HARMONEE Results Indepth with Bioabsorbable PolymerBased Sirolimus DES

HARMONEE: Results In-depth with Bioabsorbable Polymer-Based Sirolimus DES with a Luminal EPC Capture Shigeru Saito, MD, FACC, FSCAI President, Director of Cardiology & Catheterization Laboratories Shonan Kamakura General Hospital Kamakura, Japan

Shigeru SAITO, MD I have no relevant financial relationships
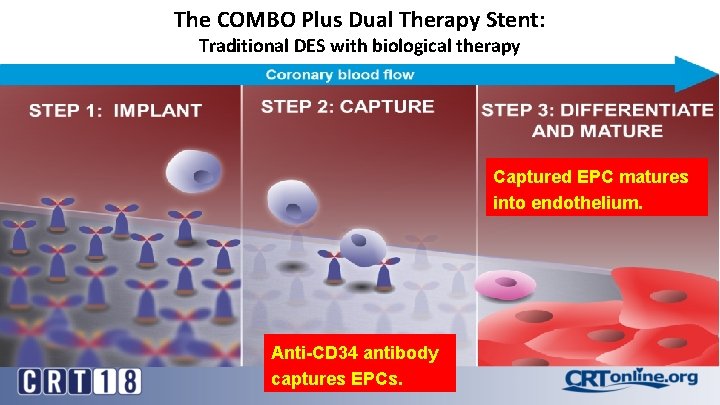
The COMBO Plus Dual Therapy Stent: Traditional DES with biological therapy Captured EPC matures into endothelium. Anti-CD 34 antibody captures EPCs.
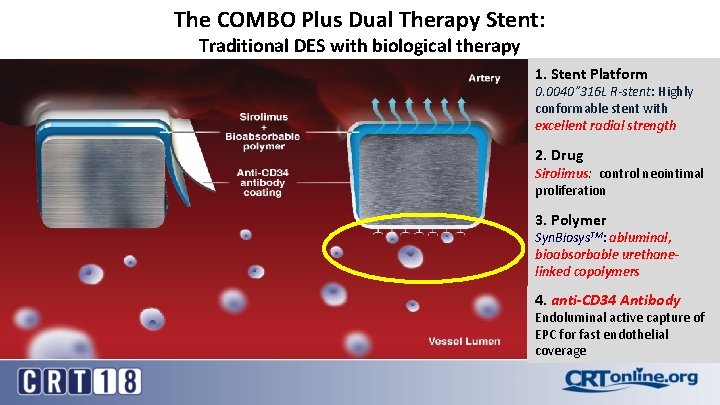
The COMBO Plus Dual Therapy Stent: Traditional DES with biological therapy 1. Stent Platform 0. 0040” 316 L R-stent: Highly conformable stent with excellent radial strength 2. Drug Sirolimus: control neointimal proliferation 3. Polymer Syn. Biosys. TM: abluminal, bioabsorbable urethanelinked copolymers 4. anti-CD 34 Antibody Endoluminal active capture of EPC for fast endothelial coverage

Randomized Registration Study for COMBO HBD Proof of Concept Global Trial Program Japan PMDA & U. S. FDA Uchida T et al, Circulation Journal 2013

HARMONEE Design & Methods Inclusion: • • • CSA/functional ischemia, USA or Non-STEMI ACS Up to 3 de novo lesions in 2 vessels 2. 5 -3. 5 mm diameter, <29 mm length Exclusion: • • • Kong DF et al Am Heart J 2017; 187: 112 -121 • • • STEMI < 7 days Unstable Sx, rhythm or BP (shock) Efx <30% Cr >2. 5 or ESRD Intolerance of 1 year DAPT Other life-limiting co-morbidity Current immunosuppressive Rx Prior murine-based Rx Anatomic: left main; SVG; CTO; bifurcation; extreme angulation; heavy calcium
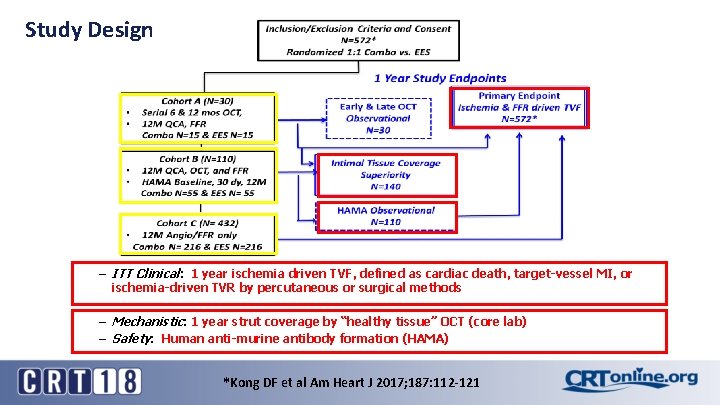
Study Design – ITT Clinical: 1 year ischemia driven TVF, defined as cardiac death, target-vessel MI, or ischemia-driven TVR by percutaneous or surgical methods – Mechanistic: 1 year strut coverage by “healthy tissue” OCT (core lab) – Safety: Human anti-murine antibody formation (HAMA) *Kong DF et al Am Heart J 2017; 187: 112 -121

HARMONEE Trial Organization
HARMONEE Japan 33 Clinical Site List with Enrollment
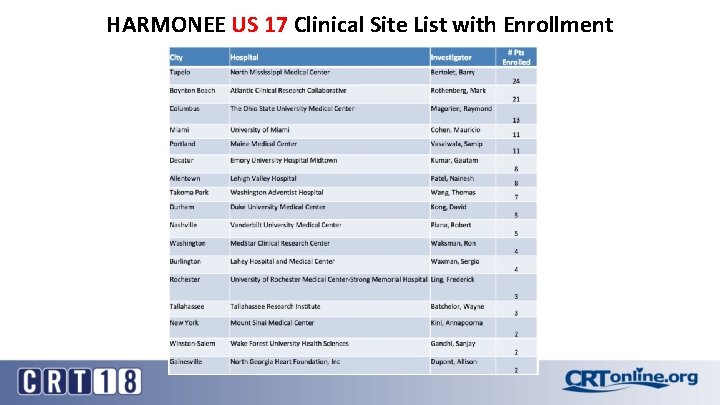
HARMONEE US 17 Clinical Site List with Enrollment

Statistical Plan Primary Clinical Endpoint: 1 year TVF • • Non-inferiority comparison of COMBO vs EES • Power assumption: 9% control arm TVF and 7% NI margin • Serial testing for superiority “Assay sensitivity”: superiority to imputed BMS* • Power assumption: 9% control arm TVF Mechanistic Endpoint: In-vivo EPC “healthy tissue” activity: • Superiority of COMBO vs EES: • % strut-level coverage with >40 micron thickness and negative FFR (OCT) Safety: • Human anti-murine anti-body (HAMA) conversion *Kong DF et al Am Heart J 2017; 187: 112 -121
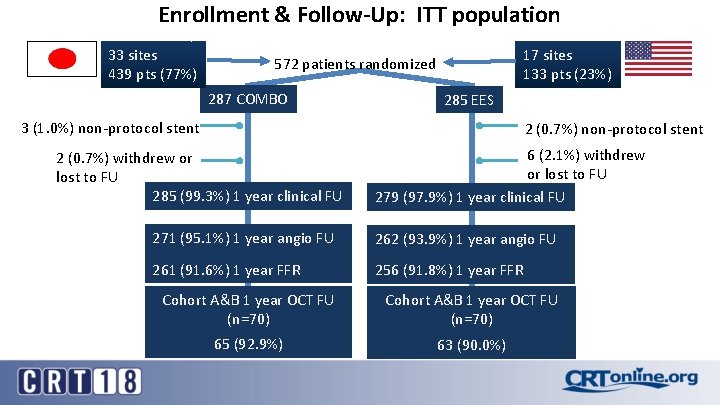
Enrollment & Follow-Up: ITT population 33 sites 439 pts (77%) 17 sites 133 pts (23%) 572 patients randomized 287 COMBO 285 EES 3 (1. 0%) non-protocol stent 2 (0. 7%) withdrew or lost to FU 285 (99. 3%) 1 year clinical FU 6 (2. 1%) withdrew or lost to FU 279 (97. 9%) 1 year clinical FU 271 (95. 1%) 1 year angio FU 262 (93. 9%) 1 year angio FU 261 (91. 6%) 1 year FFR 256 (91. 8%) 1 year FFR Cohort A&B 1 year OCT FU (n=70) 65 (92. 9%) 63 (90. 0%)
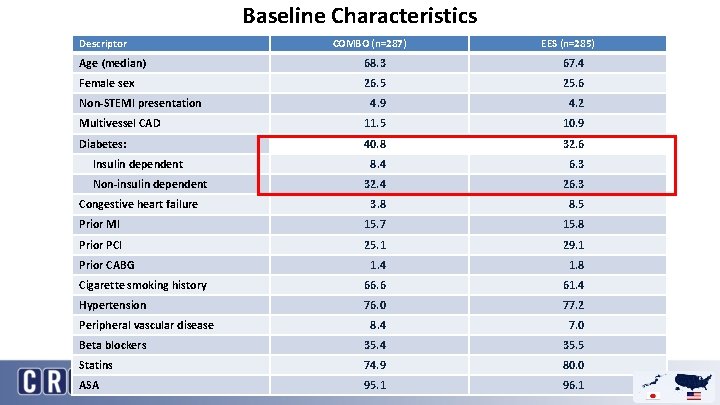
Baseline Characteristics Descriptor COMBO (n=287) EES (n=285) Age (median) 68. 3 67. 4 Female sex 26. 5 25. 6 4. 9 4. 2 Multivessel CAD 11. 5 10. 9 Diabetes: 40. 8 32. 6 8. 4 6. 3 32. 4 26. 3 3. 8 8. 5 Prior MI 15. 7 15. 8 Prior PCI 25. 1 29. 1 1. 4 1. 8 Cigarette smoking history 66. 6 61. 4 Hypertension 76. 0 77. 2 8. 4 7. 0 Beta blockers 35. 4 35. 5 Statins 74. 9 80. 0 ASA 95. 1 96. 1 Non-STEMI presentation Insulin dependent Non-insulin dependent Congestive heart failure Prior CABG Peripheral vascular disease
![ITT Cohort Site-read Angiographic Characteristics [mean (SD)] Descriptor COMBO (n=287) EES (n=285) LAD PCI ITT Cohort Site-read Angiographic Characteristics [mean (SD)] Descriptor COMBO (n=287) EES (n=285) LAD PCI](http://slidetodoc.com/presentation_image/3e0f47a438f5b87b7be6bb1e64498763/image-14.jpg)
ITT Cohort Site-read Angiographic Characteristics [mean (SD)] Descriptor COMBO (n=287) EES (n=285) LAD PCI (%) 43. 9 52 Lesion Length (mm) 14. 82 (5. 95) 14. 69 (5. 76) Reference Vessel Diameter (mm) 3. 02 (0. 36) 3. 06 (0. 38) Stent length (mm) 20. 0 (6. 2) 19. 6 (6. 0) % stenosis pre- 83. 6 (9. 6) 84. 5 (9. 4) % stenosis post- 1. 0 (4. 8) 0. 9 (3. 8)

Primary Clinical Endpoint: 1 year TVF non-inferiority

1 Year Target Vessel Failure (TVF)* Combo vs. EES Non-inferiority** (ITT Population) TVF Combo (N=287) EES (N=285) 20 (7. 0%) 12 (4. 2%) Difference (95%CI) 0. 028 (-0. 010, 0. 065) Non-inferiority p-value 0. 020 *Composite of cardiac death, target-vessel MI, or ischemia-driven target-vessel revascularization (TVR) by percutaneous or surgical methods. ** Non-inferiority margin 0. 07
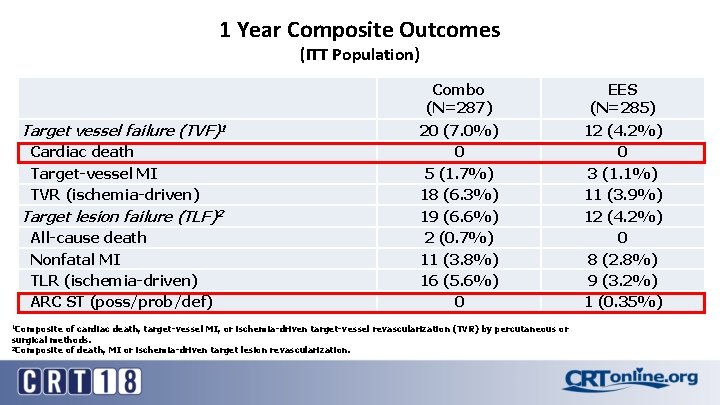
1 Year Composite Outcomes (ITT Population) Target vessel failure (TVF)1 Cardiac death Target-vessel MI TVR (ischemia-driven) Target lesion failure (TLF)2 All-cause death Nonfatal MI TLR (ischemia-driven) ARC ST (poss/prob/def) Combo (N=287) EES (N=285) 20 (7. 0%) 0 5 (1. 7%) 18 (6. 3%) 19 (6. 6%) 2 (0. 7%) 11 (3. 8%) 16 (5. 6%) 0 12 (4. 2%) 0 3 (1. 1%) 11 (3. 9%) 12 (4. 2%) 0 8 (2. 8%) 9 (3. 2%) 1 (0. 35%) 1 Composite of cardiac death, target-vessel MI, or ischemia-driven target-vessel revascularization (TVR) by percutaneous or surgical methods. 2 Composite of death, MI or ischemia-driven target lesion revascularization.
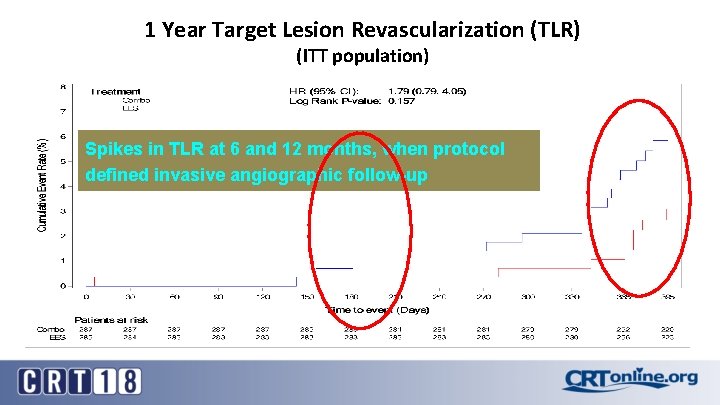
1 Year Target Lesion Revascularization (TLR) (ITT population) Spikes in TLR at 6 and 12 months, when protocol defined invasive angiographic follow-up
![1 Year QCA Core Laboratory Late Loss [mean (SD)]* (Cohorts A and B: N=140) 1 Year QCA Core Laboratory Late Loss [mean (SD)]* (Cohorts A and B: N=140)](http://slidetodoc.com/presentation_image/3e0f47a438f5b87b7be6bb1e64498763/image-19.jpg)
1 Year QCA Core Laboratory Late Loss [mean (SD)]* (Cohorts A and B: N=140) In-segment late loss or %restenosis were similar. N (lesions/patients) Combo EES 77/65 76/66 In-stent late loss (mm) 0. 293 (0. 435) 0. 219 (0. 352) In-segment late loss (mm) 0. 229 (0. 398) 0. 220 (0. 359) In-stent 1 (1. 3%) 2 (2. 6%) In-segment 2 (2. 5%) 3 (3. 9%) Restenosis * All comparisons p=NS

To test EPC Technology: Primary Mechanistic Endpoint 1 year “healthy tissue”-- superiority (OCT core laboratory)
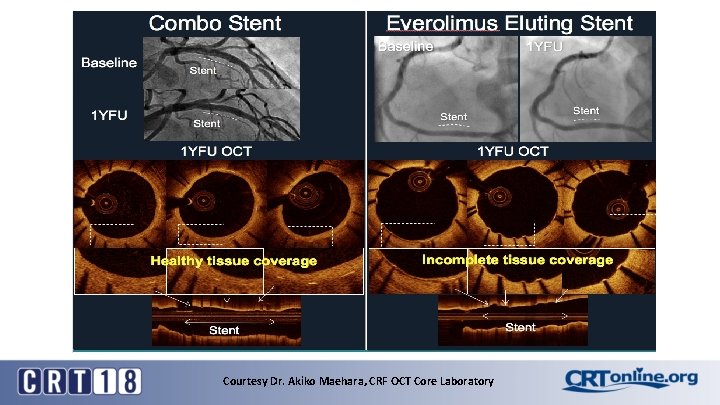
Courtesy Dr. Akiko Maehara, CRF OCT Core Laboratory
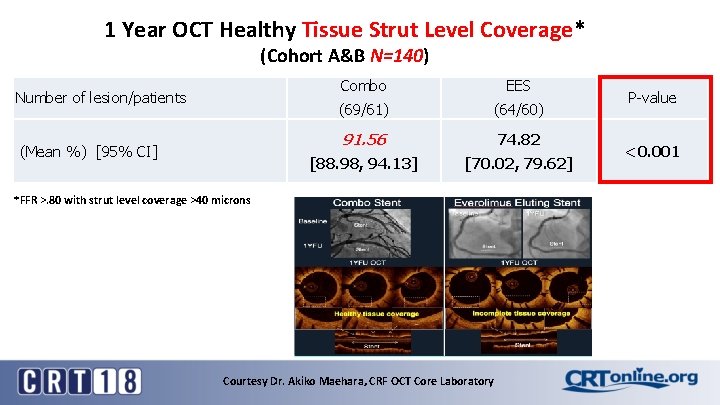
1 Year OCT Healthy Tissue Strut Level Coverage* (Cohort A&B N=140) Number of lesion/patients (Mean %) [95% CI] Combo EES (69/61) (64/60) 91. 56 74. 82 [88. 98, 94. 13] [70. 02, 79. 62] *FFR >. 80 with strut level coverage >40 microns Courtesy Dr. Akiko Maehara, CRF OCT Core Laboratory P-value <0. 001

1 Year OCT Qualitative Homogeneous Neointimal Tissue (Cohorts A and B, N=140) Number of lesion/patients Homogenous NIT Combo EES (69/61) (64/60) 81. 2% 68. 8%

At 1 year, those were not observed: Zero HAMA conversions Zero Stent Thrombosis

Conclusions 1. 1 year TVF outcomes with Combo vs. EES met the non-inferiority boundary 2. OCT suggests an active mechanistic role of EPC technology in vivo, including: • • Superior healthy tissue strut coverage with Combo vs. EES More homogeneous tissue qualitatively with Combo vs. EES 3. Combo 1 year late loss and binary restenosis were similar to EES 4. There were no safety concerns with Combo (0 HAMA conversions, 0 ST) 5. TVR events “spikes” were clearly evident in conjunction with 1 year protocol re-catheterization, despite the use of FFR 6. International Japan-USA collaboration between regulatory authorities, clinical sites and manufacturers provides a unique platform for investigational devices studies

Thank you
- Slides: 26