HAPPY FRIDAY TAKS OBJ 3 QUIZ NOTES MIXTURES

HAPPY FRIDAY! TAKS OBJ 3 QUIZ NOTES MIXTURES, SOLUTIONS, and SOLUBILITY

MATTER WHAT IS MATTER?

Atoms are matter • Carbon is an element • Elements are made of atoms • Diamonds are made of atoms of the element Carbon • Foil is made of the atoms of the element Aluminum.
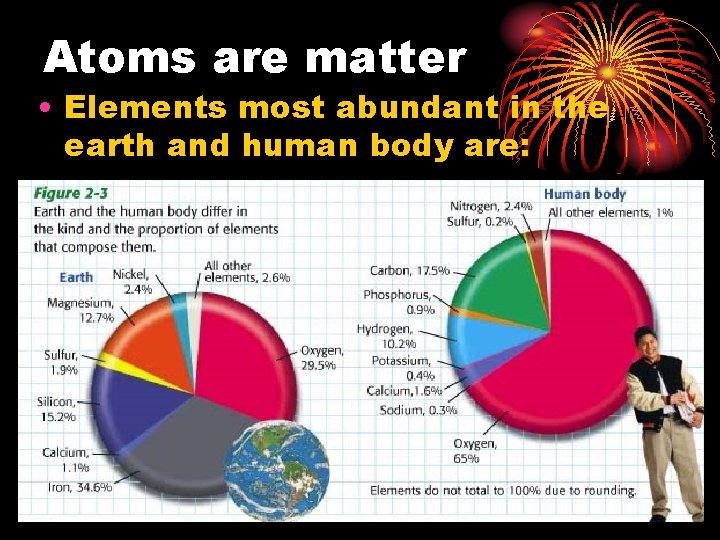
Atoms are matter • Elements most abundant in the earth and human body are:

Element • Pure substance that consists entirely of one type of atom • Hydrogen sulfur magnesium

COMPOUNDS • 2 or more elements combine to make a compound • Nylon is an example of a compound • Nylon contains atoms of Carbon, Hydrogen, Nitrogen, and Oxygen, but each strand actually contains hundreds of these units linked together.

Compounds… • Compounds are different from the elements in which they contain.

Molecules • Smallest unit of a substance that exhibits all of the properties characteristic of that substance • They act as a unit • Example: water H 2 O

Molecules • Most molecules are made of atoms of different elements, but some can be made of atoms of the same element.

Pure Substance • Any matter that has a fixed composition and definite properties • In Chemistry, a pure substance is MATTER WITH A FIXED COMPOSITION AND DEFINITE PROPERTIES

Pure Substance vs. Mixture • When people say “Pure Grape Juice” this means it contains only the juice of the grapes with NOTHING added or taken away • In reality, grape juice is NOT a pure substance. It is a MIXTURE!!!

Mixture • A combination of more than one substance • Grape juice is a mixture because it contains water, sugars, acids, and vitamins • The composition of grape juice is not fixed; it can have different amounts of water, sugars, or other compounds

Mixtures vs. Pure Substances • Almost all things we eat are mixtures • The air we breathe is a mixture of nitrogen and oxygen • Pure Substance: (ex: water) • Cannot be broken down by physical actions such as boiling, melting, or grinding

Pure substances blended together make mixtures • A mixture may have some properties similar to the pure substances that make it • Although you cannot see the different substances in grape juice, the mixture has chemical and physical properties in common with its components • Ex: fluid like water; sweet like sugar

Mixtures • There are 2 types of mixtures that we will deal with: • Heterogeneous mixture • Homogenous mixture

Heterogeneous Mixtures • Mixtures of this type are not the same throughout. • Example: Fruit Salad • Each spoonful of fruit salad would give you a different variety – no 2 spoonfuls would be exactly the same.

Homogenous Mixtures • These mixtures are uniform, in that no matter where you get your sample from in the mixture – each sample will be the exact same.

homogeneous heterogeneous

Mixtures classified by how thoroughly the substances mix Solids + Liquids examples to compare: Salt + water vs Flour + water

Mixtures classified by how thoroughly the substances mix • Flour doesn’t mix very well with water- it gives a cloudy white mixture The flour doesn’t dissolve in water This is an example of a heterogeneous mixture because the substances are not uniformly mixed.

Mixtures Salt + Water You cannot see the salt and the mixture is clear WHY? ? − SALT DISSOLVES IN WATER Even if you leave the mixture for a long time, the salt will not settle out. This is an example of a: homogenous mixture Mixing between the individual units and is the same throughout.

Obj. 1 cont… Pure or Mixture? !? ! 1. Kool-aid mixture 2. distilled water 3. tap water pure mixture 4. chocolate chip cookie 5. oxygen gas 6. atmosphere pure mixture 7. Glucose (C 6 H 12 O 6) pure
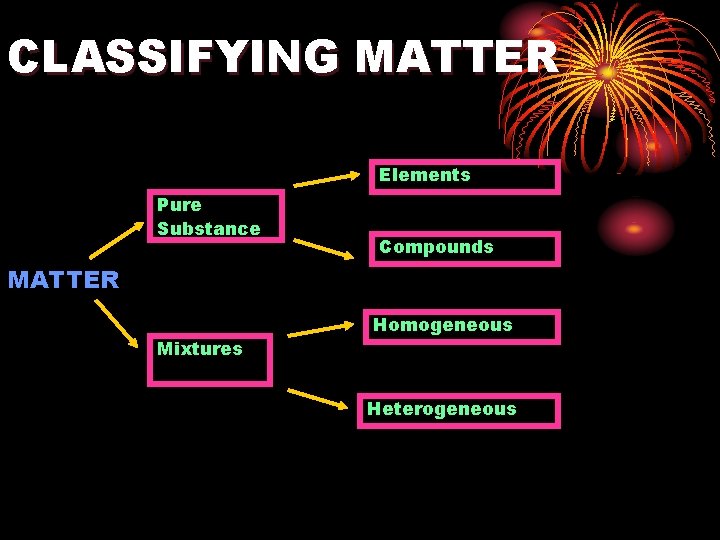
CLASSIFYING MATTER Elements Pure Substance Compounds MATTER Mixtures Homogeneous Heterogeneous

Solutions and Homogenous mixtures SOLUTIONS & HOMOGENOUS MIXTURES are synonymous. Homogenous mixtures are mixed completely, all the way down to their most fundamental particlesatoms, molecules, or ions.

Mixtures • Gasoline is a liquid mixture… • A homogenous mixture of at least 100 compounds in various quantities • The compounds within gasoline are miscible, thus gasoline looks like a pure substance even though it is not.

MISCIBLE Describes 2 or more liquids that are able to dissolve into each other in various proportions Ex: Water and Rubbing alcohol

IMMISCIBLE • If you shake a mixture of oil and water, the water will settle out after a while. • Oil and water form a heterogeneous mix • Oil and Water are immiscible thus you can see layers in the mixture.

IMMISCIBLE • 2 or more liquids that do NOT mix into each other

Gases can mix with liquids • Carbonated drinks are homogeneous mixtures. • They contain sugar, flavorings, and CO 2 gas, dissolved in water. • When carbonated drinks are manufactured, the CO 2 gas is mixed into the liquid under pressure and forms a solution.

Gases can mix with liquids • Liquids not under pressure can also contain dissolved gases. • If you let a glass of ice water sit out over night, you may see bubbles on the sides of the glass the next morning. The bubbles are some of the air that was dissolved in the cold water.

2. 1 Section Review On a separate sheet of notebook paper answer the following questions: 1: List the 2 types of pure substances 2: Complete the following: A heterogeneous mixture is to a homogeneous mixture as immiscible liquids are to _______. 3: Classify each as an element or mixture: Sulfur (S 8); Carbon Monoxide(CO); Methane (CH 4) 4: Give an example of a mixture and a pure substance.


Solutions and Other Mixtures (Ch 6 -1 and 6 -2) OBJECTIVES • Homogeneous vs. Heterogeneous mixtures • Compare and Contrast: solutions, colloids, suspensions • Identify ways to separate mixtures
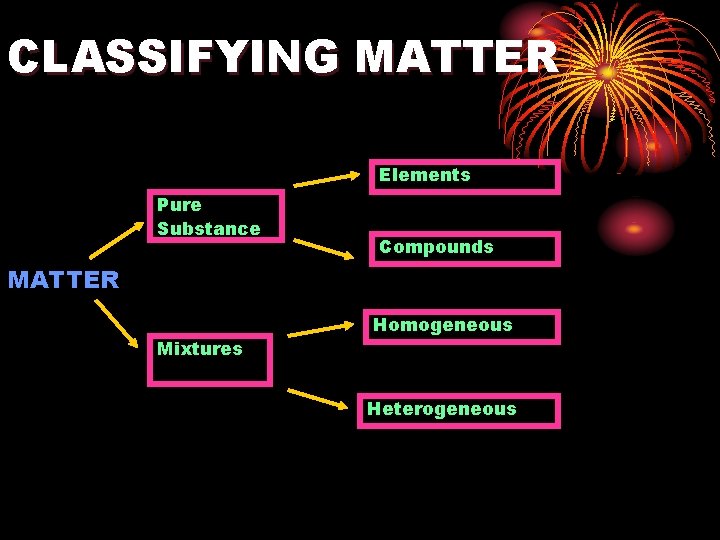
CLASSIFYING MATTER Elements Pure Substance Compounds MATTER Mixtures Homogeneous Heterogeneous

Solutions and Homogenous mixtures SOLUTIONS & HOMOGENOUS MIXTURES are synonymous. Homogenous mixtures are mixed completely, all the way down to their most fundamental particlesatoms, molecules, or ions.

Solutions and Other Mixtures • Heterogeneous Mixtures • Ex: Fruit Salad − NOT the same throughout − Quantity of each fruit varies with each spoonful

Heterogeneous Mixture

SUSPENSION • Have you ever forgotten to shake the OJ bottle before you poured a glass? • Probably tasted watery • Due to suspensions of orange pulp in OJ, which is mostly water

SUSPENSION a mixture that looks uniform when stirred or shaken that separates into different layers when it is no longer agitated

Suspension • SUSPENSIONS a mixture that looks uniform when stirred or shaken that separates into different layers when it is no longer agitated • OJ: • If carton is not shaken, then the pulp goes to the bottom and the watery liquid sits on top.

Suspension • Not all OJ has pulp • It can be separated out • Particles in suspensions are usually large enough they can be filtered out of mixture − (pulp stays behind)

Solution Suspension • All components are evenly distributed • dissolved • (salt in water) Mixture of water and nondissolved materials (flour in water)

COLLOID • Gelatin heterogeneous mix • Gelatin is a colloid • Colloid a mixture of very tiny particles of pure substances that are dispersed in another substance but do not settle out of the substance

Colloids • Differences between suspensions and colloids: • Particles in colloids are much smaller • Because they are so small, they do NOT separate out or settle to the bottom in colloids • Particles stay dispersed throughout the mixture

Colloids • EXAMPLES: • Egg white • Paint • Blood • Whipped cream (dispersing gas in a liquid) • Marshmallows (dispersed gas in a solid)

Heterogeneous liquid-liquid mixtures • Oil + vinegar 2 layers • They are immiscible • Oil floats on vinegar b/c less dense • To separate mixture: • Cup with spout • Cooks use this to separate fat from meat juices; fat is less dense thus stays behind in the cup

Some immiscible liquids can mix in EMULSIONS EMULSION: Any mixture of immiscible liquids in which the liquids are spread throughout one another

Emulsions • EXAMPLE: • Mayonnaise mixture of oil suspended in vinegar − They stay mixed (unlike that in salad dressing) because mayonnaise also contains egg yolk − Egg yolk coats the oil droplets keeping them from joining to form a separate layer − Thus, mayonnaise is an emulsion – (colloid where liquids normally do not mix are spread throughout each other)

Homogeneous Mixtures • Not only look uniform, they ARE uniform • Ex: Salt water • If you add salt to a glass of pure water and mix it, it will eventually look like pure water • Looks uniform because the components of the mixture are too small to be seen

Homogeneous Mixtures • When salt and water mix, no chemical reaction takes place • Easy to separate the 2 substances by evaporating or boiling the water • Once boiled, only left with salt

Solutions are Homogenous Mixtures • When you add aquarium salt to water and stir, the solid seems to disappear. • What is really happening? ?

Solutions are Homogenous Mixtures • The solid DISSOLVES in water to form a solution • In this example, the aquarium salt is the solute, and the water is the solvent.

Solute and Solvent… • A SOLUTE is the substance that DISSOLVES in a solution. • A SOLVENT is the substance that dissolves the solute to make a solution.

SOLUTIONS • When a solute completely dissolves in a solution, the dissolved particles are so small that you cannot see them • Solutes and solvents can be in any state of matter.

SOLUTIONS • Ex: • VINEGAR A solution of Acetic Acid, a liquid, dissolved in water, another liquid • A tank of air used by a scuba diver can be thought of as a solution of oxygen and several other gases.

Miscible Liquids mix to make solutions Water + Isopropanol rubbing alcohol (both liquids) Acetic Acid + Water solution Vinegar

Miscible Liquids • Since miscible liquids DO NOT separate into layers, they do not separate as easily as immiscible liquids do. • One way to separate miscible liquids is by distillation

Miscible Liquids • Distillation is the easiest way to separate two miscible liquids • Distillation can be used when wanting to separate water and methanol − Boiling points of the 2 are significantly different − BP WATER = 100 − BP Methanol = 67

Distillation 1. heat entire mixture until it boils 2. Liquid with lower boiling point would vaporize first (methanol @ 67 deg) 3. Some water would vaporize, but most would stay behind

Chromatography • If 2 miscible liquids have similar boiling points it can be even harder to separate by distillation • Chromatography can be used anytime it is too difficult to separate by distillation

Chromatography the science which studies the separation of molecules based on differences in their structure and/or composition. Chromatographic separations can be carried out using a variety of supports, including: immobilized silica on glass plates (thin layer chromatography) volatile gases (gas chromatography) paper (paper chromatography) and liquids which may incorporate hydrophilic insoluble molecules (liquid chromatography).

DISSOLVING AND SOLUBILITY

DISSOLVING AND SOLUBILITY

Dissolving Rate • Factors that affect dissolving rate of a solute in a solvent • Can you think of any on your own? ? ?

SURFACE AREA • Solutes with a larger surface area dissolve faster A substance in small pieces dissolves faster than the same substance in big pieces • EXAMPLE: − Loose sugar vs. Sugar cube − Chewable Vitamin C vs. pill swallowed

Shaking or Stirring If you pour sugar in a glass and let it sit without stirring, it will take longer for the sugar to dissolve completely --sugar sitting at the bottom of the glass is surrounded by dissolved sugar molecules – these molecules will slowly diffuse throughout the solution Until they spread through the entire solution the dissolved sugar keeps the water molecules from reaching the sugar crystals that haven’t dissolved yet.

Shaking or Stirring If you pour sugar in a glass and let it sit without stirring, it will take longer for the sugar to dissolve completely stirring or shaking the solution moves the dissolved sugar away from the sugar crystals. Now more water molecules can interact with the solid, so the sugar crystals dissolve faster.

TEMPERATURE • Sugar and other solutes dissolve faster in HOT water than in cold water. • Remember, that as a substance is heated, its particles move faster • As a result there are more collisions between particles and each collision transfers energy

TEMPERATURE • For gases, it is different… • Solubility is INVERSELY PROPORTIONAL to temperature for gases • The lower the temperature of the solvent, the higher the solubility • EX: Hot sodas will go flat!!

PRESSURE DOES NOT affect the solubility of solids!!! The solubility of a gas is affected by pressure… • Solubility is DIRECTLY PROPORTIONAL to pressure for gases − The HIGHER the pressure, the HIGHER the solubility − Think SCUBA divers and their gas

NOT EVERY SUBSTANCE WILL DISSOLVE • If a solute DISSOLVES in a solvent then it is said to be SOLUBLE • Example: Salt in Water • If a solute DOES NOT DISSOLVE in a solvent then it is said to be INSOLUBLE • Example: Olive Oil in water – They form two separate layers

Water • Most abundant compound in most living things!

WATER: A common Solvent of the Earth’s surface is water • Liquids we drink mostly water • ¾ of body weight is water • Many substances can dissolve in water therefore, WATER IS THE UNIVERAL SOLVENT!!! • 2/ 3

WATER • Water is the universal solvent • The structure of water helps it dissolve charged particles • Water is polar – uneven distribution of electrons
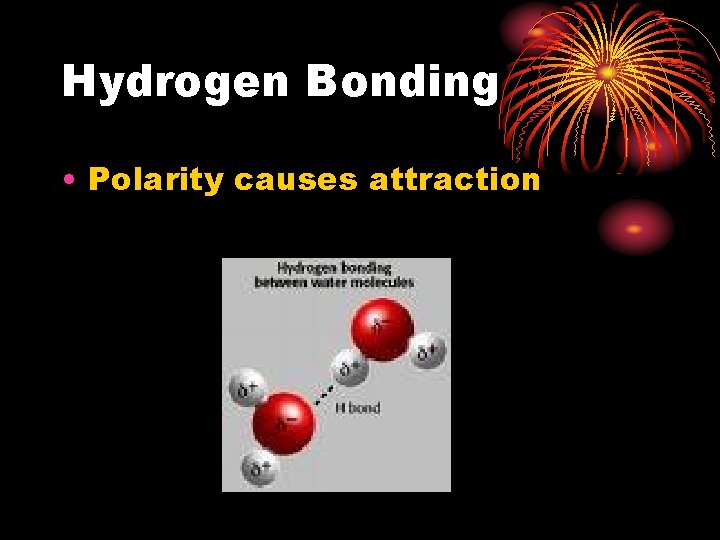
Hydrogen Bonding • Polarity causes attraction

WATER = THE Universal Solvent The figure below shows why Na. Cl easily dissolves in water… • Sodium ions are attracted to the partially negative oxygen atom, and Chloride ions are attracted to the partially positive Hydrogen atoms • Interactions between the ions and water molecules pull the ions away from the solid

Like Dissolves Like Water can dissolve MANY substances, but not all • EX: oil or gasoline doesn’t dissolve in water • Methanol is soluble in water because both liquids are polar − They have partially charged atoms that are attracted to one another • Gasoline: NOT SOLUBLE in water because its components are nonpolar Nonpolar = molecules do not have partial charges on opposite ends

CONCENTRATION Solutions can have different concentrations depending on how much solute and solvent are present

CONCENTRATION THE QUANTITY OF SOLUTE DISSOLVED IN A GIVEN QUANTITY OF SOLUTION

CONCENTRATION If a small quantity of solute is dissolved in a large volume of solvent, the resulting solution is said to be dilute A concentrated solution has a large quantity of solute dissolved in the solvent.

Saturated Solutions • Saturated Solution: contains the greatest quantity of solute that will dissolve in a given quantity of solvent • SOLUBILITY: the greatest quantity of a solute that will dissolve in a given quantity of solvent to produce a saturated solution

SOLUBILITY Solubility the maximum amount of a solute that can be dissolved in a given solvent at a certain temperature and pressure Try thinking of it like “solute capacity”, like the seating of a restaurant -solubility = seating capacity - solute = people - solvent = seats EVERY “restaurant” (or solvent) is different!!!

Saturated and Unsaturated Solutions Unsaturated Solution • a solution that is able to dissolve more solute Saturated Solution • A solution that cannot dissolve any more solute at the given conditions

SUPERSaturated Solutions • Supersaturated Solution: a solution holding more dissolved solute than is specified by its solubility at a given temperature • Supersaturated solutions are unstable systems because the solute’s solubility is exceeded for a short time.

SUPERSaturated Solutions Adding a single crystal of sodium causes the excess sodium acetate to quickly crystallize out of the solution until the solution is saturated at the cooler temperature

SUPERSaturated Solutions • 2 ways to bring a supersaturated solution back to a saturated solution… 1 heating up the solution to dissolve more solute 2 adding more solvent to the solution in order to dissolve the solute
- Slides: 86