Halogenoalkanes AH Chemistry Unit 3b Background Also known

Halogenoalkanes AH Chemistry Unit 3(b)


Background • Also known as haloalkanes or alkyl halides • Rare in the natural world • Widely used • Synthesised in the lab

Nomenclature practise • 2, 3 -dichloropentane • 3 -bromobut-1 -ene • 2, 3 -dibromo-1 -chloro-2 -methylbutane

Primary, secondary, tertiary • Draw a primary halogenoalkane and name it • Draw a secondary halogenoalkane and name it • Draw a tertiary halogenoalkane and name it

Reactions • Depends on two factors… – Type of halogen atom – Position in molecule • Two characteristic reactions: – Nucleophilic substitution – Elimination

Nucleophilic substitution
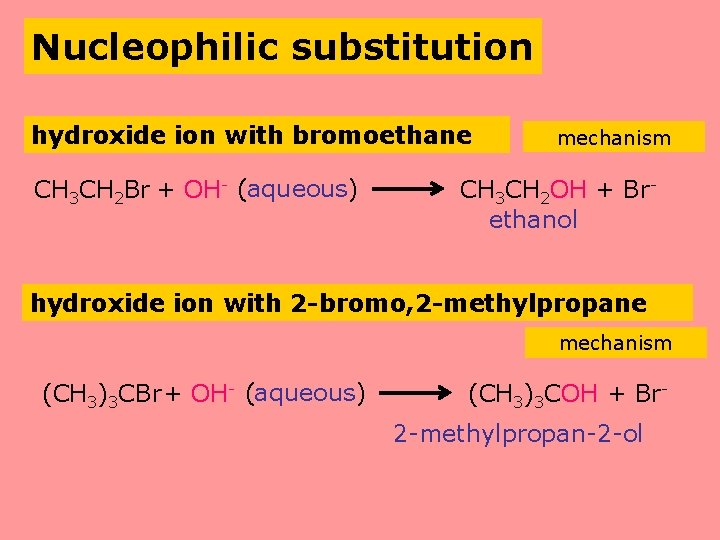
Nucleophilic substitution hydroxide ion with bromoethane CH 3 CH 2 Br + OH- (aqueous) mechanism CH 3 CH 2 OH + Brethanol hydroxide ion with 2 -bromo, 2 -methylpropane mechanism (CH 3)3 CBr+ OH- (aqueous) (CH 3)3 COH + Br 2 -methylpropan-2 -ol
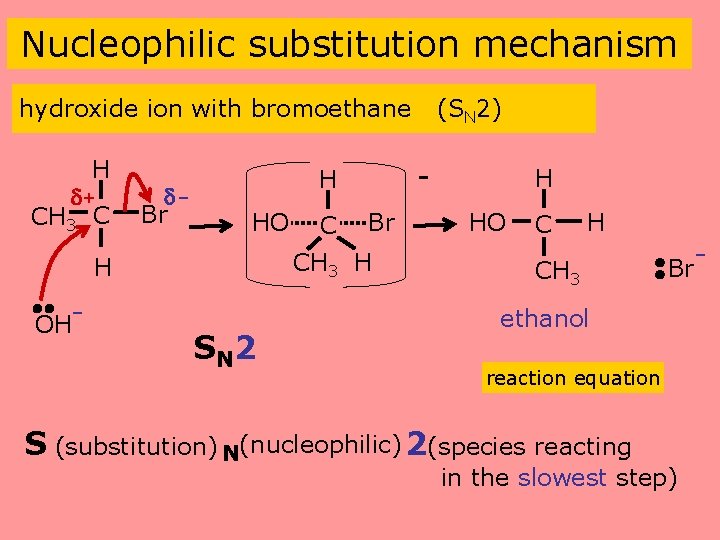
Nucleophilic substitution mechanism hydroxide ion with bromoethane H + CH 3 C Br CH 3 H H OH- - H HO SN 2 (SN 2) H HO C H CH 3 Br ethanol reaction equation S (substitution) N(nucleophilic) 2(species reacting in the slowest step)
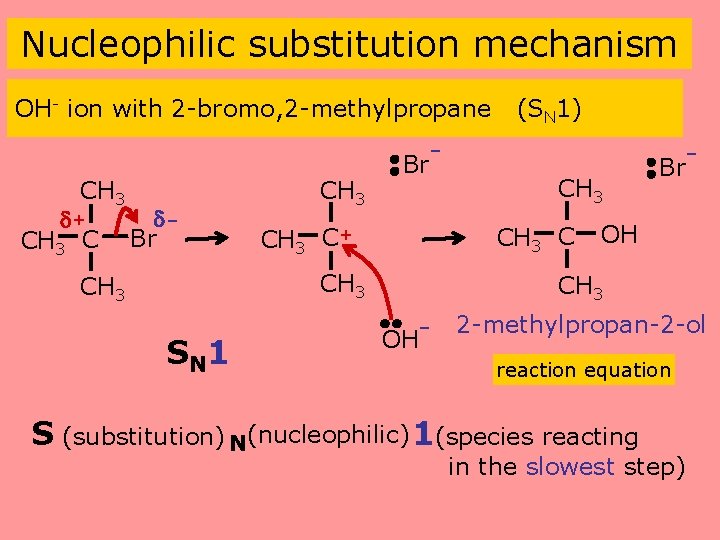
Nucleophilic substitution mechanism OH- ion with 2 -bromo, 2 -methylpropane (SN 1) - CH 3 + CH 3 C Br CH 3 C+ CH 3 SN 1 CH 3 Br OH CH 3 OH- 2 -methylpropan-2 -ol reaction equation S (substitution) N(nucleophilic) 1(species reacting in the slowest step)

General rule • Primary and secondary halogenoalkanes tend to undergo SN 2 reactions • Tertiary halogenoalkanes tend to undergo SN 1 reactions • Can you suggest why?

Important substitution reactions Water or aqueous alkali Alcohols Can then be converted to aldehydes, ketones or carboxylic acids Ammonia Amines Alcoholic alkoxides Ethers A sodium alkoxide is produced in the reaction of sodium with a dry alcohol Potassium cyanide (ethanolic solution) Nitriles Increases length of carbon chain
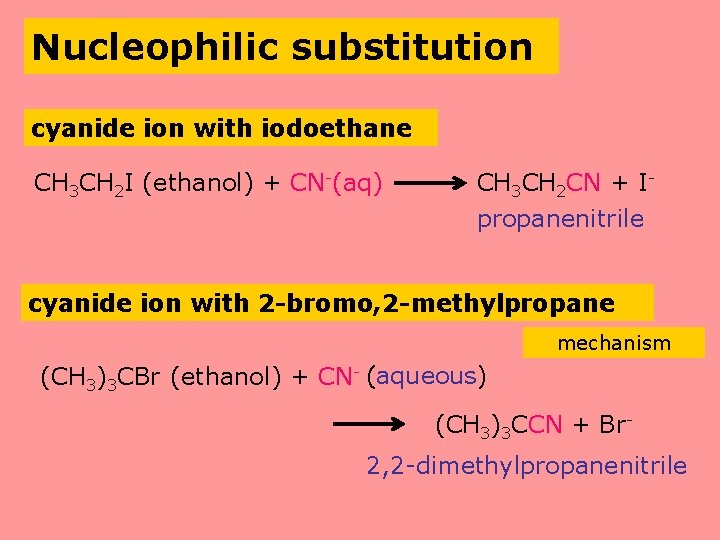
Nucleophilic substitution cyanide ion with iodoethane CH 3 CH 2 I (ethanol) + CN-(aq) CH 3 CH 2 CN + Ipropanenitrile cyanide ion with 2 -bromo, 2 -methylpropane mechanism (CH 3)3 CBr (ethanol) + CN- (aqueous) (CH 3)3 CCN + Br 2, 2 -dimethylpropanenitrile
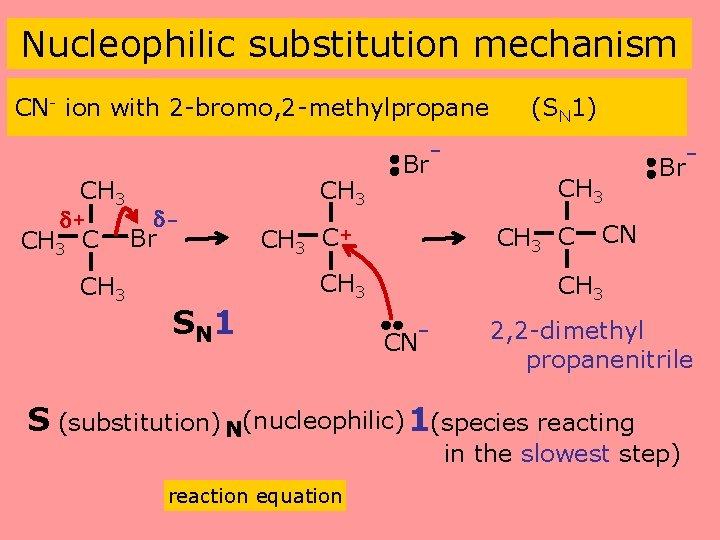
Nucleophilic substitution mechanism CN- ion with 2 -bromo, 2 -methylpropane (SN 1) - CH 3 + CH 3 C Br CH 3 C+ CH 3 C CH 3 SN 1 CH 3 Br CN CH 3 CN- 2, 2 -dimethyl propanenitrile S (substitution) N(nucleophilic) 1(species reacting in the slowest step) reaction equation
- Slides: 14