Hantavirus Stephanie Bagley Eugene Khandros Nicholas Bevins History

Hantavirus Stephanie Bagley Eugene Khandros Nicholas Bevins

History • Hantaviruses are rodent-borne viruses which may be transmitted to humans in aerosolized urine, feces, or saliva, and occasionally by bite. • Hantaviruses often cause either hemorrhagic fever with renal syndrome (HFRS) or hantavirus pulmonary syndrome (HPS) • Other hantaviruses are not known to be human pathogens.

Hantavirus Outbreak in the US • HPS was first described in the United States in May 1993 during the investigation of a cluster of cases of acute adult respiratory distress in the Four Corners region. • HPS was found to be caused by a previously unknown hantavirus, Sin Nombre, detected in deer mice. • Sin Nombre caused approximately 200 confirmed cases of HPS during the outbreak, that led to a 50% mortality rate.

Hantavirus in the US • Prior to the HPS outbreak, the only known hantaviruses were those that caused HFRS • At least three other hantaviruses, New York Virus, Bayou Virus, and Black Creek Canal virus have since been confirmed to cause HPS in the U. S.

HPS in South America • The discovery of HPS in North America led to retrospective studies in South America. • More than 140 cases of HPS confirmed in Argentina. • The Andes hantavirus was discovered in long tailed pygmy rice rats in southern Argentina. • Andes Virus is the only known hantavirus to be transmitted person-to-person.

History of HFRS • While HPS has only been identified since 1993, HFRS has a much longer and complex history. • HFRS may have been recognized in China as early as 1000 years ago. • HFRS described in 1913 Russian records

History of HFRS Outbreaks of HFRS in the 1930’s: • Russia (1932) • Japanese troops in Manchuria (1934) • Sweden (1934)

Characterization of HFRS • 1934 -Japanese outbreak led to studies by Japanese physicians • 1940 - Japanese physicians compiled a clinical and pathological description of what was then called “epidemic hemorrhagic fever. ” • First to implicate mice in disease transmission. • Physicians injected filtrates of tissue from Apodemus agrarius carrying the hantavirus Hantaan into human subjects.

History of HFRS • 1939 -Russian studies also implicated mice in disease transmission. • 1961 - Moscow outbreak affecting 113 of 186 workers linked to rodent shipment

Characterization of HFRS 1967 -Russian scientists Yankovski and Povalishina provide more insight into disease. • Incubation period up to 6 weeks • Cycles of virus activity parallel population cycles of field mice • Transmission of disease occurred via inhalation of dust contaminated by rodent excretions.

Recent HFRS Cases • 1951 - outbreak in Korea during the war affected 3, 200 soldiers and resulted in a 7 -15% mortality rate. • Korean hemorrhagic fever (KHF) drew widespread international attention and was reclassified as HFRS in 1983 by the WHO. • 1983 -Several past outbreaks including KHF, “epidemic hemorrhagic fever” , Russian outbreaks all reclassified as HFRS by the WHO

Family Bunyaviridae 5 genera, 250 species Genus Human disease Bunyavirus La. Crosse encephalitis, others Phlebovirus Rift Valley fever, sandfly fever Nairovirus Crimean-Congo hemorrhagic fever Tospovirus Plant virus, no known human disease Hantavirus Hemorrhagic fever with renal syndrome Hantavirus pulmonary syndrome

Hantavirus Genus • Hantavirus Similarities – RNA viruses – Lipid membrane – Tri-segmented genome • Hantavirus Differences – Hantavirus transmitted through aerosolized rodent urine, feces and saliva. – Others genera transmitted through arthropod vectors.

Epidemiology and Rodent Hosts • Each strain of hantavirus has a specific rodent host • Hantavirus species appear to have coevolved with host rodent species • Rodents carrying hantavirus are asymptomatic
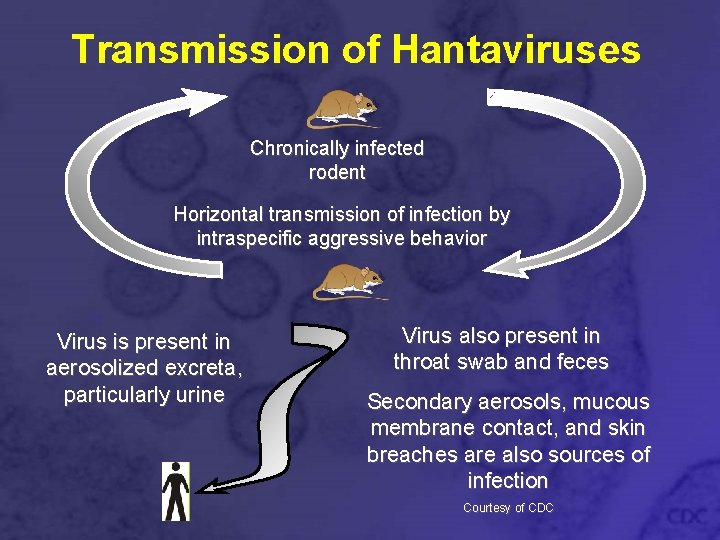
Transmission of Hantaviruses Chronically infected rodent Horizontal transmission of infection by intraspecific aggressive behavior Virus is present in aerosolized excreta, particularly urine Virus also present in throat swab and feces Secondary aerosols, mucous membrane contact, and skin breaches are also sources of infection Courtesy of CDC
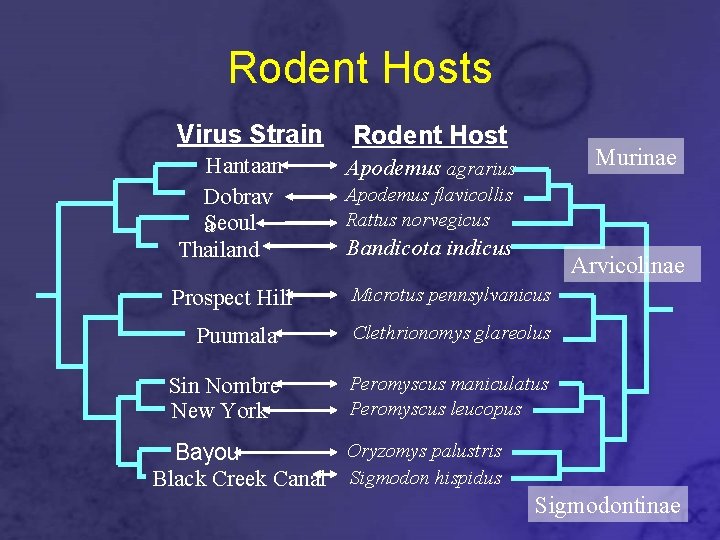
Rodent Hosts Virus Strain Rodent Host Murinae Hantaan Dobrav a. Seoul Thailand Bandicota indicus Prospect Hill Microtus pennsylvanicus Puumala Clethrionomys glareolus Sin Nombre New York Peromyscus maniculatus Peromyscus leucopus Apodemus agrarius Apodemus flavicollis Rattus norvegicus Arvicolinae Oryzomys palustris Bayou Black Creek Canal Sigmodon hispidus Sigmodontinae

New World Hantaviruses New York Sin Nombre Peromyscus leucopus Peromyscus maniculatus Prospect Hill Muleshoe Microtus pennsylvanicus Bloodland Lake Sigmodon hispidus Microtus ochrogaster Isla Vista Bayou Oryzomys palustris Black Creek Canal Microtus californicus El Moro Canyon Reithrodontomys megalotis Calabazo Zygodontomys Caño Delgadito brevicauda Choclo Sigmodon alstoni Oligoryzomys Rio Mamore fulvescens Oligoryzomys microtis Orán Oligoryzomys longicaudatus Bermejo Oligoryzomys chacoensis Andes Oligoryzomys longicaudatus Sigmodon hispidus Rio Segundo Reithrodontomys mexicanus Juquitiba Unknown Host Laguna Negra Calomys laucha Maciel Necromys benefactus Hu 39694 Unknown Host Lechiguanas Oligoryzomys flavescens Pergamino Akodon azarae Courtesy of CDC
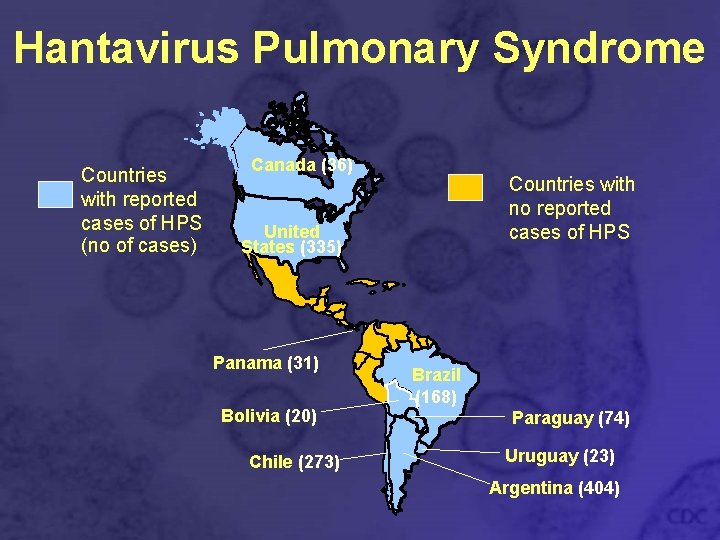
Hantavirus Pulmonary Syndrome Countries with reported cases of HPS (no of cases) Canada (36) Countries with no reported cases of HPS United States (335) Panama (31) Bolivia (20) Chile (273) Brazil (168) Paraguay (74) Uruguay (23) Argentina (404)

Rodent Hosts in the United States Deer mouse (Peromyscus maniculatus) Carrier of Sin Nombre strain, primary agent of HPS in the US. 250 -300 cases since discovery.

Rodent Hosts in the United States White-footed Mouse (Peromyscus leucopus) Carrier of New York strain.

Molecular Biology of Hantavirus • Physical Properties • Structure • Genetics • Replication Cycle • Pathogenesis

Molecular Biology of Hantavirus • Physical Properties • Structure • Genetics • Replication Cycle • Pathogenesis
Virion Properties • Spherical or oval-shaped. • 80 -120 nm diameter • Unique grid-like surface pattern, with 7 -8 nm projections • Lipid bilayer envelope • Granulofilamentous interior • Survive 12 hours at 4 C, high salt concentration and nonphysiological p. H. • Survives 1 -3 days after drying. • Exposure to lipid solvents and nonionic detergents destroys viral envelope
Molecular Biology of Hantavirus • Virion Properties • Structure • Genetics • Replication Cycle • Pathogenesis
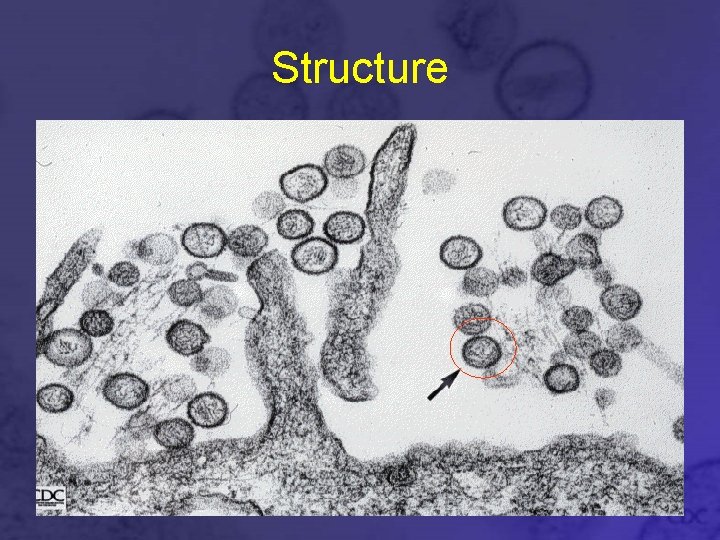
Structure
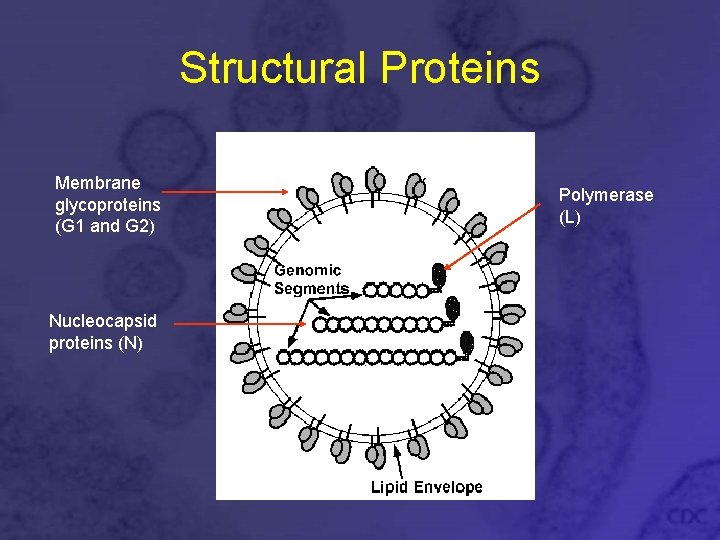
Structural Proteins Membrane glycoproteins (G 1 and G 2) Nucleocapsid proteins (N) Polymerase (L)

Membrane Glycoproteins • • G 1: 64 -67 k. Da G 2: 54 k. Da, highly conserved Integral membrane proteins G 1 -G 2 heterodimers form 8 nm projections on virion surface • Cysteine-rich • Contain asparagine-linked sugar groups • Important in cell entry and pathogenesis

Nucleocapsid Protein (N) • 48 k. DA • Complexes with genomic v. RNA in virus, as well as with c. RNA after infection, but not with m. RNA • Necessary for virus replication and packaging

Polymerase (L) • • • 247 k. DA RNA-dependent RNA polymerase (Rd. Rp) Complexed with ribonucleocapsids in virion Endonuclease activity to cleave host m. RNA Transcriptase activity for making c. RNA and m. RNA from v. RNA • Helicase activity to unwind v. RNA during transcription

Molecular Biology of Hantavirus • Physical Properties • Structure • Genetics • Replication Cycle • Pathogenesis

Genomic Organization • Tripartite negative sense genome • Small (S) segment, 1. 7 -2. 1 kb, codes for N nucleocapsid protein • Medium (M) segment, 3. 6 -3. 7 kb, codes for G 1 and G 2 glycoproteins • Large (L) segment, 6. 5 kb, codes for L polymerase protein
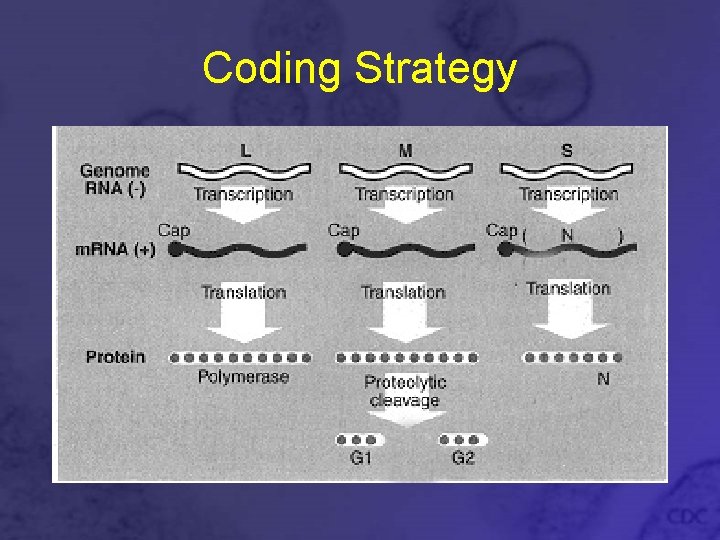
Coding Strategy
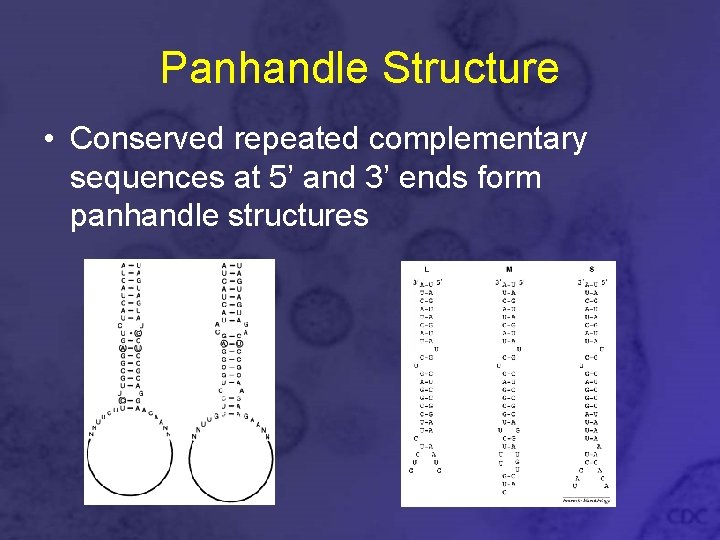
Panhandle Structure • Conserved repeated complementary sequences at 5’ and 3’ ends form panhandle structures

Transcription • Viral polymerase transcribes negative-strand v. RNA to m. RNA • Polymerase acts as a endonuclease and cleaves host m. RNAs 7 -18 nt from the 5’ cap • The capped oligonucleotides act as primers required to initiate transcription • After transcription is primed and the first repeat of the terminal sequence is transcribed, polymerase slips and realigns the nascent RNA, then continues transcription

Replication • Viral polymerase transcribes negativestrand v. RNA to sense c. RNA • c. RNA is used as template to make more negative-strand v. RNA • ppp. G is used to prime c. RNA and v. RNA synthesis • Same “prime and realign” strategy
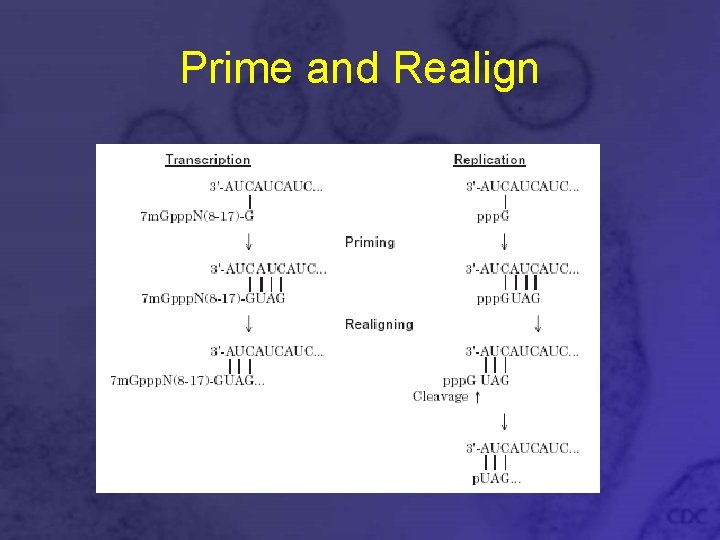
Prime and Realign
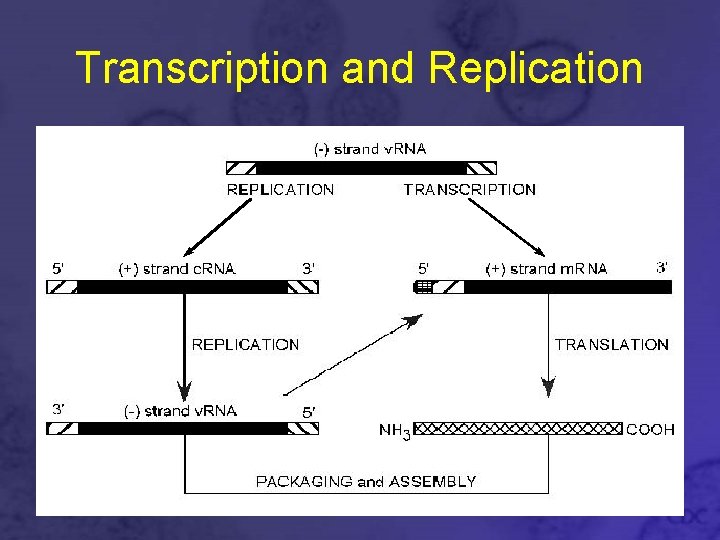
Transcription and Replication

Molecular Biology of Hantavirus • Physical Properties • Structure • Genetics • Replication Cycle • Pathogenesis
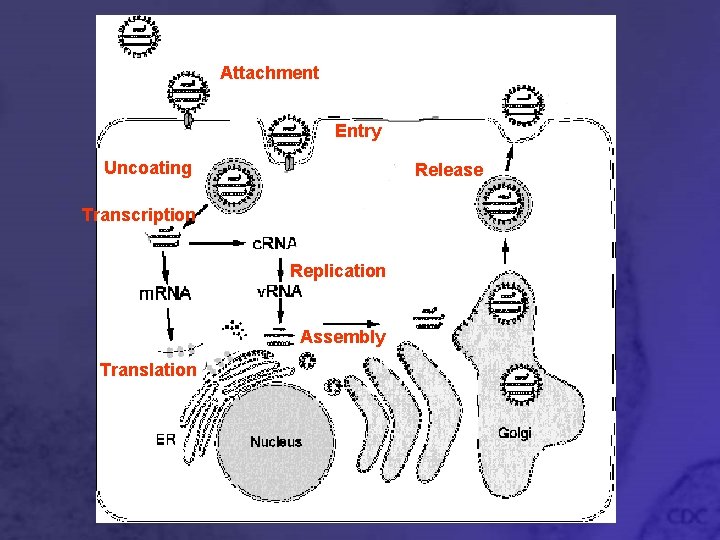
Attachment Entry Uncoating Release Transcription Replication Assembly Translation

Attachment • Viral G 1 and G 2 glycoproteins interact with cell surface receptors • Pathogenic hantaviruses bind 3 integrins • Non-pathogenic hantaviruses bind 1 receptors

Entry and Uncoating • Virus particles bound to integrin receptors are taken in by receptor mediated endocytosis • Newly formed vesicles are acidified • Acidic environment changes conformation of G 1 and G 2 • Viral and cell membrane fuse • Genomic material and polymerase are released into cytoplasm

Entry

Primary Transcription • Transcription of negative sense v. RNA to m. RNA • Viral polymerase (Rd. Rp) transcribes nucleoprotein-coated v. RNA • Capped oligonucleotides from cell’s own m. RNA are used to prime transcription • Follows “prime and realign” model

Translation • L and S segment m. RNA is translated on free ribosomes in cytoplasm • M segment m. RNA translated on ER-bound ribosomes • G 1 and G 2 peptides produced from M m. RNA are cleaved cotranslationally • Separate signal sequences for G 1 and G 2 cause ER attachment and embed the peptides in ER membrane (Signal Hypothesis)
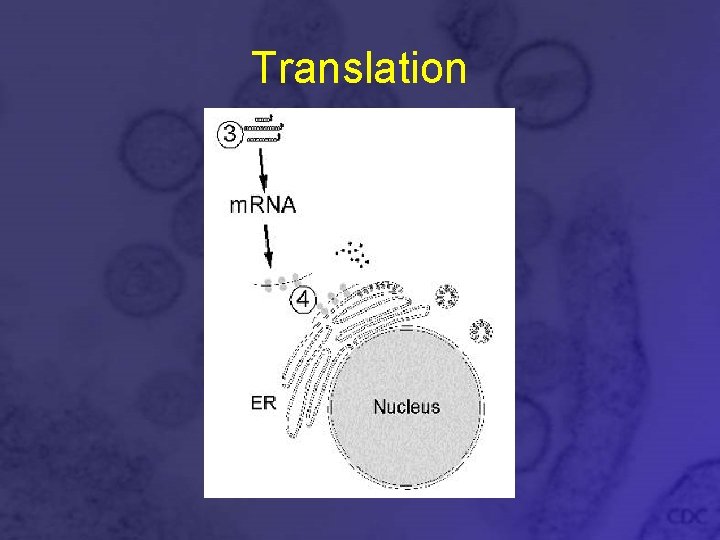
Translation

Genome Replication • v. RNA is used as a template by viral polymerase to make sense strand c. RNA • c. RNA is used as a template to make more negative strand v. RNA • More genetic material means more virions produced

Secondary Transcription • Extra v. RNA synthesized during replication is used as template to make m. RNA • Since more template is present after v. RNA is replicated, more m. RNA can be transcribed, and more viral proteins can be made
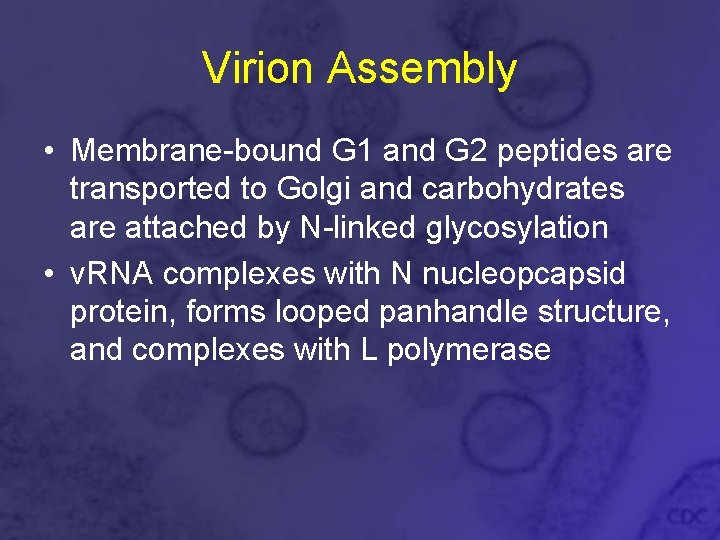
Virion Assembly • Membrane-bound G 1 and G 2 peptides are transported to Golgi and carbohydrates are attached by N-linked glycosylation • v. RNA complexes with N nucleopcapsid protein, forms looped panhandle structure, and complexes with L polymerase
Virion Assembly

Virion Release – Scenario 1 • Nucleocapsid complexes bud into the Golgi membrane with G 1 and G 2 embedded • Virion particle is formed inside the Golgi • Virions are transported to cell membrane by vesicles and released by exocytosis, just like in secretion • Viruses may prefer different cell surfaces for release
Virion Release – Scenario 2 • Sin Nombre and Black Creek Canal viruses • G 1 and G 2 embedded into cell membrane through Golgi vesicles • Virions bud from cell membrane, not through Golgi

Molecular Biology of Hantavirus • Physical Properties • Structure • Genetics • Replication Cycle • Pathogenesis

Hantavirus and Host Cells • Virus replication typically halts host macromolecule synthesis • Hantavirus replication does not affect host cell’s natural functions • Hantavirus release does not require host cell lysis • Hantavirus is able to establish a persistent infection in rodent host cells

Integrins • Heterodimeric receptors composed of α and β subunits • Present on endothelial cells, macrophages, and platelets – cells affected by Hantavirus infection • Normally involved in regulation of endothelial cell adhesion, platelet aggregation, Ca++ channel activation, and extracellular matrix interactions, including cell migration

β 3 -Integrins • Required for infection by pathogenic Hantaviruses • β 1 integrins are used by non-pathogenic strains • Attachment of G 1/G 2 proteins of viroid to integrin initiates endocytosis, but also activates the receptor • Variation in virus G 1/G 2 protein may account for severity of disease

Hantavirus Infection Pathogenesis • Binding of Hantavirus glycoproteins to β 3 integrin causes disruption of vascular integrity • Capillaries become more permeable • Arteriole vasoconstriction and vasodilation are disrupted • Binding to platelet receptors affects clotting and platelet function
Immune Reaction • Immune system activated against Hantavirus epitopes • Virus epitopes expressed on surface of host cells triggers cytotoxic T-cell attack on host tissues • Symptoms are consistent with inflammatory response

Laboratory Diagnosis of Hantavirus • Hantavirus is difficult to culture, so morphological identification is difficult • RT-PCR using primers for conserved genome regions allows confirmation of infection • PCR product can be sequenced and compared to known viral sequence database for species identification

Clinical Presentation of Hantavirus Infection Three different clinical manifestations of hantavirus infection caused by different viral strains Hemorrhagic fever with renal syndrome (HFRS) • Found in Europe and Asia Nephropathia Epidemica (NE) • Found in Europe Hantavirus pulmonary syndrome (HPS) • Found in north and south America

HFRS • A group of clinically similar diseases that occur throughout Europe and Asia • Includes several diseases that formerly had other names, including Korean hemorrhagic fever, epidemic hemorrhagic fever and nephropathia epidemica • ~15% fatality

Stages of Hemorrhagic Fever with Renal Syndrome (HFRS) 1)Incubation (4 -40 days) 2)Febrile Phase (3 -5 days) 3)Hypotensive Phase (hours to days) 4)Oliguric Phase (3 -7 days) Recovery: 5)Diuretic Phase (2 -21 days) 6)Convalescent Phase (2 -3 months)
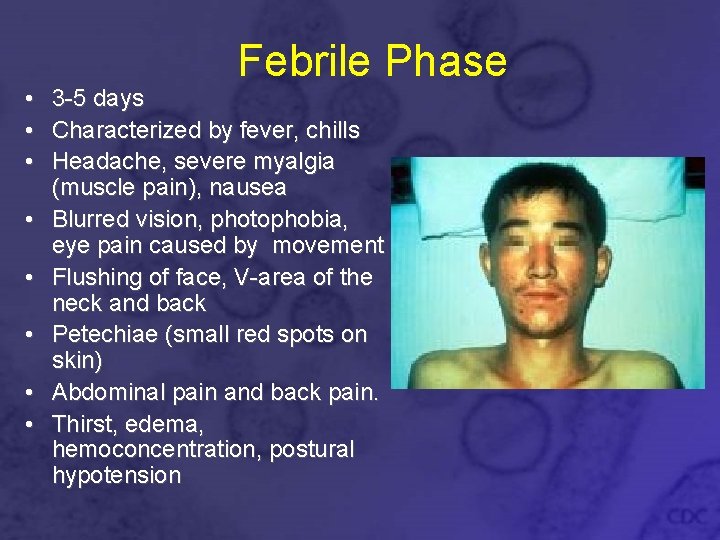
Febrile Phase • 3 -5 days • Characterized by fever, chills • Headache, severe myalgia (muscle pain), nausea • Blurred vision, photophobia, eye pain caused by movement • Flushing of face, V-area of the neck and back • Petechiae (small red spots on skin) • Abdominal pain and back pain. • Thirst, edema, hemoconcentration, postural hypotension

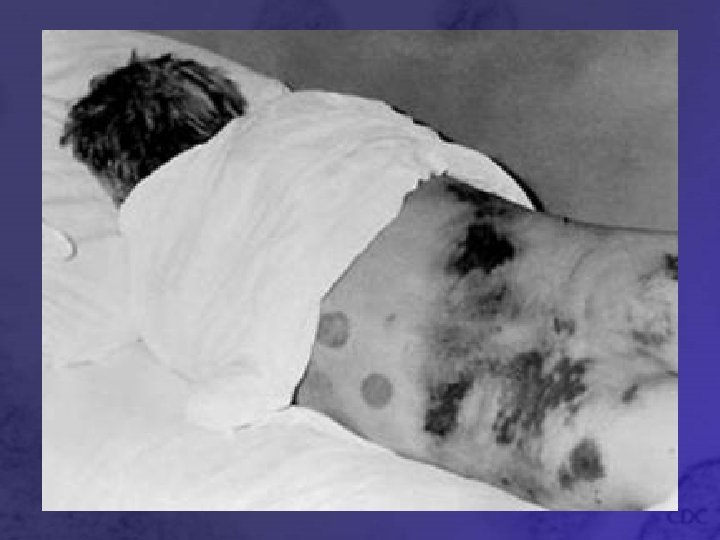
Hypotensive phase • Hours to days • Blood pressure decrease, hypovolemia (decreased blood volume), shock • Worsening of bleeding manifestations: petechiae, epistaxis (nosebleed), gastrointestinal and intracranial bleeding • Levels of urea and creatinine in blood rise, proteinuria (excessive protein in urine) • Leukocytosis, thrombocytopenia (decreased # of platelets)

Oliguric Phase • 3 -7 days • Marked by decreased urine production due to renal (kidney) dysfunction • Hypervolemia (high blood volume) leading to hypertension • Blood electrolyte imbalance • Continuation of hemorrhagic symptoms • Severe complications: cardiac failure pulmonary edema (swelling of lungs), and cerebral bleeding

Diuretic Phase • Several days to several weeks • Beginning of recovery • 3 -6 liters of urine/ day; return to normal renal activity • Anorexia, fatigue due to dehydration
Convalescent Phase • 2 -3 months • Progressive improvement in glomerular filtration, renal blood flow, and urine concentrating ability

Clinical Testing for HFRS • Thrombocytopenia (low platelet count) is a signifier • Urine tests for albuminuria (abnormally high amounts of the plasma protein albumin in the urine) • Urine tests for microhematuria (microscopic amounts of blood in the urine)

Problems Diagnosing HFRS • Early symptoms resemble influenza • More serious symptoms of hypotensive phase have acute onset

Nephropathia Epidemica (NE) • Puumala hantavirus strain • Common mild form of HFRS in Europe • Similar sequence of symptoms as HFRS, but much milder • Only 6% of serologically confirmed cases require hospitalization

HPS • 1993 four corners outbreak • Cases found in almost all of the Americas • ~50% fatality
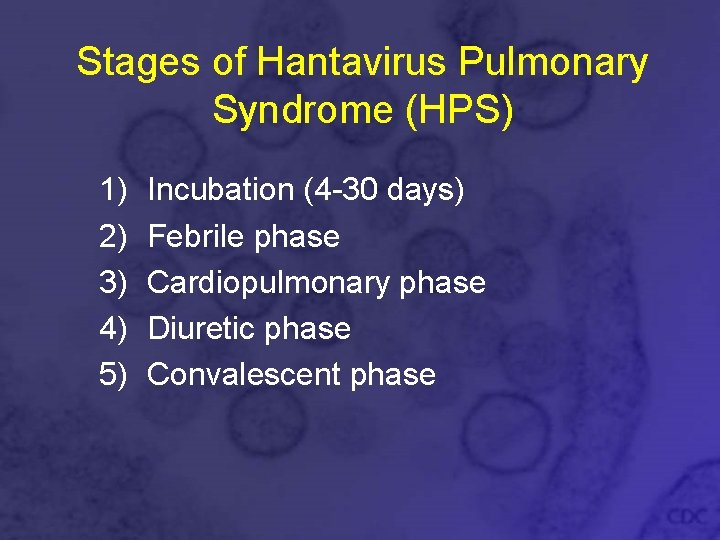
Stages of Hantavirus Pulmonary Syndrome (HPS) 1) 2) 3) 4) 5) Incubation (4 -30 days) Febrile phase Cardiopulmonary phase Diuretic phase Convalescent phase
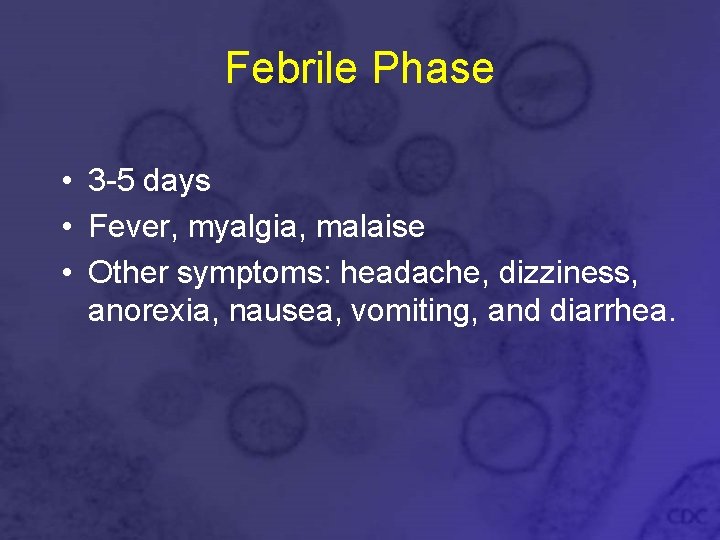
Febrile Phase • 3 -5 days • Fever, myalgia, malaise • Other symptoms: headache, dizziness, anorexia, nausea, vomiting, and diarrhea.

Cardiopulmonary Phase • 4 -24 hours • Presentation and rapid progression of shock and pulmonary edema (4 -24 h non-productive cough and tachypnea (shortness of breath) • Hypovolemia due to progressive leakage of high protein fluid from blood to lung interstitium and alveoli • Hypotension and oliguria • Thrombocytopenia (often present in febrile phase as well) • Death within 24 -48 hours due to hypoxia (lack of oxygen) and/or myocardial failure

Diuretic Phase • • • Several days to several weeks Beginning of recovery Rapid clearance of pulmonary edema Resolution of fever and shock Anorexia, fatigue due to dehydration
Convalescent Phase • Up to 2 months • Results in chronic decreased small-airway volume and diminished alveolar diffusing capacity

Clinical Testing for HPS • Many lab tests and radiographs appear normal • Serological tests more effective • ELISA Ig. M capture assay, using either SNV, Laguna Negra, or Andes antigens are used in all countries that have previously detected cases • Immunofluorescent test for the presence of antibodies • Blood analysis also may find thrombocytopenia with platelet count less than 150, 000 mm in 98% of cases

Problems Diagnosing HPS • Symptoms often confused with influenza • Common signs of upper respiratory disease such as sore throat, sinusitis, and ear pain not usually present • Abdominal pain often misinterpreted as appendicitis • Many doctors outside endemic regions fail to recognize or have sufficient testing
Treatment of Hantavirus Infection • General care, alleviation of symptoms • Ribavirin (HFRS) • ECMO (HPS)
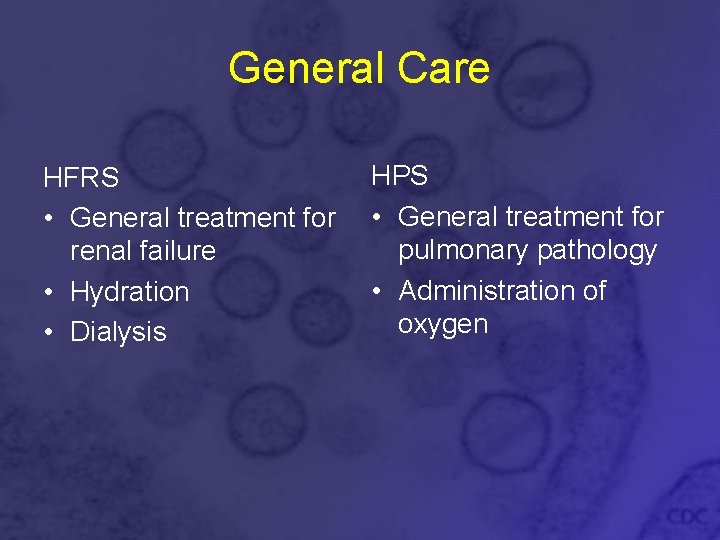
General Care HFRS • General treatment for renal failure • Hydration • Dialysis HPS • General treatment for pulmonary pathology • Administration of oxygen

Extra Corporeal Membrane Oxygenation • Removes blood from the body and artificially removes CO 2 and adds O 2 • Costly • Difficult
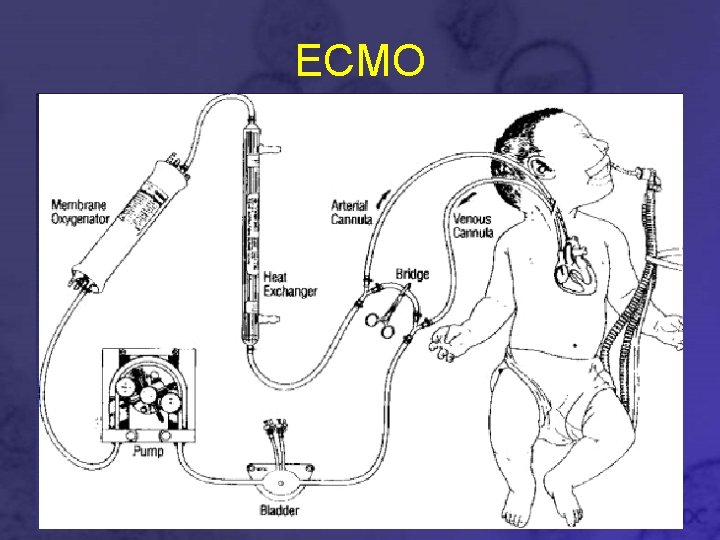
ECMO

Ribavirin • Administered intravenously • Shown to be effective against HFRS causing strains • Not shown to be effective against HPS causing strains

Terrorism

Terrorism A policy intended to strike with terror those against whom it is adopted; the employment of methods of intimidation; the fact of terrorizing or condition of being terrorized.

One man’s terrorist is another man’s freedom fighter.

Considerations of Pathogens for Use in Bioterrorism • • • Health effects Epidemiology Cost effectiveness Psychological effects Economic impact

CDC classifications Category A • Highest priority organisms that can be easily disseminated or transmitted form person to person, results in high mortality rates and have the potential for major public health impact, might cause public panic and social disruption, and require special action for public health preparedness.

CDC Classifications Category A • Anthrax (bacillus anthracis) • Botulism (clostridium botulinum) • Plague (Yersinia pestis) • Smallpox (variola major) • Tularemia (Francisella tularensis) • Viral hemorrhagic fevers

CDC Classifications Category B • Second highest priority organisms that are moderately easy to disseminate, results in moderate mortality rates, and require specific enhancements of CDC's diagnostic capacity and enhanced disease surveillance.

CDC Classifications Category B • Typhus fever • Viral encephalitis • Ricin toxin • Food and water safety threats

CDC Classifications Category C • Third highest priority organisms that include emerging pathogens that could be engineered for mass dissemination in the future because of availability, ease of production and dissemination, and potential for high morbidity and mortality rates and major health impact.

CDC Classifications Category C • Nipah Virus • Crimean-Congo Hemorrhagic Fever virus • Yellow fever • Multi-drug resistant TB • Influenza • Rabies

CDC Classifications • HFRS causing strains are Category A because of high infectivity and morbidity • HPS causing strains are Category C because of low infectivity

Considerations of Pathogens for Use in Bioterrorism • Health effects • Epidemiology • Cost effectiveness • Psychological effects • Economic impact
Health Effects • High lethality • No or ineffective treatment

Health Effects Hemorrhagic Fever with Renal Syndrome • Medium lethality • Dramatic visual change in patients (psychological) • Some success with antiviral treatments Hantavirus Pulmonary Syndrome • High Lethality • No effective treatment

Considerations of Pathogens for Use in Bioterrorism • Health effects • Epidemiology • Cost effectiveness • Psychological effects • Economic impact

Epidemiology • Medium incubation time in order to cause secondary infections • Rodent vector • Spread through aerosol • HFRS causing strains known to transmit human to human • Suspected human to human transmission of HPS causing strain in Argentina (Andes Virus)

Andes Virus • 1996 outbreak in rural Argentina • Spread to people whose only contact was a car ride • Spread to several doctors caring for HPS patients • Low rodent population in Argentina at the time (early spring)

Aerosolization • Virus only infectious for 1 -3 days outside of a host because of a weak lipid envelope • The number of particles needed to cause a human infection is not known

Considerations of Pathogens for Use in Bioterrorism • Health effects • Epidemiology • Cost effectiveness • Psychological effects • Economic impact

Cost Effectiveness • What resources would be required to successfully disseminate Hantavirus into a large population?

Cost Effectiveness • Preparation of virus using cell culture (difficult, need training and equipment) • Aerosolization? • Contaminated water? • Creation of infected rodent population?

Considerations of Pathogens for Use in Bioterrorism • Health effects • Epidemiology • Cost effectiveness • Psychological effects • Economic impact

Psychological Effects • HFRS causes dramatic visual effects in patients • HPS would be especially difficult to diagnose outside of its normal range • Media coverage ‘super flu’ ‘hemorrhagic fever’
Mouse Virus in City! Everyone will Die! “Nothing to Fear” say Liberal ‘Doctors’

Considerations of Pathogens for Use in Bioterrorism • Health effects • Epidemiology • Cost effectiveness • Psychological effects • Economic impact

Economic Impact What effect would an outbreak of hantavirus have on the economy? • Treatment • Disruption of work • Hysteria • Even a small outbreak could cause a large disruption

Why Hantavirus Would Be a Good Terrorist Weapon • There is no cure for HPS infection • Fairly long incubation period between infection and onset of symptoms • Difficult diagnosis of HPS • High lethality of HPS

Why Hantavirus Would Not Be a Good Terrorist Weapon • Low infectivity of HPS • Difficult production • Not stable

Prevention • Vaccines • Hygiene
Vaccines • E. Coli expressed truncated nucleocapsid as an immunogen • Naked DNA • Recombinant non-pathogenic virus • Rodent brain-derived • Cell culture derived • Inactivated virus

Hygiene • Prevent aerosolization of virus from rodent excrement • Dampen surfaces with detergent before cleaning • General hygiene

“Weapons of mass destruction, including evil chemistry and evil biology, are all matters of great concern, not only to the United States but also to the world community” ~ John Ashcroft
- Slides: 115