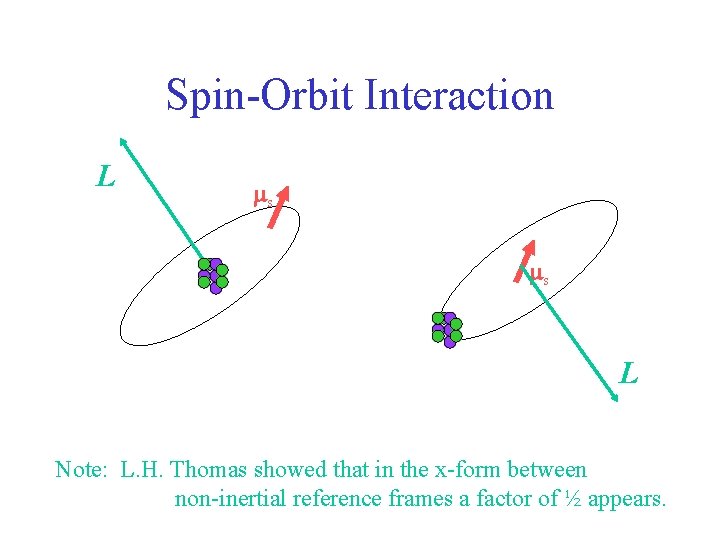
Spin-Orbit Interaction L ms ms L Note: L. H. Thomas showed that in the x-form between non-inertial reference frames a factor of ½ appears.
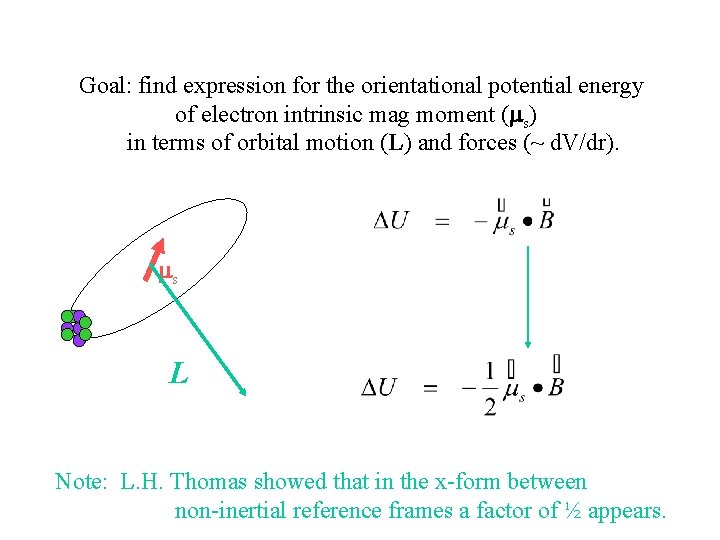
Goal: find expression for the orientational potential energy of electron intrinsic mag moment (ms) in terms of orbital motion (L) and forces (~ d. V/dr). ms L Note: L. H. Thomas showed that in the x-form between non-inertial reference frames a factor of ½ appears.
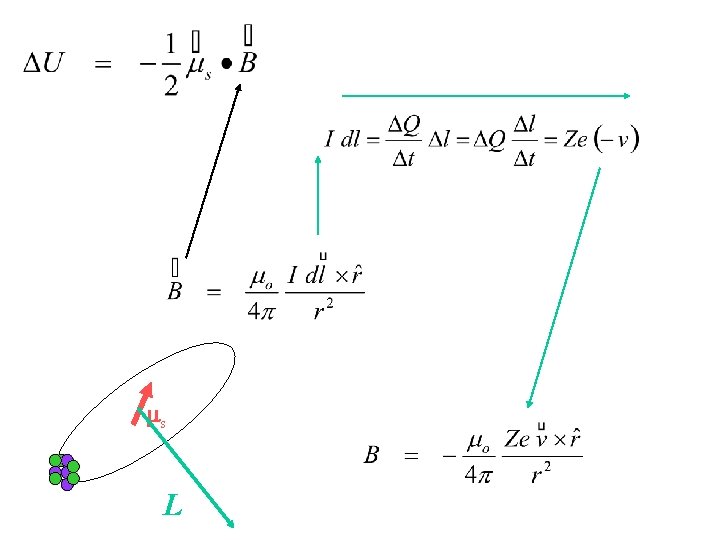
ms L
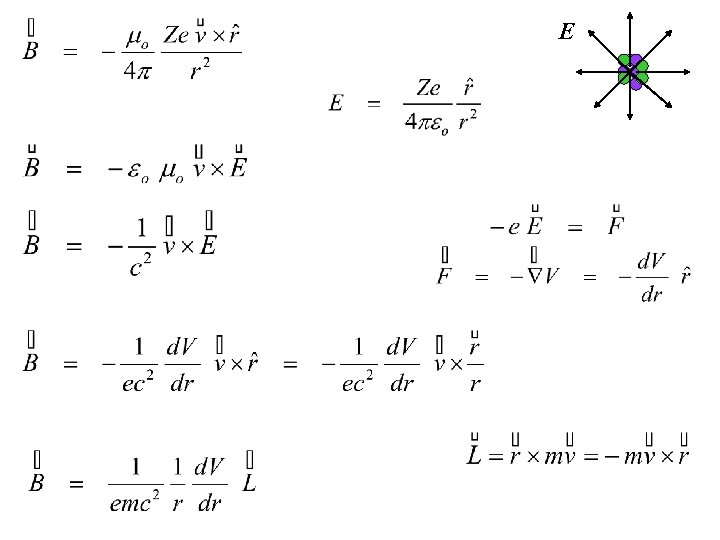
E
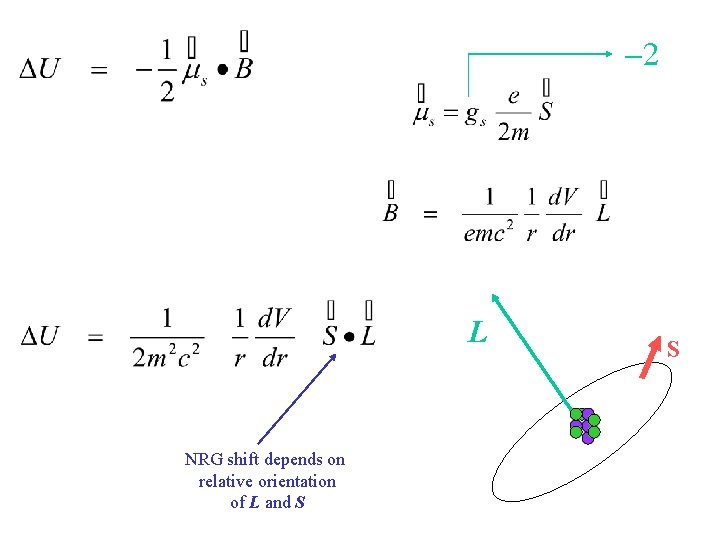
-2 L NRG shift depends on relative orientation of L and S S
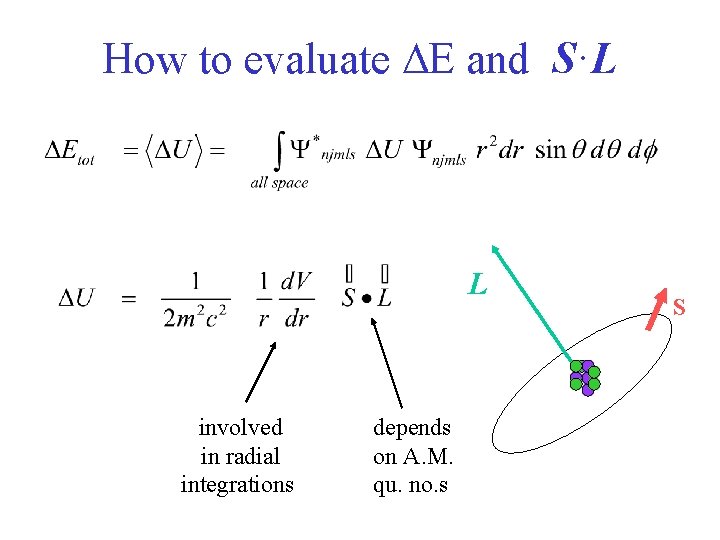
How to evaluate DE and S·L L involved in radial integrations depends on A. M. qu. no. s S
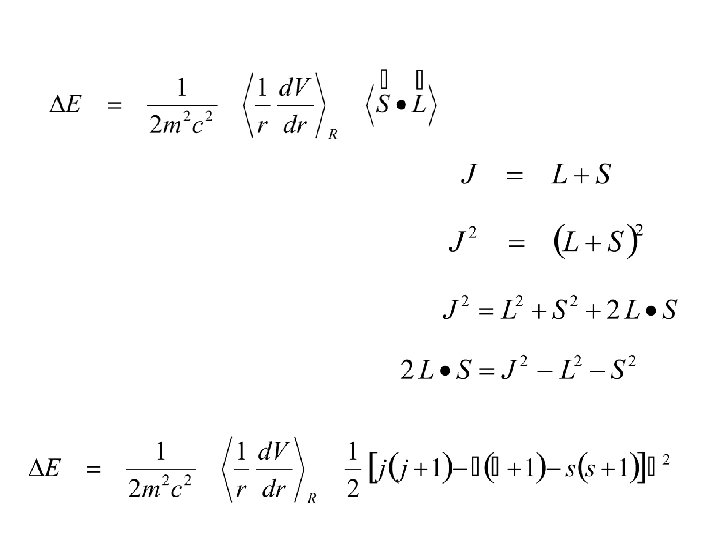
electron. Spin-Orbit “locks” the angle between L & S J is now a well-defined direction. S NOTE Lz is no longer well-defined ml not a good q. no. L J S L

Revised H-atom Level Scheme add in spin-orbit correction nlj not required to specify NRG j mj l ml s ms not required to specify NRG mj ml s ms absolutely worthless

electron Spin-Orbit is more important in higher-Z atoms fn’l expression only for H-atom, for all others, must come from Hartree procedure Splitting (e. V) Li Na K Rb Cs 0. 42 E-4 21. E-4 72. E-4 295. E-4 687. E-4
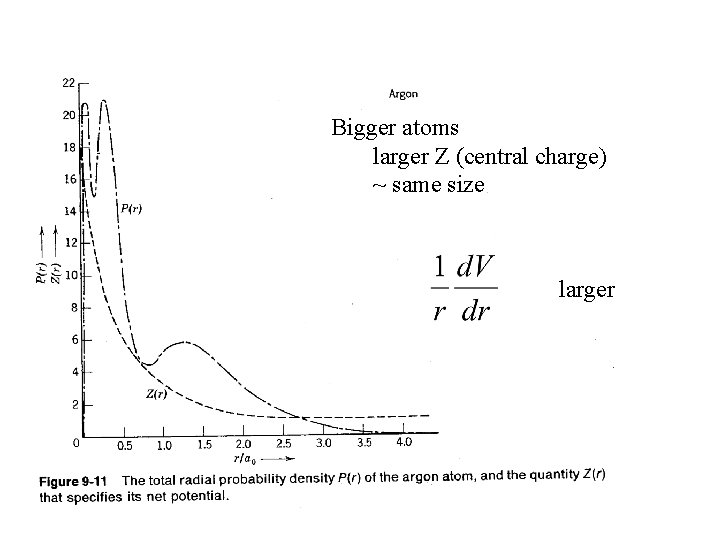
Bigger atoms larger Z (central charge) ~ same size larger

7. 7 QM Angular Momentum
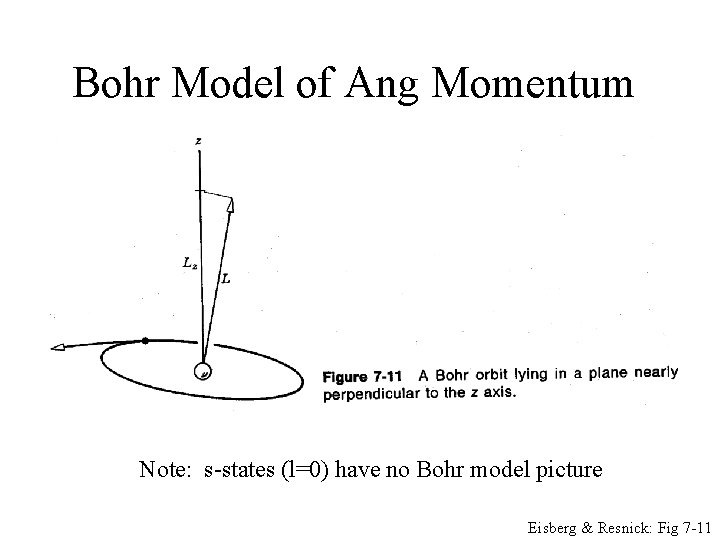
Bohr Model of Ang Momentum Note: s-states (l=0) have no Bohr model picture Eisberg & Resnick: Fig 7 -11
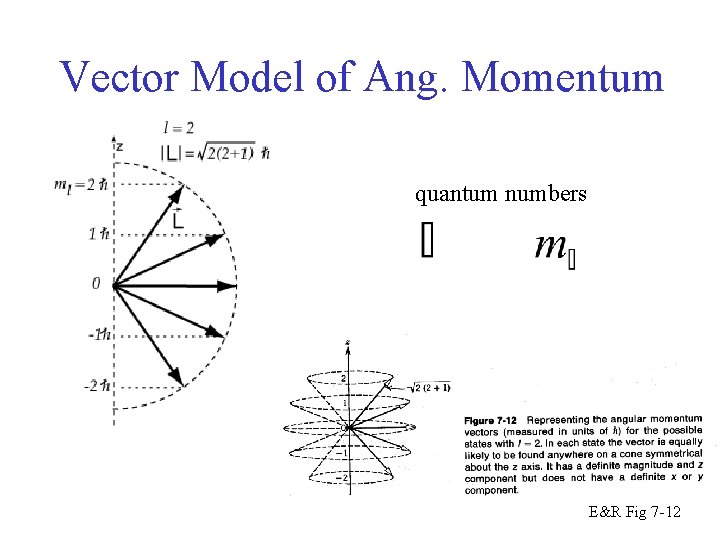
Vector Model of Ang. Momentum quantum numbers E&R Fig 7 -12

Edmonds “A. M. in QM” pg 19: “We might imagine the vector moving in an unobservable way about the z-axis. . . ” pg 29: “The QM probability density, not being time dependent, gives us no information about the motion of the particle in it’s orbit. ”

Morrison, Estle, Lane “Understanding More QM”, Prentice-Hall, 1991

ADDITION OF ANGULAR MOMENTUM L 2 Ltot = L 1 + L 2 L 1
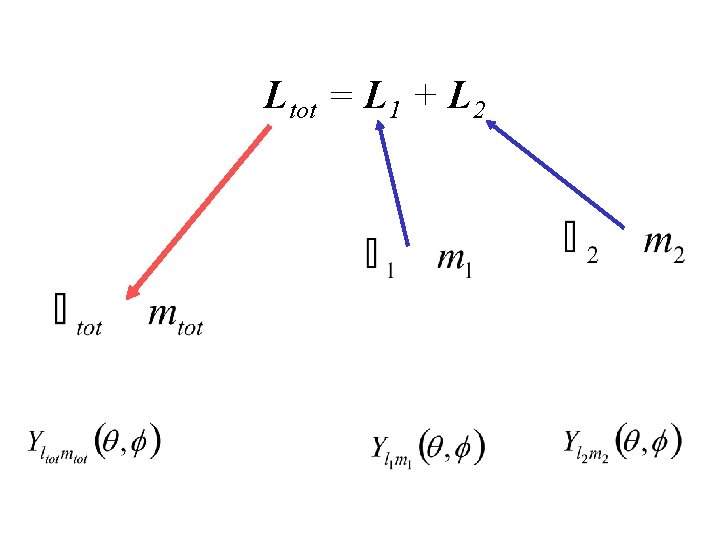
Ltot = L 1 + L 2
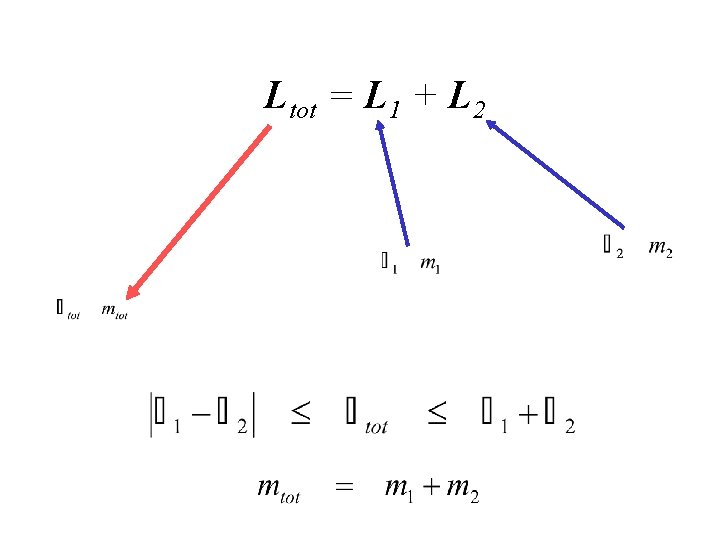
Ltot = L 1 + L 2

Addition of Angular Momentum www. bokerusa. com aligned configuration “aligned” does not mean straight www. cartowning. co. za/DBNRECGC. htm jack-knife configuration “jack-knife” does not mean antiparallel


Detailed Example L 1 L 2 Problem: Two objects each travel in a p-orbit ( l=1 ). The total energy of each object is degenerate wrt ml, so we have no detailed knowledge of ml. What are the allowed values of ltot, mtot ?
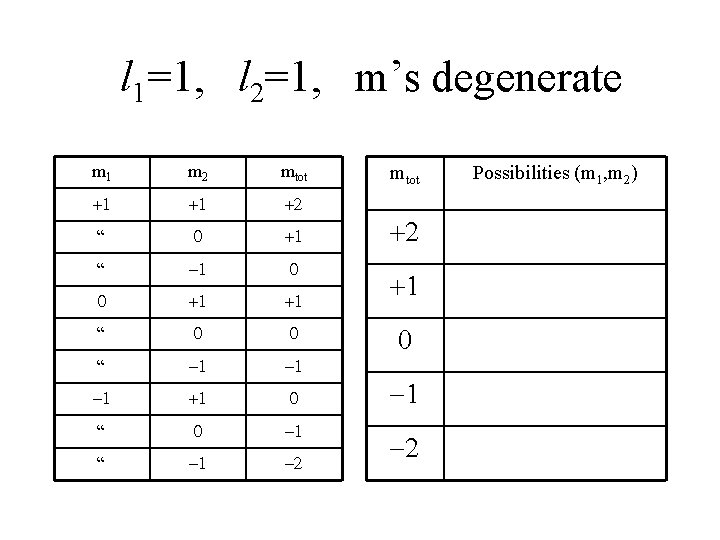
l 1=1, l 2=1, m’s degenerate m 1 m 2 mtot +1 +1 +2 “ 0 +1 “ -1 0 0 +1 +1 “ 0 0 “ -1 -1 -1 +1 0 “ 0 -1 “ -1 -2 mtot +2 +1 0 -1 -2 Possibilities (m 1, m 2)

Allowed Values of ltot mtot

Basic A. M. Math J=L+S J S L

Vector Representation of J
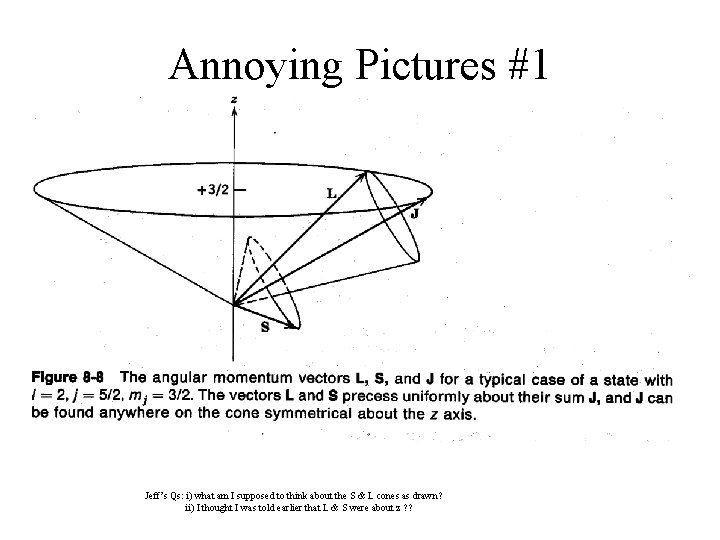
Annoying Pictures #1 Jeff’s Qs: i) what am I supposed to think about the S & L cones as drawn? ii) I thought I was told earlier that L & S were about z ? ?

Annoying Pictures #2 Jeff: Pictures such as this confuse the vector symbols L and S with the quantum numbers ℓ and s. For instance, how could L and S ever point in the same direction?

TOTAL ANGULAR MOMEMTUM J=L+S

More Detailed H-atom Level Scheme Energies & Spectra not sensitive to l ml Energies & Spectra not sensitive to j mj l ml s ms till next page
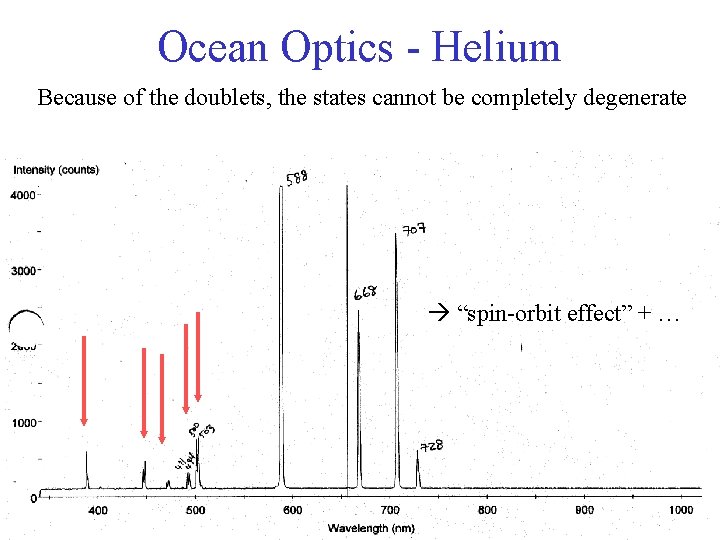
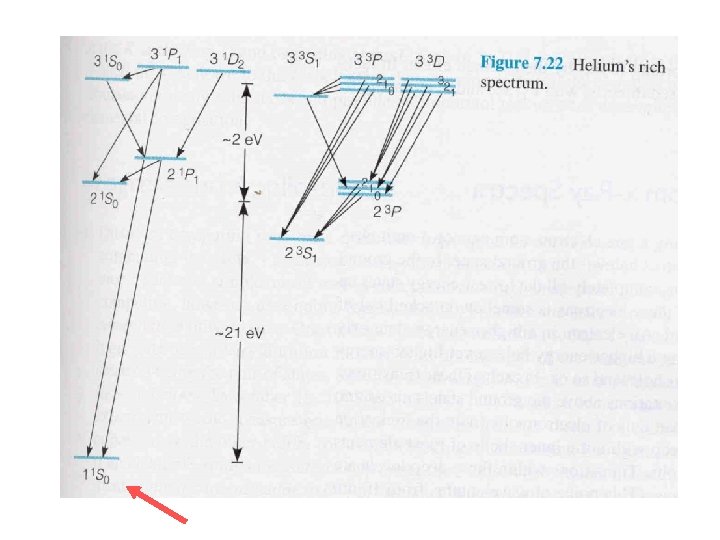
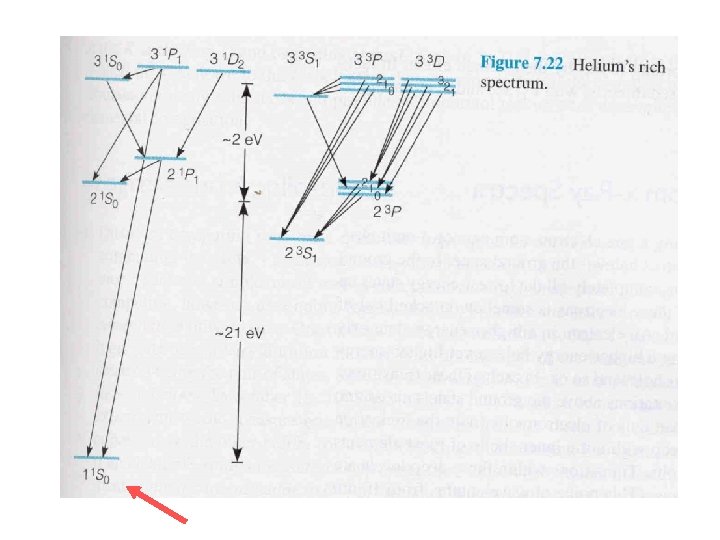
Ocean Optics - Helium Because of the doublets, the states cannot be completely degenerate “spin-orbit effect” + …
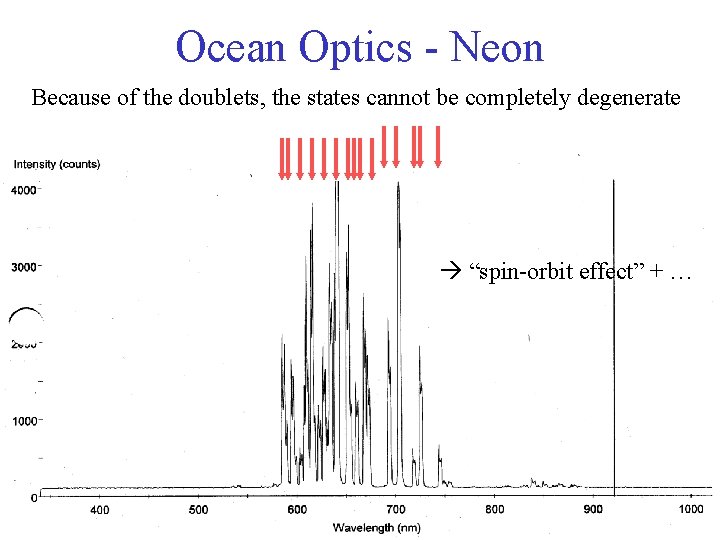
Ocean Optics - Neon Because of the doublets, the states cannot be completely degenerate “spin-orbit effect” + …

7. 9 Multi-electron Spectra

Multi-e Spectroscopic Notation QUANTUM NUMBERS principal: n ltot , stot jtot. Stot = S 1 + S 2 + … Ltot = L 1 + L 2 + … Jtot = Ltot + Stot stot = 1, ltot=0, jtot=1 2 S 1

Two Kinds of Notation • Where an individ electron is at • n l j – – 1 s 1/2 2 p 3/2 • A. M. for whole atom • 2 Stot+1 ltot jtot – 1 S 0 – 3 S 1 – 3 P 0 , 3 P 1, 3 P 2
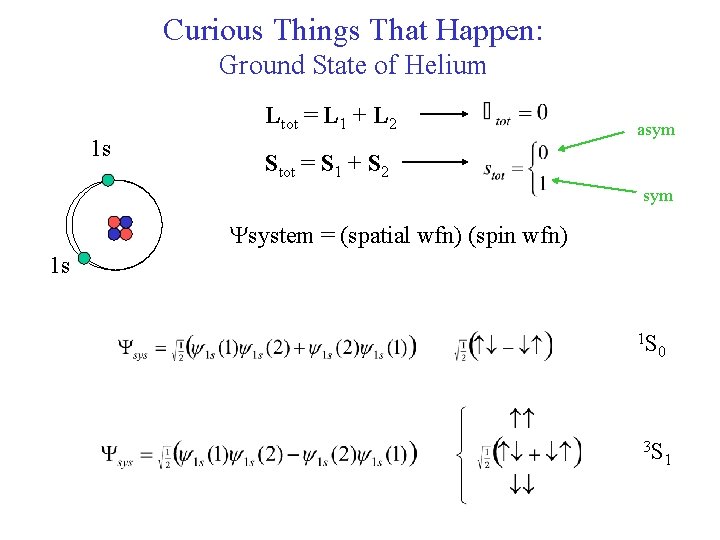
Curious Things That Happen: Ground State of Helium Ltot = L 1 + L 2 1 s asym Stot = S 1 + S 2 sym Ysystem = (spatial wfn) (spin wfn) 1 s 1 S 0 3 S 1

7. 8 Atoms in External Magnetic Fields -- the Zeeman Effect

Corrections to the Coulomb Potl for H-atom • • Central Potential Spin-Orbit (electron viewpoint) Relativistic Spin (Thomas precession) Relativistic Kinetic Energy Spin-Orbit (nucleus viewpoint) Spin-Spin Impact of External Fields – Zeeman Effect (applied B-field) – Stark Effect (applied E-field)

Weak-Field Zeeman • Hartree-Fock Coulomb & related Procedures • Fine Structure – spin-orbit ( jtot becomes important ) – relativistic • Zeeman H’Zeeman = - mtot * Bext < few 0. 1’s Tesla
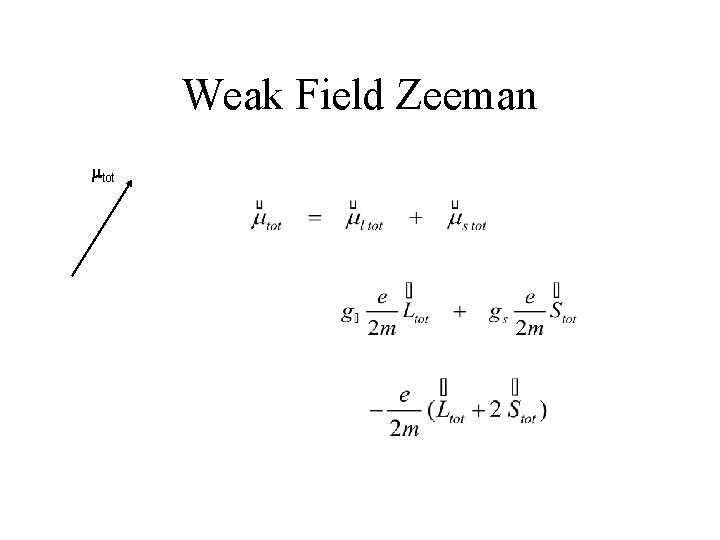
Weak Field Zeeman mtot
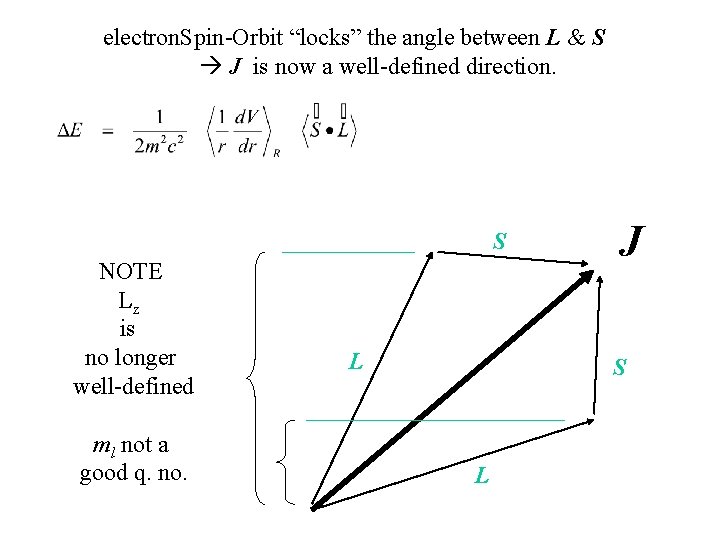
electron. Spin-Orbit “locks” the angle between L & S J is now a well-defined direction. S NOTE Lz is no longer well-defined ml not a good q. no. L J S L

Weak-Field Zeeman e. SO makes jtot good quantum number, mtot g Jtot mltot & mstot become ‘confused’ (near worthless). Jtot is ‘well-defined’ direction; jtot mjtot project average mtot onto Jtot
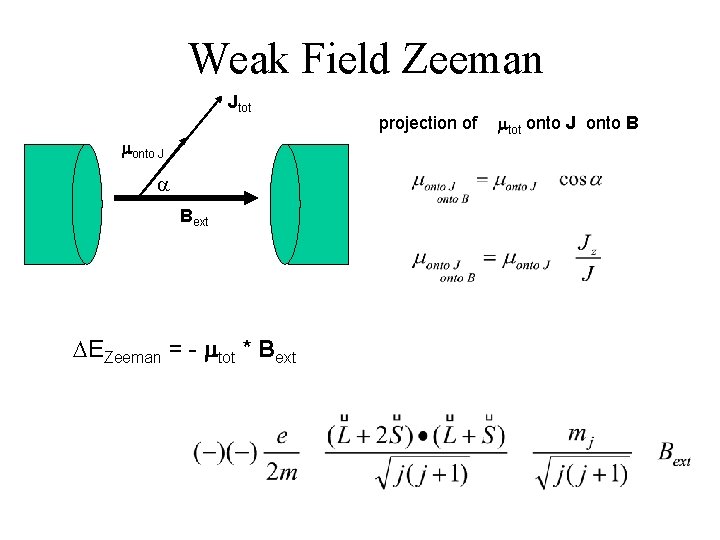
Weak Field Zeeman Jtot monto J a Bext DEZeeman = - mtot * Bext projection of mtot onto J onto B
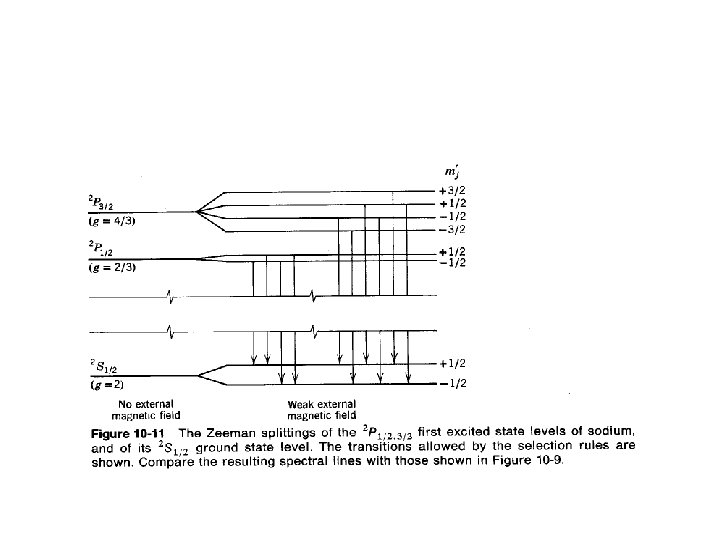
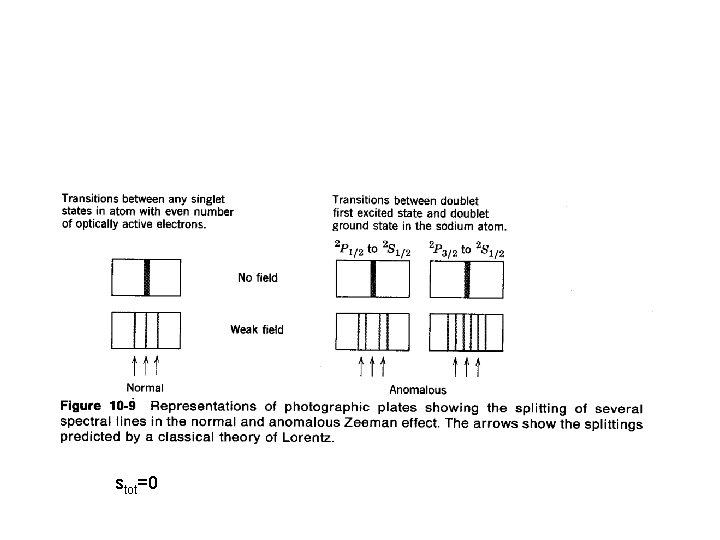
stot=0

Strong-Field Zeeman • Hartree-Fock Coulomb & related procedures • Zeeman • Fine Structure – spin-orbit – relativistic H’Zeeman = - mtot * Bext
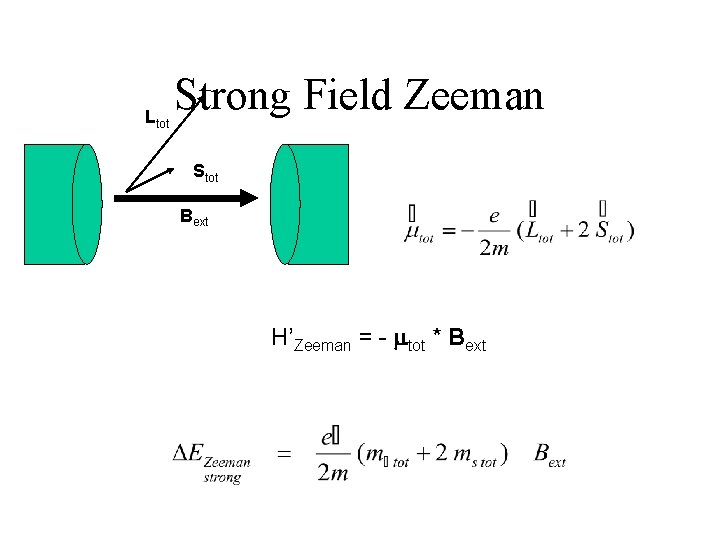
Ltot Strong Field Zeeman Stot Bext H’Zeeman = - mtot * Bext

- Slides: 103