Happy Monday Take out your notebook and a

Happy Monday! • Take out your notebook and a pen/pencil • First volunteer group earns a 10% boost in overall score. Talk to me. • Summer Reading Book Presentations 1
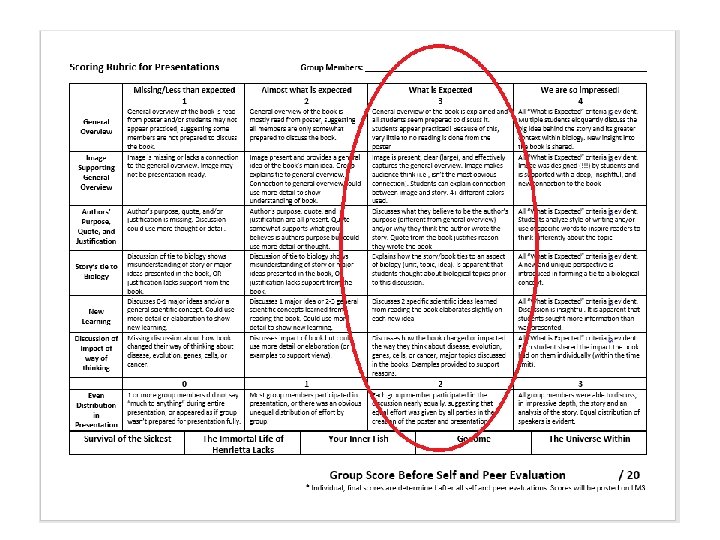

Notes on Summer Reading Books Names of Peers Book they read Their general overview Their connection to Biology

Happy Tuesday! • Summer Reading Book Presentations, c’td • Self and Peer Assessment – Final grades only posted once this is completed by ALL group members and group receives rubric (physical copy) • Reflection (due Weds) 4

Silent Reflection One of the goals of the summer reading assignment was to open your mind to ideas you will learn this year in biology and give you a little context for what’s ahead. After listening to your peers discuss the 5 books from the presentations, explain in a short paragraph what you think we will cover this year in biology, and elaborate. To support each idea you list, use specific examples that were mentioned during presentations. If you finish, see me for stamps. If you need more time, this is tonight’s homework. Due Wednesday

DO NOW Wednesday 10/5 Refer back to the Evidence Based Arguments form (brown) we started last week. List the reactants and products of the ethanol burning reaction. Checking: Open notebooks to Summer Reading Book Reflection and await on-time “stamp” 6

Evidence-Based Arguments 7

Evidence-Based Arguments (Per 1) - BTB turned green Mass of Ethanol decreased - CO 2 moved from ethanol to BTB - BTB went from blue green Condensation! - When ethanol burned, CO 2 was produced Water was produced! - CH 3 CH 2 OH CO 2 + H 2 O - Ethanol has chemical potential energy When lit, it has (releases, changes, produces) thermal and light energy - - There was a flame The flame died - - What other molecules left/moved from ethanol? 8

Evidence-Based Arguments (Per 3) - - Mass of ethanol decreased Condensation! Color of BTB: blue yellow - CO 2 moved into BTB Water moved from ethanol to side of the container BTB went from blue. teal yellow. greenblue Fire went out Condensation - CH 3 CH 2 OH + O 2 CO 2 + H 2 O Fire went out Mass of ethanol decreased 9

Evidence-Based Arguments (Per 4) - m - BTB - Fire - CO 2 m 10

Evidence-Based Arguments (Per 5) - m - BTB - Fire - CO 2 m 11

Evidence-Based Arguments (Per 6) - m - BTB - Fire - CO 2 m 12

Carbon: Transformations in Matter and Energy Environmental Literacy Project Michigan State University Systems and Scale Unit Activity 4. 4 Molecular Models for Ethanol Burning
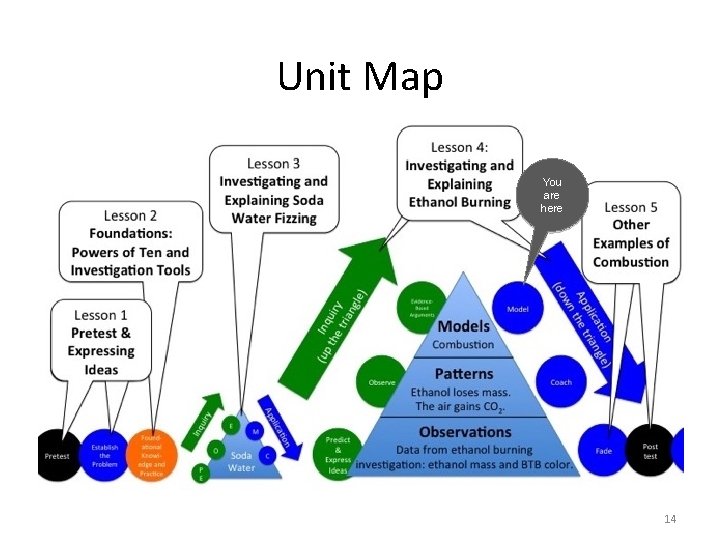
Unit Map You are here 14

Discuss: Why is the burning of ethanol important or worth studying? 15

ZOOMING INTO A Flame 16
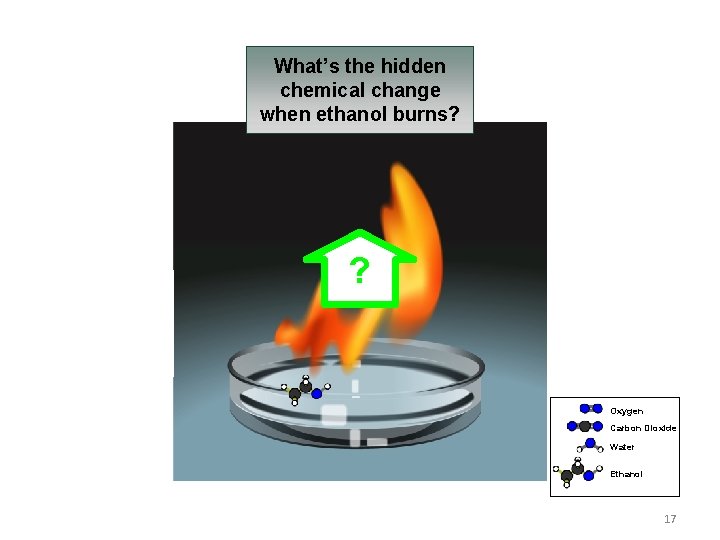
What’s the hidden chemical change when ethanol burns? ? Oxygen Carbon Dioxide Water Ethanol 17
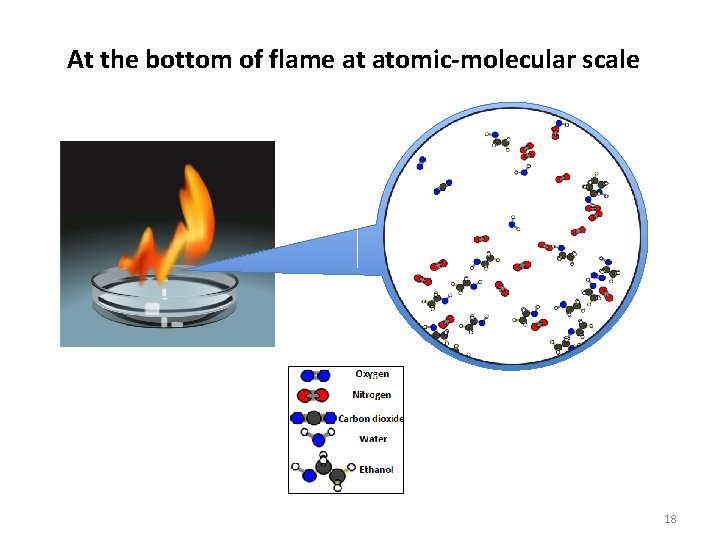
At the bottom of flame at atomic-molecular scale 18
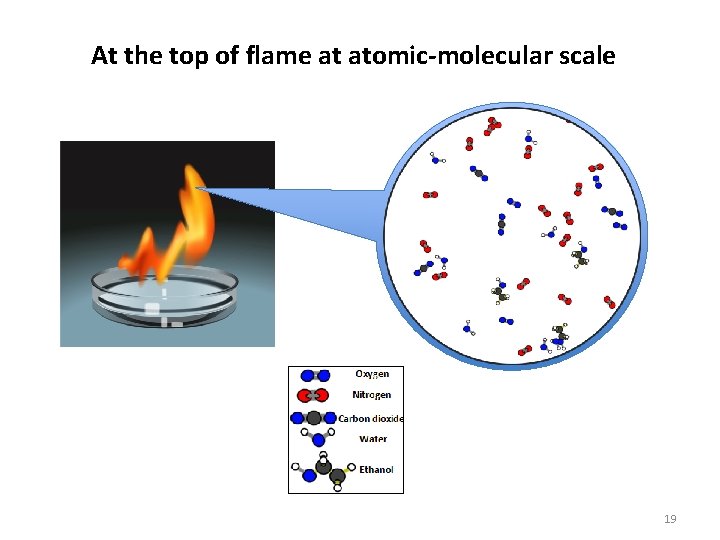
At the top of flame at atomic-molecular scale 19

How Atoms Bond Together in Molecules • Atoms in stable molecules always have a certain number of bonds to other atoms. • Oxygen atoms do NOT bond to other oxygen atoms if they can bond to carbon or hydrogen instead. • Chemical energy is stored in bonds between atoms AND the overall structure of the molecule. • C-C and C-H bonds are high energy bonds • C-O and H-O bonds are low energy bonds 20

An example… To show that chemical energy is stored within bonds AND molecular structure of the molecule: Isopropyl Alcohol vs. 1 -Propanol Curious about why? 21

DO NOW Thursday 10/6 Write out the molecular formula for ethanol. Extra challenge: Ethanol can be written in several ways. Write the various ways ethanol can be written. 22

Making the reactant molecules: ethanol and oxygen Remember the Bonding Rules: Carbon – 4 Oxygen – 2 Hydrogen – 1

Photo of reactant molecules: C 2 H 5 OH (ethanol) and O 2 (oxygen) Start by making the molecules and energy units of the reactants and putting them on the reactants side, then rearrange the atoms and energy units to show the products. Chemical change Ethanol Oxygen Reactants Products Remember: Atoms last forever (so you can rearrange atoms into new molecules, but can’t add or subtract atoms). Energy lasts forever (so you can change forms of energy, but energy units can’t appear or go away) 24
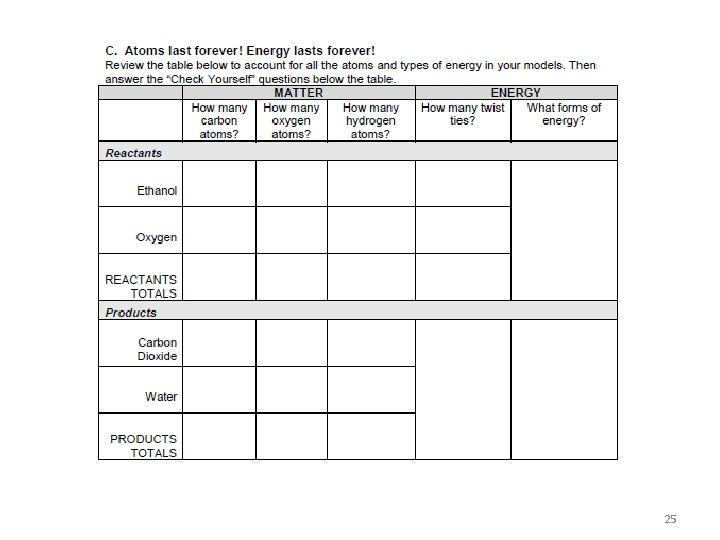
25

Important: When you are finished constructing the reactants, put all extra pieces away. 26

Rearranging to Make Product Molecules: Carbon Dioxide and Water
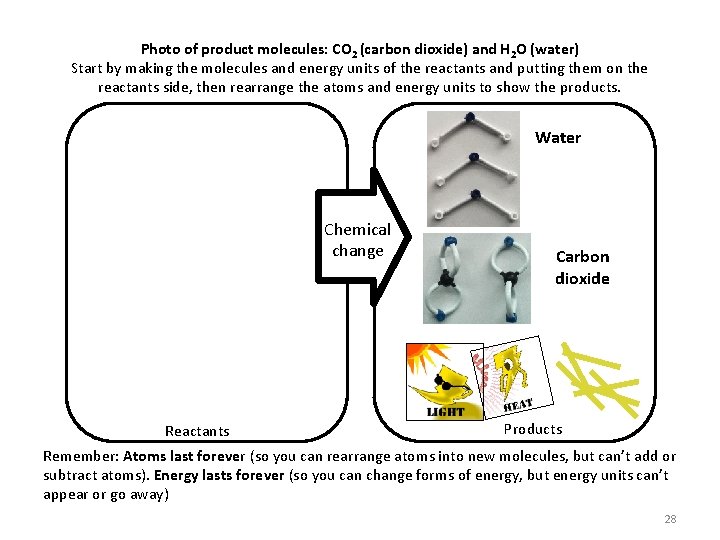
Photo of product molecules: CO 2 (carbon dioxide) and H 2 O (water) Start by making the molecules and energy units of the reactants and putting them on the reactants side, then rearrange the atoms and energy units to show the products. Water Chemical change Reactants Carbon dioxide Products Remember: Atoms last forever (so you can rearrange atoms into new molecules, but can’t add or subtract atoms). Energy lasts forever (so you can change forms of energy, but energy units can’t appear or go away) 28
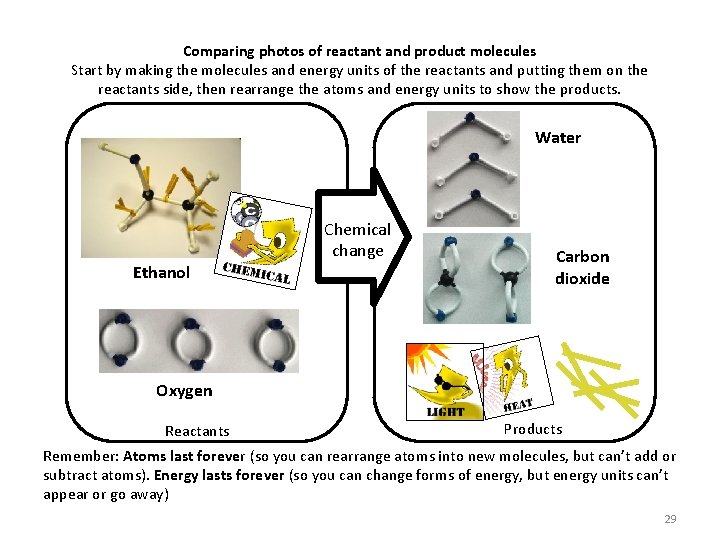
Comparing photos of reactant and product molecules Start by making the molecules and energy units of the reactants and putting them on the reactants side, then rearrange the atoms and energy units to show the products. Water Chemical change Ethanol Carbon dioxide Oxygen Reactants Products Remember: Atoms last forever (so you can rearrange atoms into new molecules, but can’t add or subtract atoms). Energy lasts forever (so you can change forms of energy, but energy units can’t appear or go away) 29
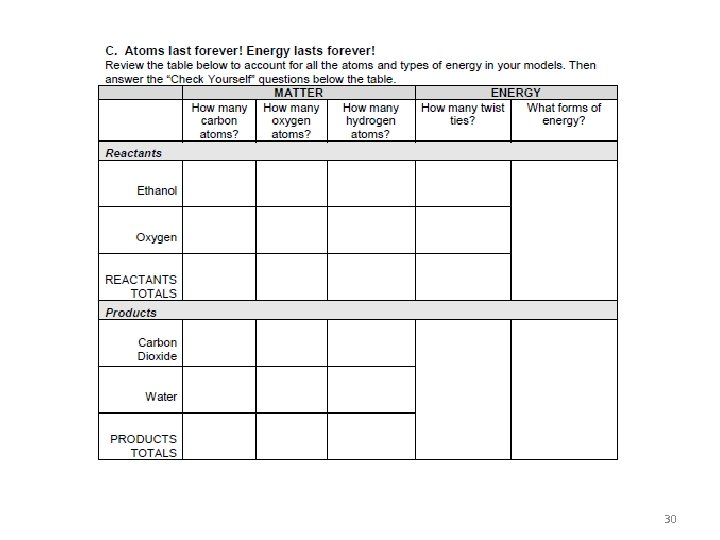
30
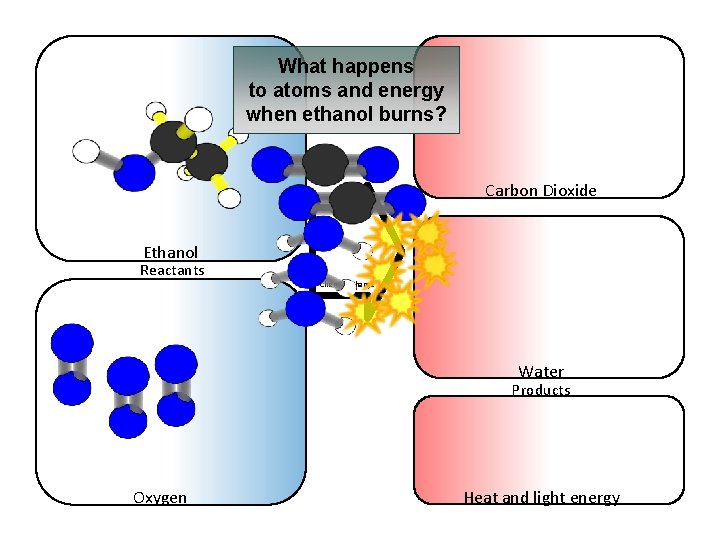
What happens to atoms and energy when ethanol burns? Carbon Dioxide Ethanol Reactants Chemical change Water Products Oxygen Heat and light energy
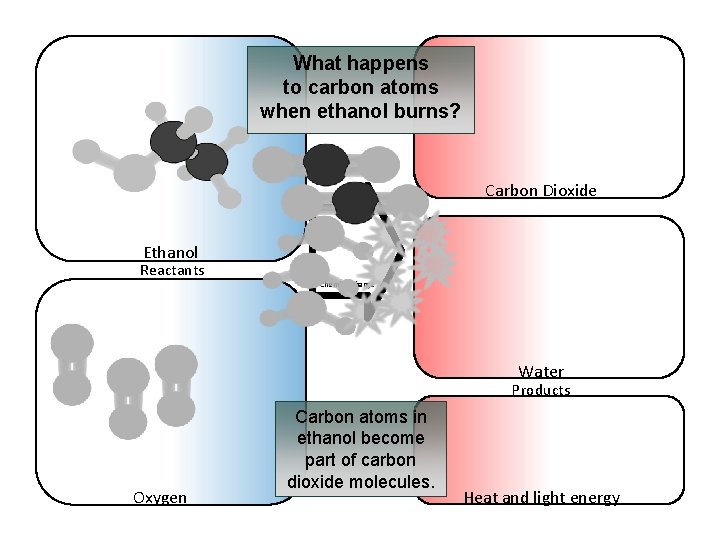
What happens to carbon atoms when ethanol burns? Carbon Dioxide Ethanol Reactants Chemical change Water Products Oxygen Carbon atoms in ethanol become part of carbon dioxide molecules. Heat and light energy
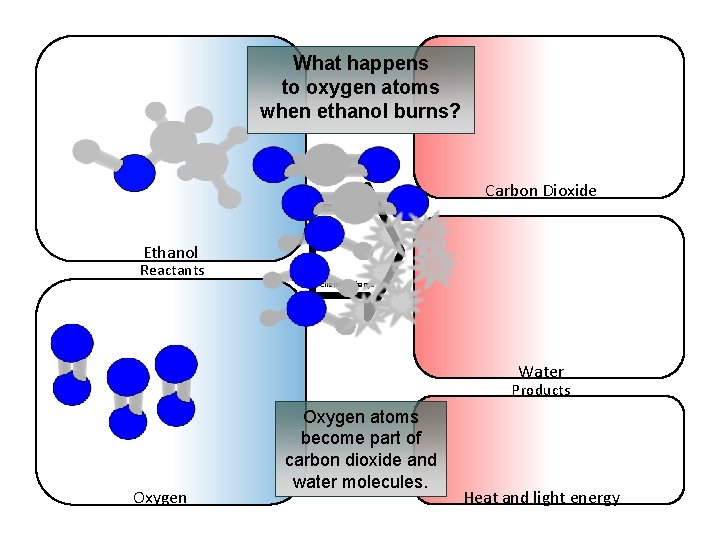
What happens to oxygen atoms when ethanol burns? Carbon Dioxide Ethanol Reactants Chemical change Water Products Oxygen atoms become part of carbon dioxide and water molecules. Heat and light energy
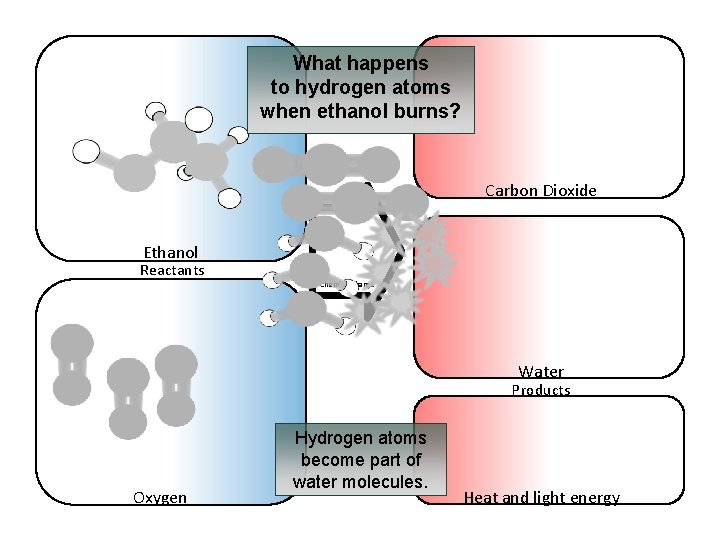
What happens to hydrogen atoms when ethanol burns? Carbon Dioxide Ethanol Reactants Chemical change Water Products Oxygen Hydrogen atoms become part of water molecules. Heat and light energy
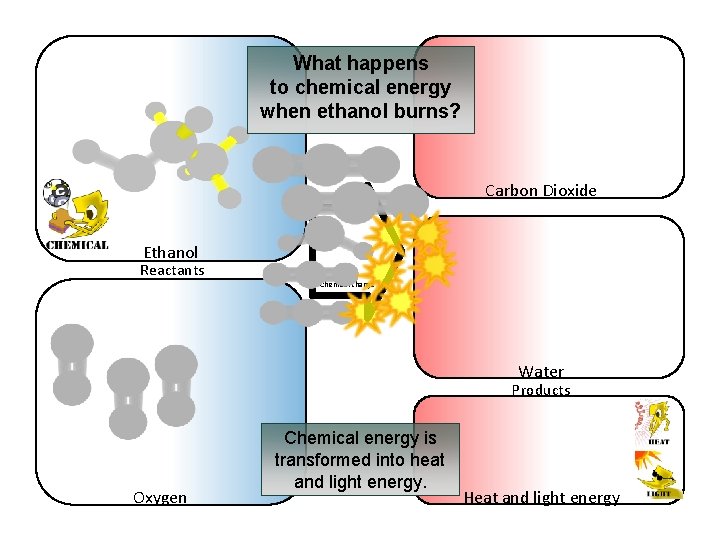
What happens to chemical energy when ethanol burns? Carbon Dioxide Ethanol Reactants Chemical change Water Products Oxygen Chemical energy is transformed into heat and light energy. Heat and light energy
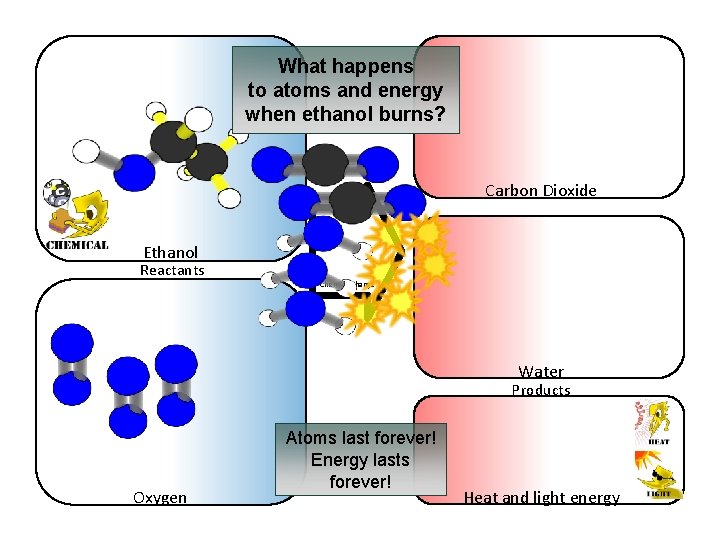
What happens to atoms and energy when ethanol burns? Carbon Dioxide Ethanol Reactants Chemical change Water Products Oxygen Atoms last forever! Energy lasts forever! Heat and light energy

Discuss with neighbors 1. Did the number and type of atoms stay the same at the beginning and end of the chemical change? ____ 2. Did the number of twist ties (representing energy) stay the same at the beginning and end of the chemical change? ____ 3. Why do the numbers of atoms and twist ties have to stay the same? 37

Review: Writing a Chemical Equation • Chemists use chemical equations to show atoms of reactant molecules are rearranged to make product molecules. • Writing the equation in symbols. Chemists use an arrow to show reactants change into products. • Equations must be balanced. Remember: Atoms last forever. • An example: The breakdown of hydrogen peroxide. 38

Group Challenge: Using the molecular models for the reactants, figure out how to write a balanced chemical equation for the ethanol burning reaction. Consider the products discussed on the Evidence Based Arguments worksheet. First group with the correct answer (and can show us) wins a prize! 39

Chemical equation for ethanol burning C 2 H 5 OH + 3 O 2 2 CO 2 + 3 H 2 O Write it in words: Submit when finished 40

DO NOW Friday 10/7 The ethanol burning reaction has a general name. You observed a chemical change involving the rapid reaction of a substance (fuel) with O 2 to produce heat and light. Using these context clues, can you name the chemical change that happens when ethanol burns? Submit Molecular Modeling for Ethanol Burning Worksheet 41

Prepare for Quiz (5 mins) • Revisit Unanswered Questions from your Ethanol Burning Worksheet. Can you answer them now? • Are there any questions left unanswered, regarding the burning of ethanol? Extra Credit Question – Period 1 Will put up in the last 5 minutes; Write both question and answer to earn points. These questions vary from class to class, so this is a must. Methane, CH 4, is a combustible fuel. Write the balanced chemical equation for this reaction. 42

Prepare for Quiz (5 mins) • Revisit Unanswered Questions from your Ethanol Burning Worksheet. Can you answer them now? • Are there any questions left unanswered, regarding the burning of ethanol? Extra Credit Question – Period 3 Will put up in the last 5 minutes; Write both question and answer to earn points. These questions vary from class to class, so this is a must. Butane, C 4 H 10, is a combustible fuel. Write the balanced chemical equation for this reaction. 43

Prepare for Quiz (5 mins) • Revisit Unanswered Questions from your Ethanol Burning Worksheet. Can you answer them now? • Are there any questions left unanswered, regarding the burning of ethanol? Extra Credit Question – Period 4 Will put up in the last 5 minutes; Write both question and answer to earn points. These questions vary from class to class, so this is a must. Propane, C 3 H 8, is a combustible fuel. Write the balanced chemical equation for this reaction. 44

Prepare for Quiz (5 mins) • Revisit Unanswered Questions from your Ethanol Burning Worksheet. Can you answer them now? • Are there any questions left unanswered, regarding the burning of ethanol? Extra Credit Question – Period 5 Will put up in the last 5 minutes; Write both question and answer to earn points. These questions vary from class to class, so this is a must. Butane, C 4 H 10, is a combustible fuel. Write the balanced chemical equation for this reaction. 45

Prepare for Quiz (5 mins) • Revisit Unanswered Questions from your Ethanol Burning Worksheet. Can you answer them now? • Are there any questions left unanswered, regarding the burning of ethanol? Extra Credit Question – Period 6 Will put up in the last 5 minutes; Write both question and answer to earn points. These questions vary from class to class, so this is a must. Methane, CH 4, is a combustible fuel. Write the balanced chemical equation for this reaction. 46

Carbon: Transformations in Matter and Energy Environmental Literacy Project Michigan State University Systems and Scale Unit Activity 5. 1 Molecular Models for Methane Burning
Unit Map You are here 48
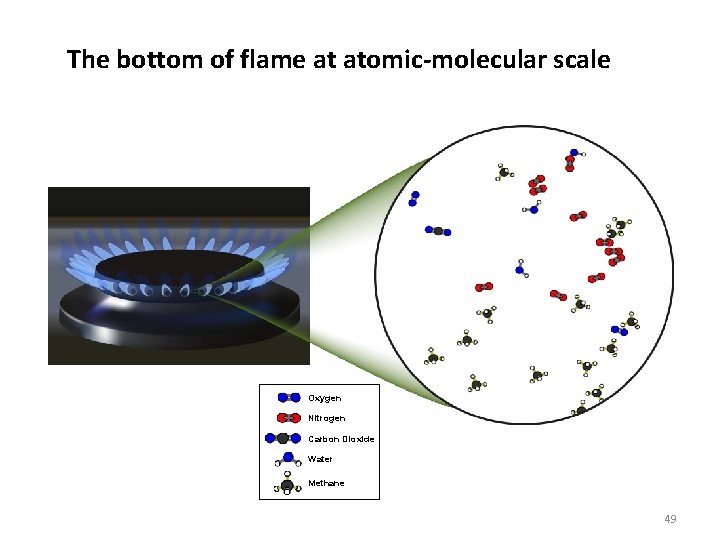
The bottom of flame at atomic-molecular scale Oxygen Nitrogen Carbon Dioxide Water Methane 49

The top of flame at atomic-molecular scale Oxygen Nitrogen Carbon Dioxide Water Methane 50
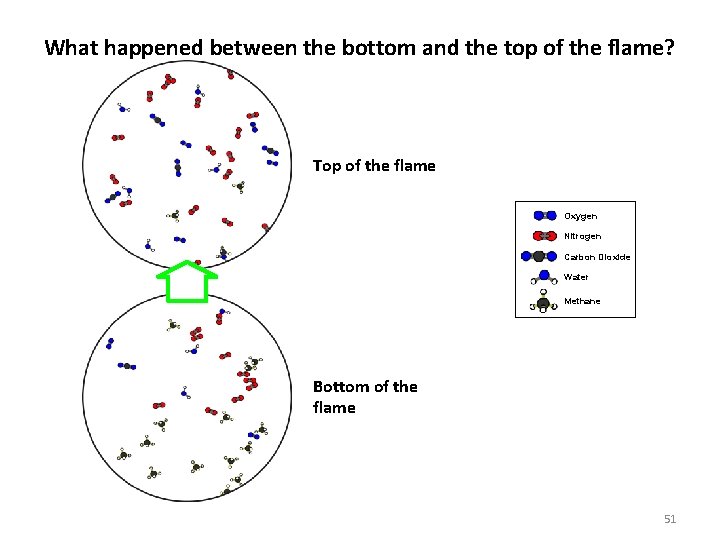
What happened between the bottom and the top of the flame? Top of the flame Oxygen Nitrogen Carbon Dioxide Water Methane Bottom of the flame 51

What’s the hidden chemical change when methane burns? ? Oxygen Carbon Dioxide Water Methane 52

The Matter Movement Question: Methane Where is matter moving? The Matter Change Question: How are the atoms in molecules being rearranged into different molecules? The Energy Question: What energy change is happening? Extra Challenge: Can you figure out the balanced chemical reaction for this combustion reaction? 53
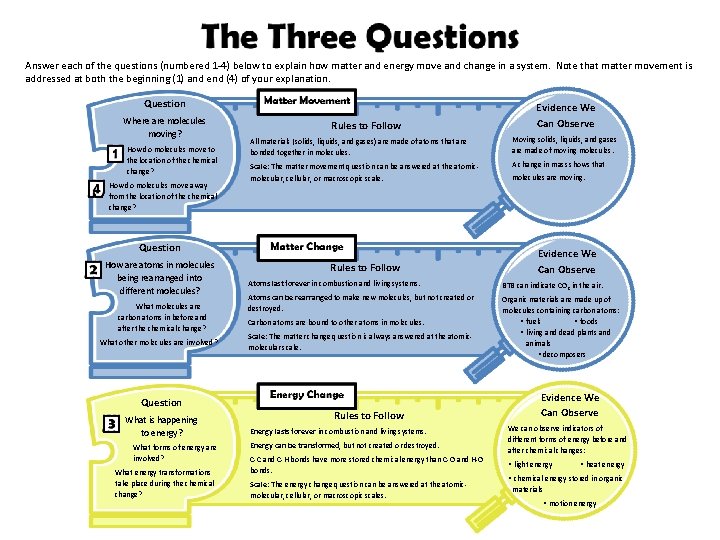
Answer each of the questions (numbered 1 -4) below to explain how matter and energy move and change in a system. Note that matter movement is addressed at both the beginning (1) and end (4) of your explanation. Question Where are molecules moving? How do molecules move to the location of the chemical change? How do molecules move away from the location of the chemical change? Rules to Follow All materials (solids, liquids, and gases) are made of atoms that are bonded together in molecules. Moving solids, liquids, and gases are made of moving molecules. Scale: The matter movement question can be answered at the atomicmolecular, cellular, or macroscopic scale. A change in mass shows that Question How are atoms in molecules being rearranged into different molecules? What molecules are carbon atoms in before and after the chemical change? What other molecules are involved? Question What is happening to energy? What forms of energy are involved? What energy transformations take place during the chemical change? Evidence We Can Observe Rules to Follow molecules are moving. Evidence We Can Observe Atoms last forever in combustion and living systems. BTB can indicate CO 2 in the air. Atoms can be rearranged to make new molecules, but not created or destroyed. Organic materials are made up of molecules containing carbon atoms: • fuels • foods • living and dead plants and animals • decomposers Carbon atoms are bound to other atoms in molecules. Scale: The matter change question is always answered at the atomicmolecular scale. Rules to Follow Energy lasts forever in combustion and living systems. Energy can be transformed, but not created or destroyed. C-C and C-H bonds have more stored chemical energy than C-O and H-O bonds. Scale: The energy change question can be answered at the atomicmolecular, cellular, or macroscopic scales. Evidence We Can Observe We can observe indicators of different forms of energy before and after chemical changes: • light energy • heat energy • chemical energy stored in organic materials • motion energy

Making the Reactant Molecules: Methane and Oxygen Remember the bonding rules: Carbon, Oxygen, Hydrogen 55
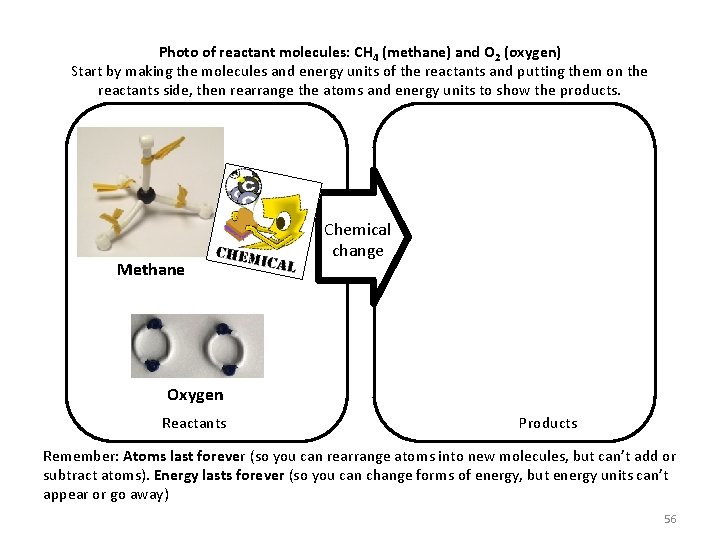
Photo of reactant molecules: CH 4 (methane) and O 2 (oxygen) Start by making the molecules and energy units of the reactants and putting them on the reactants side, then rearrange the atoms and energy units to show the products. Methane Chemical change Oxygen Reactants Products Remember: Atoms last forever (so you can rearrange atoms into new molecules, but can’t add or subtract atoms). Energy lasts forever (so you can change forms of energy, but energy units can’t appear or go away) 56

Important: When you are finished constructing the reactants, put all extra pieces away. 57

Rearranging the Atoms to Make Product Molecules: Carbon Dioxide and Water 58
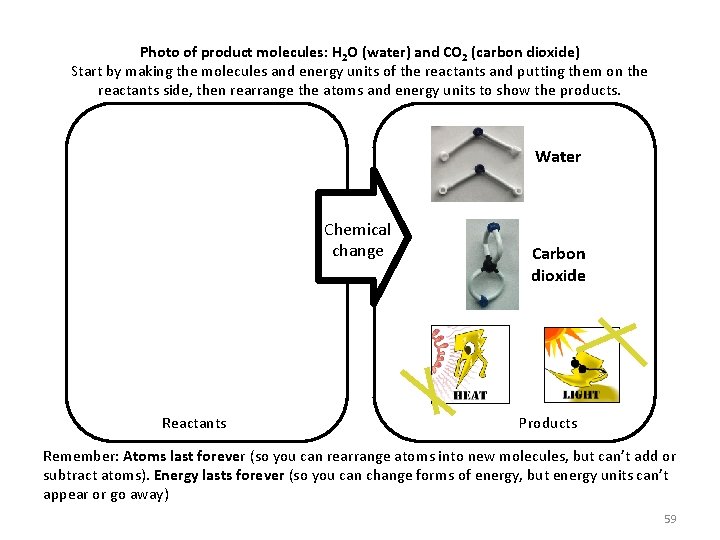
Photo of product molecules: H 2 O (water) and CO 2 (carbon dioxide) Start by making the molecules and energy units of the reactants and putting them on the reactants side, then rearrange the atoms and energy units to show the products. Water Chemical change Reactants Carbon dioxide Products Remember: Atoms last forever (so you can rearrange atoms into new molecules, but can’t add or subtract atoms). Energy lasts forever (so you can change forms of energy, but energy units can’t appear or go away) 59
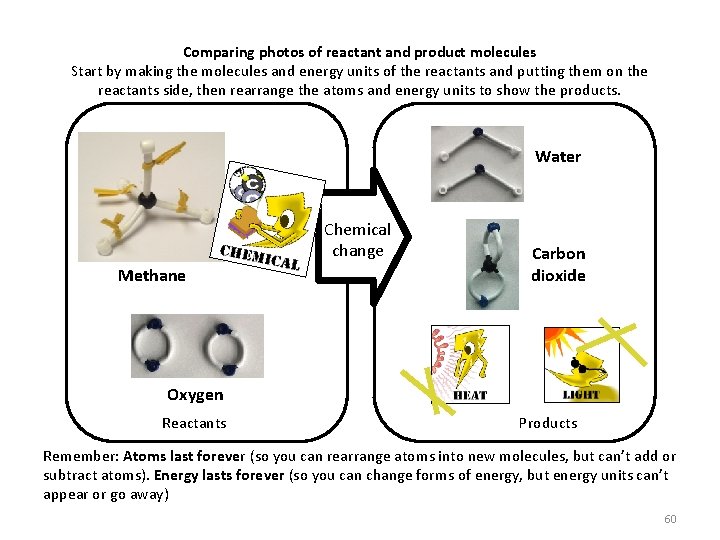
Comparing photos of reactant and product molecules Start by making the molecules and energy units of the reactants and putting them on the reactants side, then rearrange the atoms and energy units to show the products. Water Chemical change Methane Carbon dioxide Oxygen Reactants Products Remember: Atoms last forever (so you can rearrange atoms into new molecules, but can’t add or subtract atoms). Energy lasts forever (so you can change forms of energy, but energy units can’t appear or go away) 60
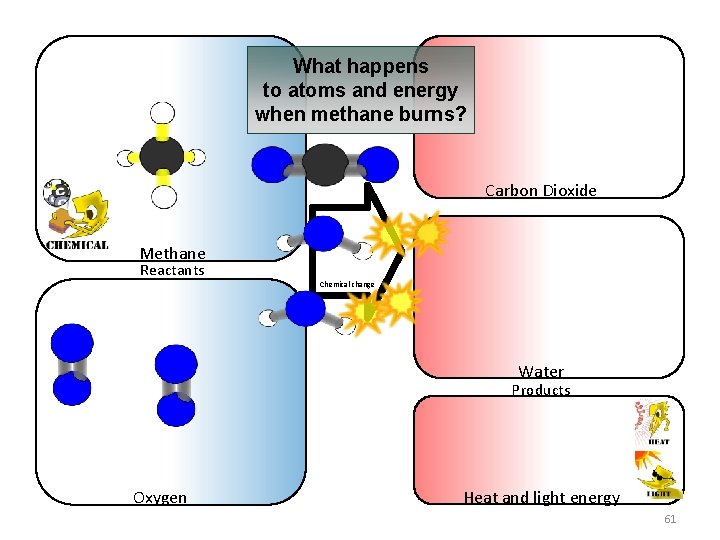
What happens to atoms and energy when methane burns? Carbon Dioxide Methane Reactants Chemical change Water Products Oxygen Heat and light energy 61
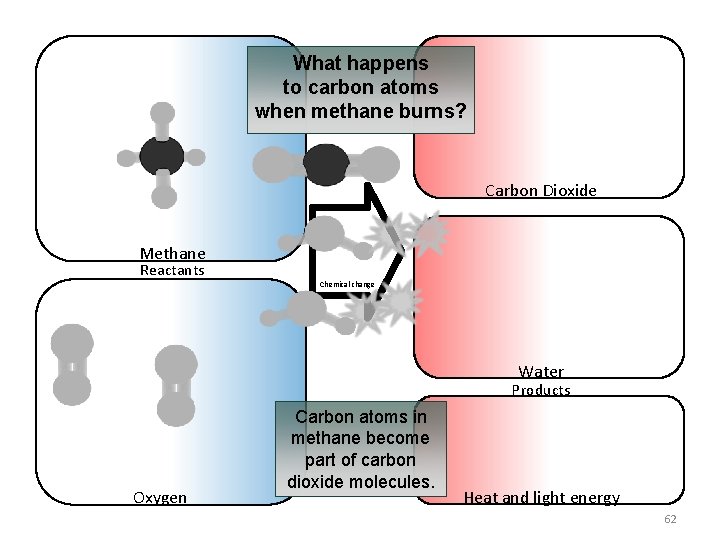
What happens to carbon atoms when methane burns? Carbon Dioxide Methane Reactants Chemical change Water Products Oxygen Carbon atoms in methane become part of carbon dioxide molecules. Heat and light energy 62
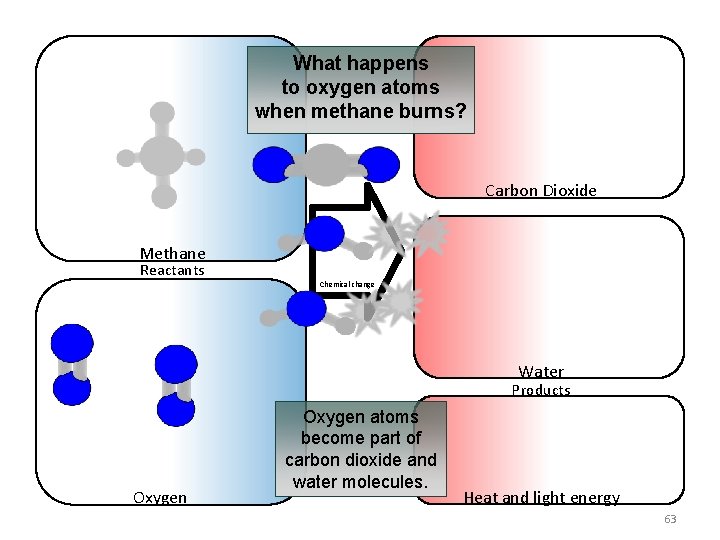
What happens to oxygen atoms when methane burns? Carbon Dioxide Methane Reactants Chemical change Water Products Oxygen atoms become part of carbon dioxide and water molecules. Heat and light energy 63
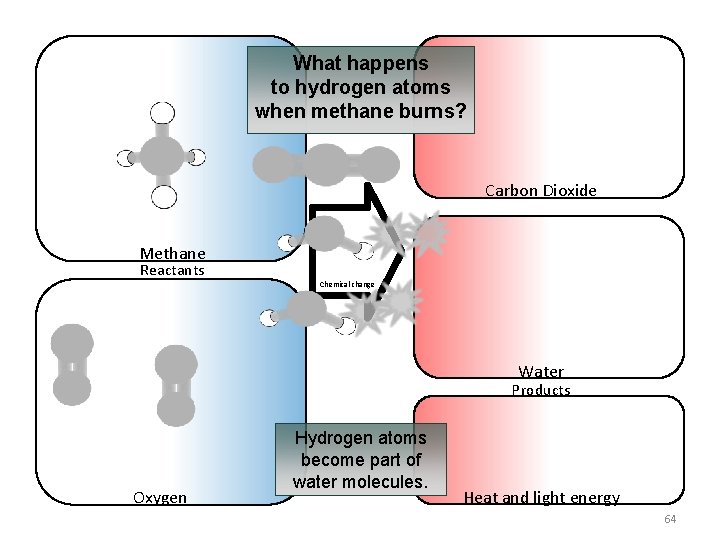
What happens to hydrogen atoms when methane burns? Carbon Dioxide Methane Reactants Chemical change Water Products Oxygen Hydrogen atoms become part of water molecules. Heat and light energy 64
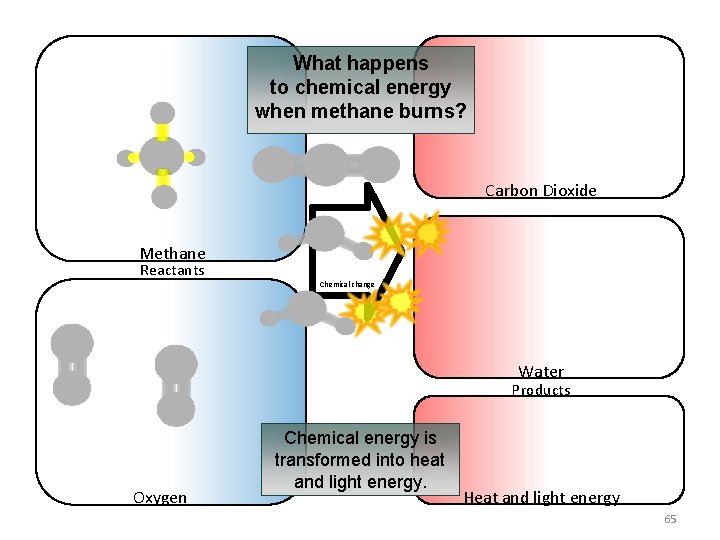
What happens to chemical energy when methane burns? Carbon Dioxide Methane Reactants Chemical change Water Products Oxygen Chemical energy is transformed into heat and light energy. Heat and light energy 65
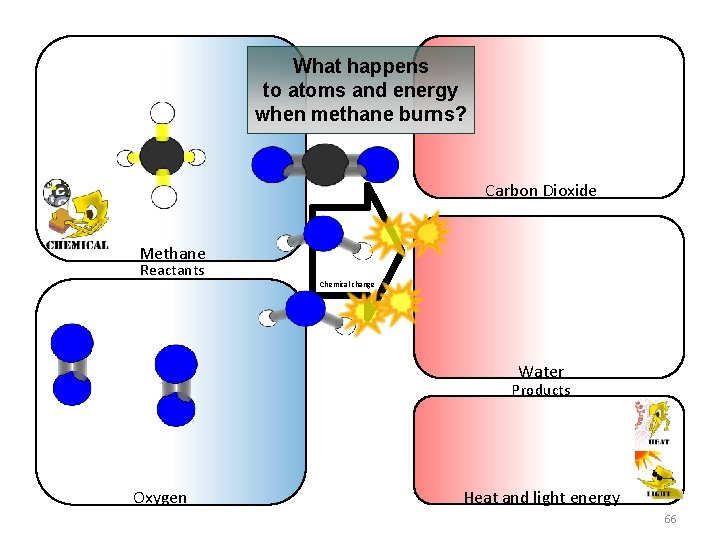
What happens to atoms and energy when methane burns? Carbon Dioxide Methane Reactants Chemical change Water Products Oxygen Heat and light energy 66

Review: Writing a Chemical Equation • Chemists use chemical equations to show atoms of reactant molecules are rearranged to make product molecules. • Writing the equation in symbols. Chemists use an arrow to show reactants change into products. • Equations must be balanced. Remember: Atoms last forever. • Try it: Write a balanced chemical equation for the burning of methane. 67

Chemical equation for methane burning CH 4 + 2 O 2 CO 2 + 2 H 2 O Write it in words: Methane reacts with oxygen to yield carbon dioxide and water 68
- Slides: 68