HALOGENS Chlorine Fluorine GROUP 7 A Bromine Iodine

HALOGENS Chlorine Fluorine GROUP 7 A Bromine Iodine
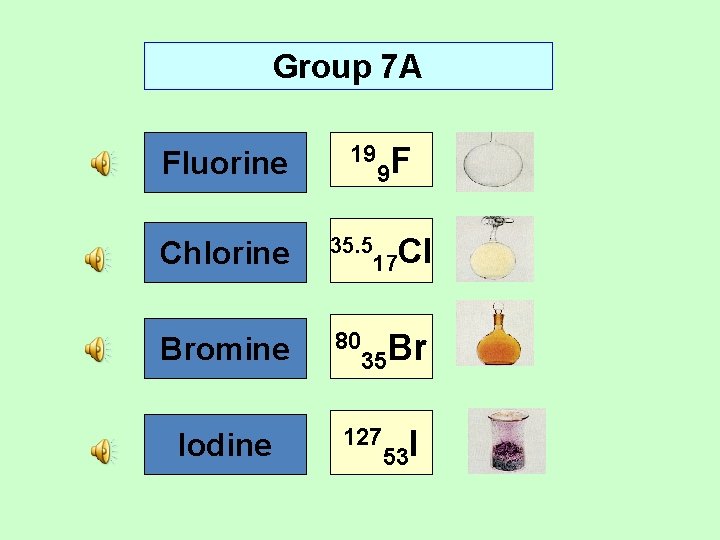
Group 7 A Fluorine 19 Chlorine 35. 5 Bromine 80 Iodine 9 F 17 Cl 35 Br 127 53 I

HALOGENS *Halos-genes is meaning of ‘salt former’ in Greek language. *They produce salts with metals. *They have 7 valence electrons. *They have -1 oxidation state in their stable compounds. *Fluorine is the most active nonmetal.

They are found as diatomic molecules in nature, F 2, Cl 2, Br 2, I 2.

Hydrogen has some properties of halogens. Come here !

Fluorine is the most active nonmetal

STATES OF HALOGENS AT ROOM TEMPERATURE • fluorine and chlorine are gases. • bromine is liquid. • iodine is solid.
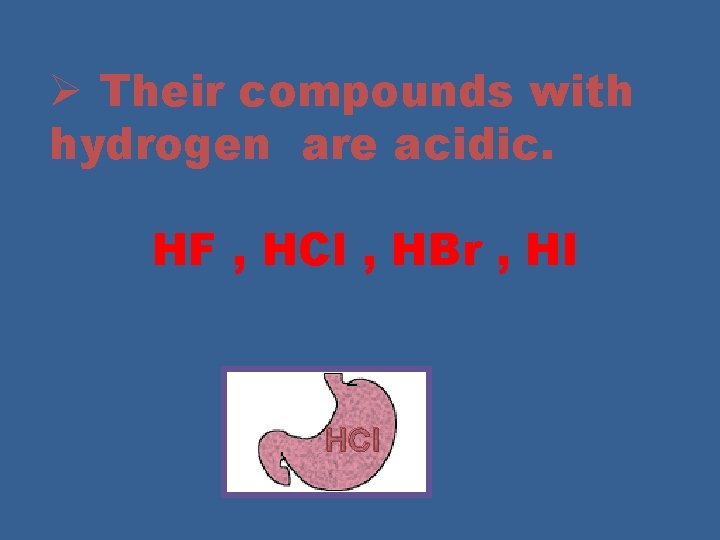
Ø Their compounds with hydrogen are acidic. HF , HCl , HBr , HI HCl

FLUORINE (F 2) Origin and meaning Fluere- to flow Earth`s crust abundance: 5. 4 x 10 -2 % Electron configuration: [He]2 s 22 p 5 Common oxidation num. Melting point: -219. 6 0 C Physical state: gas Boiling point: 188. 1 0 C Color: Pale yellow -1

FLUORINE (F 2) *The word fluorine is derived from the Latin language ‘fluere’ meaning flowing. * Fluorine is the most active nonmetal. *A gas in pale yellow in color. *Has only -1 oxidation number in compounds. *Very important for teeth, but excess amount causes darkening and decay of teeth.

FLUORINE COMPOUNDS (F 2) *Hydrofluoric acid (HF) is the only acid may react with Si. O 2 , so it can not be stored in glass container. * HF is used to decorate glassware. *Used in manufacture of polymers such as teflon. F 2 is compenent of freon gas (CCl 2 F 2) used in refrigerators.
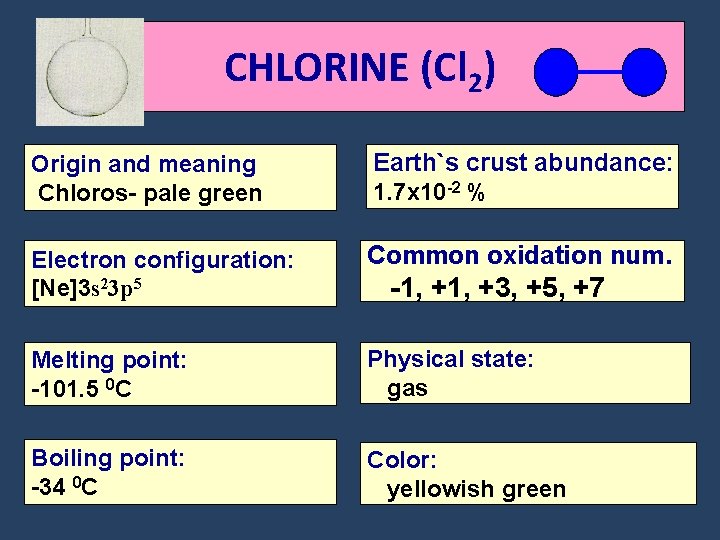
CHLORINE (Cl 2) Origin and meaning Chloros- pale green Earth`s crust abundance: 1. 7 x 10 -2 % Electron configuration: [Ne]3 s 23 p 5 Common oxidation num. Melting point: -101. 5 0 C Physical state: gas Boiling point: -34 0 C Color: yellowish green -1, +3, +5, +7

CHLORINE (Cl 2) • The word chlorine is derived from the Greek word ‘chloros’ meaning greenish yellow • Chlorine is very reactive gas. • It is found as diatomic molecule(Cl 2) • Poisonous and soluble in water. • Found in salt beds, sea water (2. 8 %) and in salts such as Na. Cl, KCl and Mg. Cl 2. )

INDUSTRIAL PRODUCTION Chlorine is prepared by the electrolysis of either molten Na. Cl salt or of an aqueous solution of Na. Cl. 2 Na. Cl(l) electrolysis 2 Na(l) + Cl 2(g) 2 Na. Cl(l) +H 2 O electrolysis 2 Na. OH(aq) + H 2 +Cl 2(g
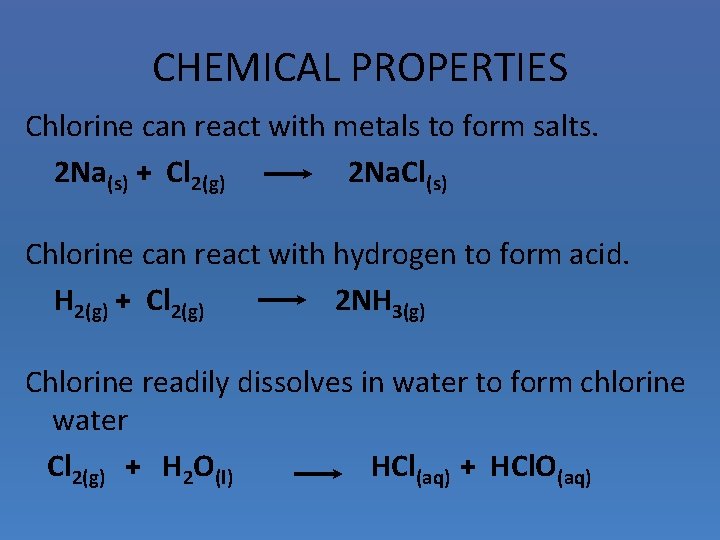
CHEMICAL PROPERTIES Chlorine can react with metals to form salts. 2 Na(s) + Cl 2(g) 2 Na. Cl(s) Chlorine can react with hydrogen to form acid. H 2(g) + Cl 2(g) 2 NH 3(g) Chlorine readily dissolves in water to form chlorine water Cl 2(g) + H 2 O(l) HCl(aq) + HCl. O(aq)

Hydrochloric acid (HCl) *It is one of the strong acids. * It is found in our stomach for digestion. *Used in manufacture of PVC, dyes, rubber and medicine. *The concentrated HCl used in the laboratories is % 36.

USES OF CHLORINE (Cl 2) *Used in manufacture of dyes, medical drugs, explosives, as a bleaching powder in tap water, insecticides , plastic polyvinyl chloride(PVC) and production of various chemicals such as HCl.

QUESTIONS *Explain why a solution of chlorine is acidic? *Why we use chlorine to clean water although chlorine is poisonous gas? * HCl acid is strong acid. If you drink this acid you will be die. How our stomach protect itself from this acid? *How salt preserve food from decay? *Why we throw salt to road at winter? * Why sea water is salty?
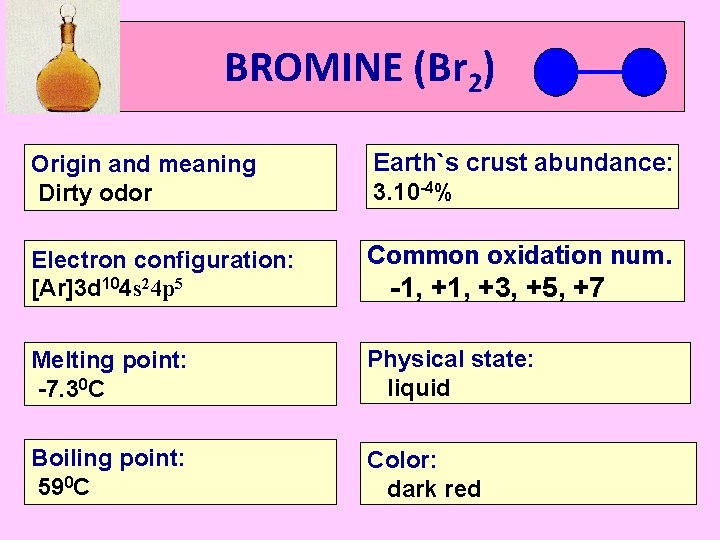
BROMINE (Br 2) Origin and meaning Dirty odor Earth`s crust abundance: 3. 10 -4% Electron configuration: [Ar]3 d 104 s 24 p 5 Common oxidation num. Melting point: -7. 30 C Physical state: liquid Boiling point: 590 C Color: dark red -1, +3, +5, +7

BROMINE (Br 2) * Only liquid nonmetal at room temperature. * A liquid with a dark red color. *Meaning of dirty odor in Latin language. * Very reactive and toxic. *Main source is sea water.
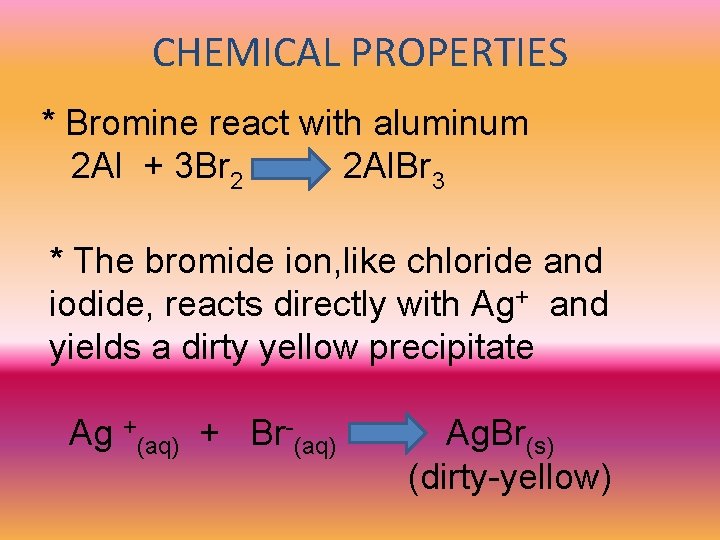
CHEMICAL PROPERTIES * Bromine react with aluminum 2 Al + 3 Br 2 2 Al. Br 3 * The bromide ion, like chloride and iodide, reacts directly with Ag+ and yields a dirty yellow precipitate Ag +(aq) + Br-(aq) Ag. Br(s) (dirty-yellow)

USES OF BROMINE Used in agriculture as drugs, in manufacture of films and drug industry. Used to protect engines from knocking. Added to fuel-oil.
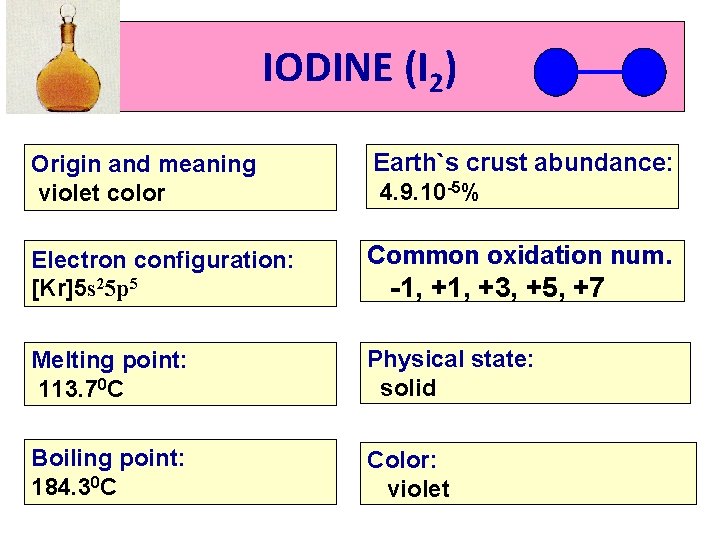
IODINE (I 2) Origin and meaning violet color Earth`s crust abundance: 4. 9. 10 -5% Electron configuration: [Kr]5 s 25 p 5 Common oxidation num. Melting point: 113. 70 C Physical state: solid Boiling point: 184. 30 C Color: violet -1, +3, +5, +7

IODINE (I 2) *A solid, violet-dark in color. *Iodos means violet color in Latin language. * Iodine is insoluble in water but soluble in alcohol. *Sublimates easily by releasing violet vapor which has unpleasant odor. * The solutions of iodine in alcohols are used as antiseptics in medicines.

USES OF IODINE *Used as antiseptic(tincture of iodine) in medicine. * Iodine is used to detect starch because it turns blue when it comes in contact with starch. *Used in halogen lamps in car headlights. *The deficiency of iodine in body causes goiter disease that is why ionized table salt is produced.

QUESTIONS • What kind of illness caused by deficiency of iodine? • What is the meaning of iodine ? • What is the first halogen found in nature? • Halogens compounds damage the ozone layer. What is the function of ozone.
- Slides: 27