Haloalkanes Learning Objectives 1 Understand reactivity of haloalkanes

Haloalkanes Learning Objectives: 1. Understand reactivity of haloalkanes 2. Explain nucleophilic substitution reactions 3. Explain elimination reactions & the conditions 4. Describe the free radical substitution mechanism to create haloalkanes

Starter chloroethane 1, 3 -dibromo-2 -chlorobutane 2 -bromomethylpropane 1 chloro-2, 2 -dimethlypropane

Cn. H 2 n+1 X or R – X Which halogen? • • How many? Fluoro Chloro Bromo Iodo • Di • Tri • Tetra- etc. . . Which carbon? Which order? • Alphabetical NOT • Numerical • Use numbers for which C the halogen attached Cl I 3 -chloro-2 -iodopentane

Using Unit 1 knowledge. . . what can you predict about physical properties? – Electronegativity – Dipoles – Boiling points

Physical properties • Polar bonds: + - C – F most polar (2. 5 & 4. 0) C–I least polar (2. 5 & 2. 6) • Solubility – insoluble in water, need organic solvent • Boiling point: – Increase with chain length – Increase down halogen group • Bond enthalpy: (Due to increased vd. W) C – F bond strongest C – I bond weakest • Bond enthalpy more important factor than polarity reactivity increases down the group

Activity 1 i. iii. iv. a) b) c) d) CH 3 CH 2 CH 2 I CH 3 CHBr. CH 3 CH 2 Cl. CH 2 CH 3 CH 2 CHBr. CH 3 1 -iodobutane 2 -bromopropane 1 -chlorobutane 2 -bromobutane Draw displayed formula for each & mark polarity of C-X bond. c) 1 -iodobutane because highest Mr, most e- & highest vd. W forces Name each. Predict which would have the highest b. p. & explain. Explain why halogens get less reactive as we go up the halogen group. d) This is because the C-X bond becomes stronger

3 types of haloalkanes • • • Primary - 2 H’s bonded to C with halogen atom Secondary - 1 H bonded to C with halogen atom Tertiary - 0 H bonded to C with halogen atom

What type of haloalkane are these? 1. 2 -chloro-2 methylbutane 2. 1 -chloropropane 3. 2 -chloropropane 1. Tertiary 2. Primary 3. Secondary

What’s a nucleophile? • Ideas? • A reagent that attacks & bonds with + C atoms: Ø Either –ve ion OR atom with δ- charge Ø With a lone pair electrons Ø Lone pair is on an electronegative atom • Bonds by donating l. p. e- to electron deficient C atom • E. g -: OH, : NH 3, -: CN A nucleophile is a species which attacks a + centre by donating a pair of electrons

Nucleophilic substitution reaction • : Nu- will replace the halogen in haloalkane

Nucleophilic substitution mechanism • All follow same pattern so apply general mechanism + - Curly arrow notation: 1. Indicates the movement of electrons 2. Tail comes from a lone pair or a bonded pair of electrons 3. Arrow goes to an atom to form a lone pair or next to an atom to form a bonded pair of electrons
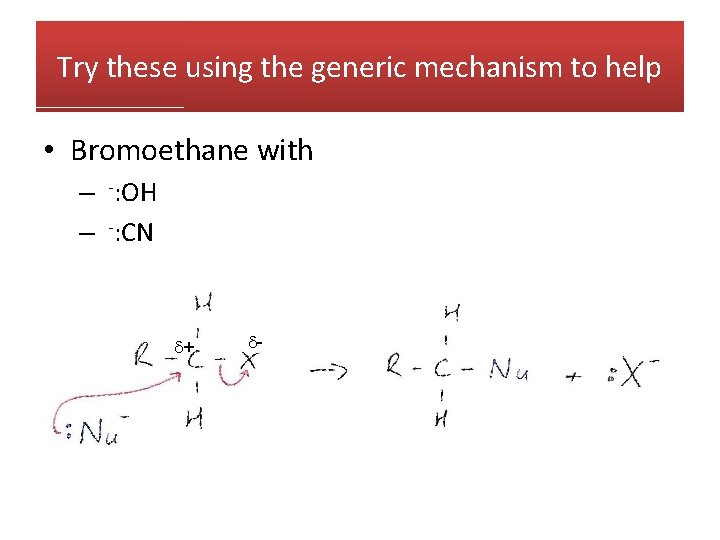
Try these using the generic mechanism to help • Bromoethane with – -: OH – -: CN + -
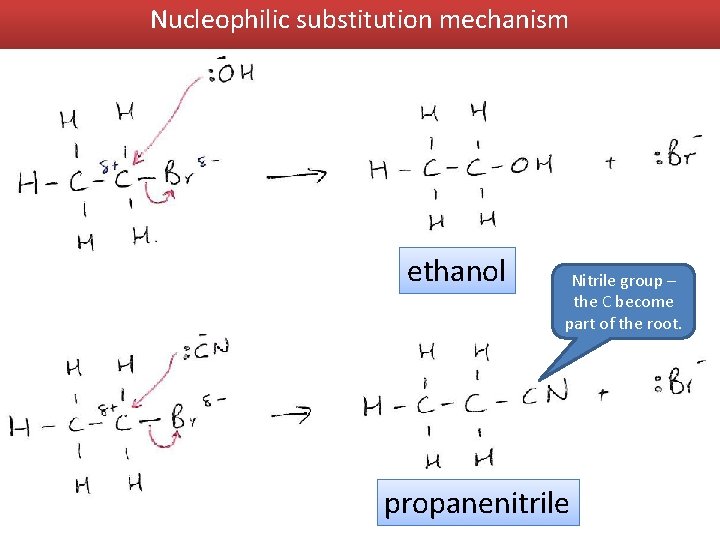
Nucleophilic substitution mechanism ethanol Nitrile group – the C become part of the root. propanenitrile
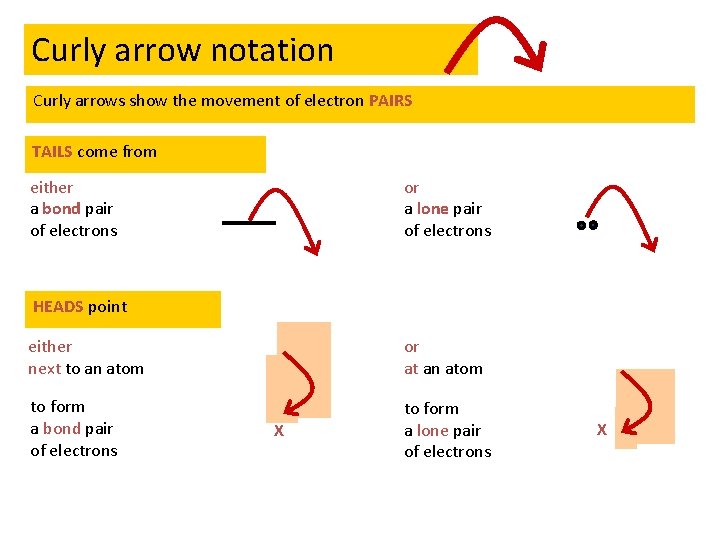
Curly arrow notation Curly arrows show the movement of electron PAIRS TAILS come from either a bond pair of electrons or a lone pair of electrons HEADS point either next to an atom or at an atom to form a bond pair of electrons to form a lone pair of electrons X X

Haloalkanes with NH 3 • Same mechanism but a 2 nd step • Overall reaction: amine
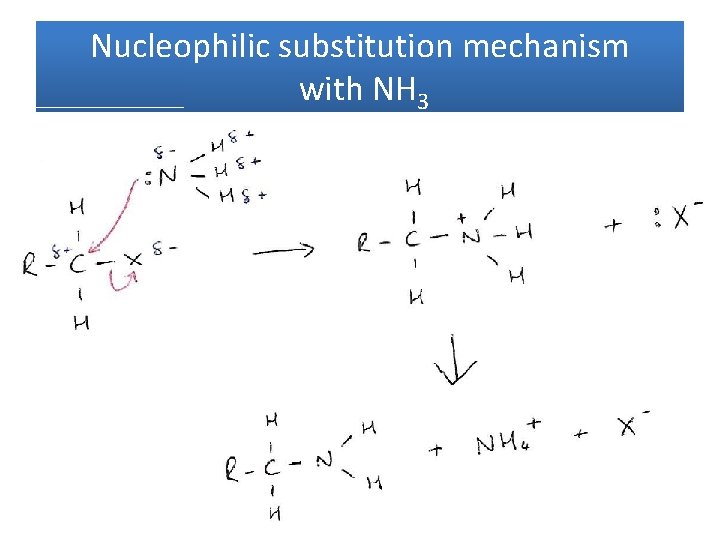
Nucleophilic substitution mechanism with NH 3

New functional groups • With -: OH to form alcohols R - OH • With -: CN to form nitriles (extends length of chain by 1 C) R – CN • With NH 3 to form amines R – NH 2

Reaction conditions • Use pgs 189 -190 to identify reaction conditions e. g. : – Solvent – Temperature
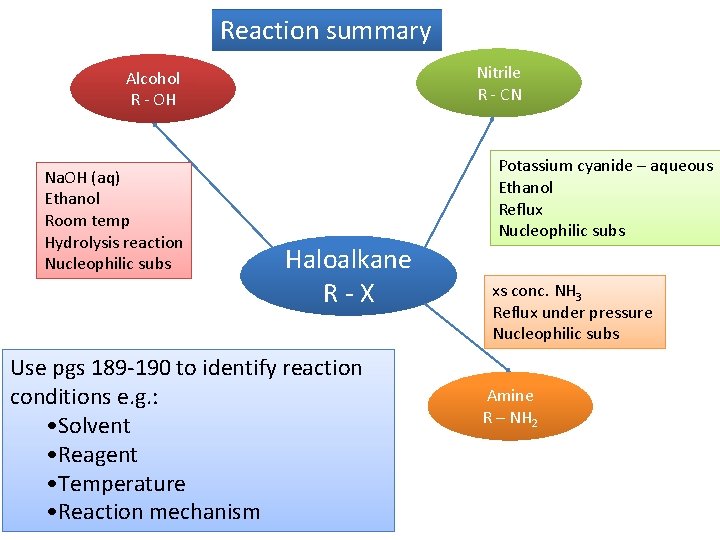
Reaction summary Nitrile R - CN Alcohol R - OH Na. OH (aq) Ethanol Room temp Hydrolysis reaction Nucleophilic subs Potassium cyanide – aqueous Ethanol Reflux Nucleophilic subs Haloalkane R-X Use pgs 189 -190 to identify reaction conditions e. g. : • Solvent • Reagent • Temperature • Reaction mechanism xs conc. NH 3 Reflux under pressure Nucleophilic subs Amine R – NH 2

Plenary • Exam question pg 197 Q 4

Haloalkanes 1. Nucleophilic substitution reactions 2. Elimination reactions – Under different conditions

Elimination reactions • H & X eliminated from the molecule • Double bond left in its place = alkene formed • Different reaction conditions: – -: OH acting as a base (not a Nu) removes H+ ion – KOH dissolved in ethanol (ethanolic KOH) – No water present – Heat required

Elimination mechanism • OH- acts a base – i. e. a proton acceptor – suggest a possible mechanism using curly arrows.

How do would you. . . show one of the products is an alkene? • Add bromine water – Decolourises in presence of a double bond

Preferences Primary Secondary Tertiary Elimination Substitution
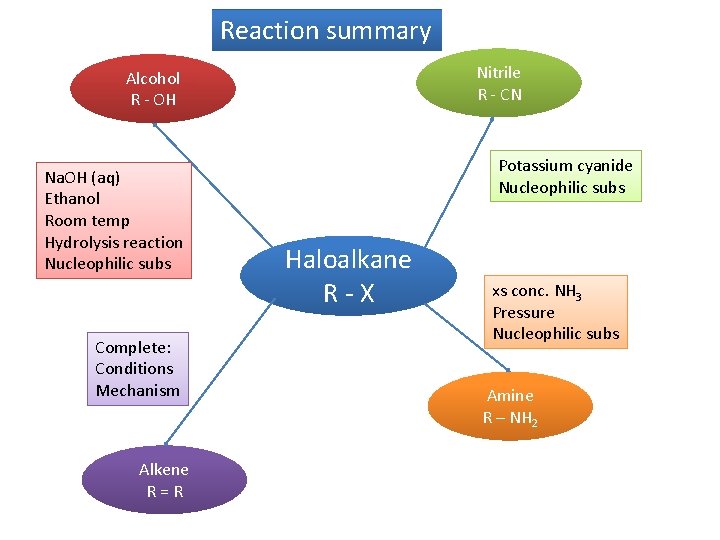
Reaction summary Nitrile R - CN Alcohol R - OH Na. OH (aq) Ethanol Room temp Hydrolysis reaction Nucleophilic subs Complete: Conditions Mechanism Alkene R=R Potassium cyanide Nucleophilic subs Haloalkane R-X xs conc. NH 3 Pressure Nucleophilic subs Amine R – NH 2

Formation of haloalkanes • Free radical substitution • UV light • Three stages: – Initiation – Propagation – Termination UV Light CH 4(g) + Cl 2(g) CH 3 Cl(g) + HCl(g)
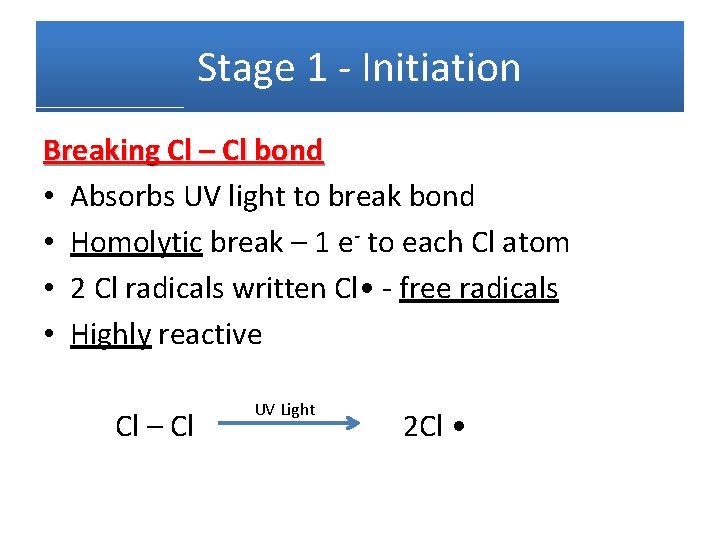
Stage 1 - Initiation Breaking Cl – Cl bond • Absorbs UV light to break bond • Homolytic break – 1 e- to each Cl atom • 2 Cl radicals written Cl • - free radicals • Highly reactive Cl – Cl UV Light 2 Cl •
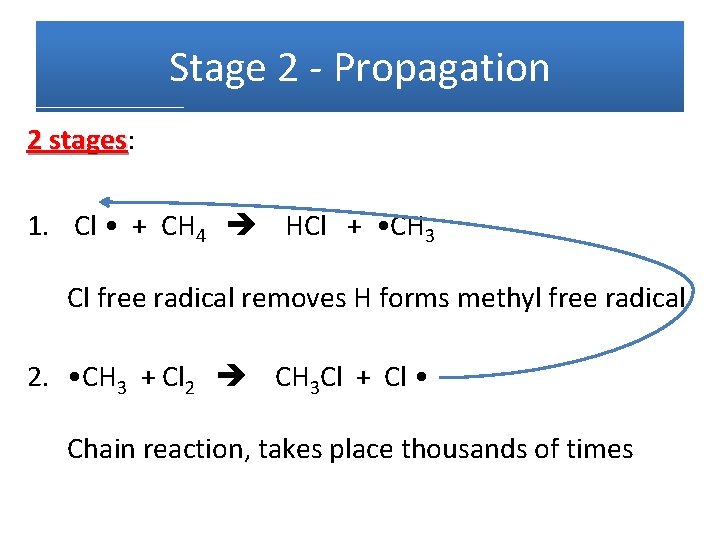
Stage 2 - Propagation 2 stages: stages 1. Cl • + CH 4 HCl + • CH 3 Cl free radical removes H forms methyl free radical 2. • CH 3 + Cl 2 CH 3 Cl + Cl • Chain reaction, takes place thousands of times
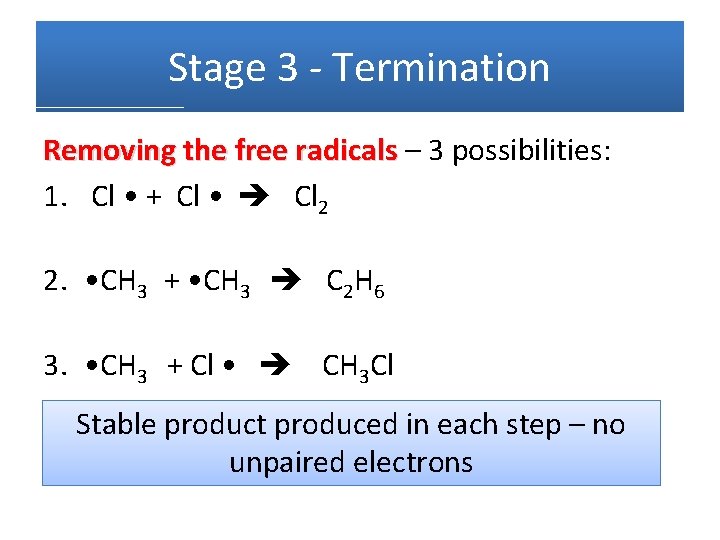
Stage 3 - Termination Removing the free radicals – 3 possibilities: 1. Cl • + Cl • Cl 2 2. • CH 3 + • CH 3 C 2 H 6 3. • CH 3 + Cl • CH 3 Cl Stable product produced in each step – no unpaired electrons
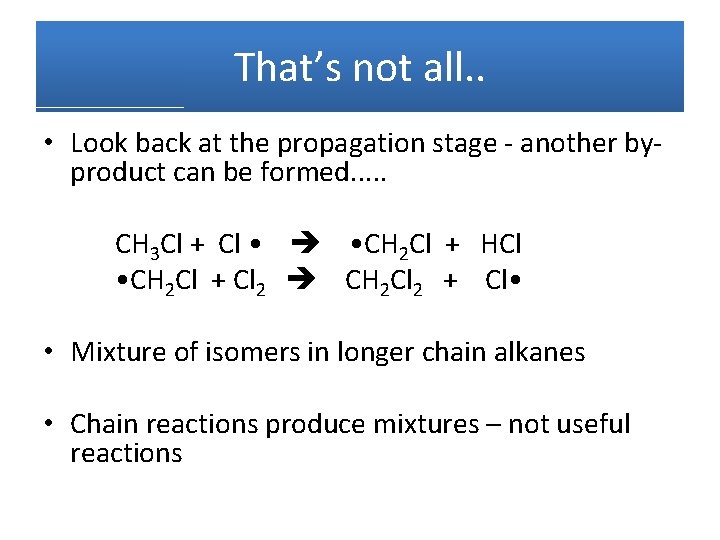
That’s not all. . • Look back at the propagation stage - another byproduct can be formed. . . CH 3 Cl + Cl • • CH 2 Cl + HCl • CH 2 Cl + Cl 2 CH 2 Cl 2 + Cl • • Mixture of isomers in longer chain alkanes • Chain reactions produce mixtures – not useful reactions

Find out why CFC’s are a problem Prepare short presentation to cover: • What are CFC’s? • What is ozone and why is it important? • Why are CFC’s and ozone not a good combination? • What is the reaction? Pages 193 & 195


(a) dichlorodifluoromethane / accept halogens reversed (1) 1 ignore number ‘l’ in name; penalise other numbers ignore hyphens and gaps (b) appropriate tetrahedral shape (1) explains that tetrahedral shape due to repulsion between (four bonding) pairs of electrons not pairs of atoms repelling (1) repulsion unequal due to bonds to different atoms / covalent radii / bond lengths / different electronegativity / dipoles (1) 3 (c) C–Cl bond weaker / longer than C-F bond / C–F bond is stronger (1) 1 [5] d) Mark for each of 3 reactions on pg 195 with description Plus acts as a catalyst.

Plenary • Exam question 3 pg 196
- Slides: 35