Halloween Lab Get your paper ready for bell

Halloween Lab! Get your paper ready for bell work. One question today so we can start quickly.

Bell Work 10 -31 -16 Which diagram best represents the phase change of gas to liquid? A. C. C. B. D. What is necessary to cause a change in phase in the three most common phases? Justify your answer!

Success Criteria: • I can identify particle movement in a solid, liquid, and gas. • I can compare particles in a solid, liquid, or gas. • I can identify sublimation. TOC #15. States of Matter & Energy #16. Halloween Lab
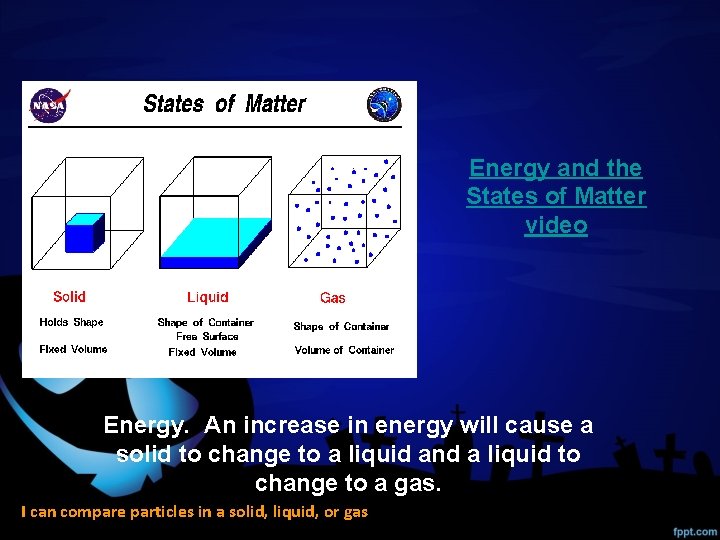
Energy and the States of Matter video Energy. An increase in energy will cause a solid to change to a liquid and a liquid to change to a gas. I can compare particles in a solid, liquid, or gas

#15. States of Matter & Energy Sublimation Melting Solid Evaporation Liquid Freezing I can compare particles in a solid, liquid, or gas Condensation Gas

#16. Halloween Lab! Success Criteria: I can identify particle movement in a solid, liquid, and gas. I can compare the particle movements in a solid, liquid, and gas. I can identify sublimation. Disappearing Ice Group break outs 1. 2. 3. Dry Ice Example: Dry Ice Demonstration Root Beer Float Exit Response

Disappearing Ice Demonstration! • Write your prediction about what’s going to happen when a piece of dry ice and an ice cube are placed on paper plates. What could we do to make them evaporate more quickly? • At the end of class, let’s see what happened. Be ready to explain! I can compare particles in a solid, liquid, or gas
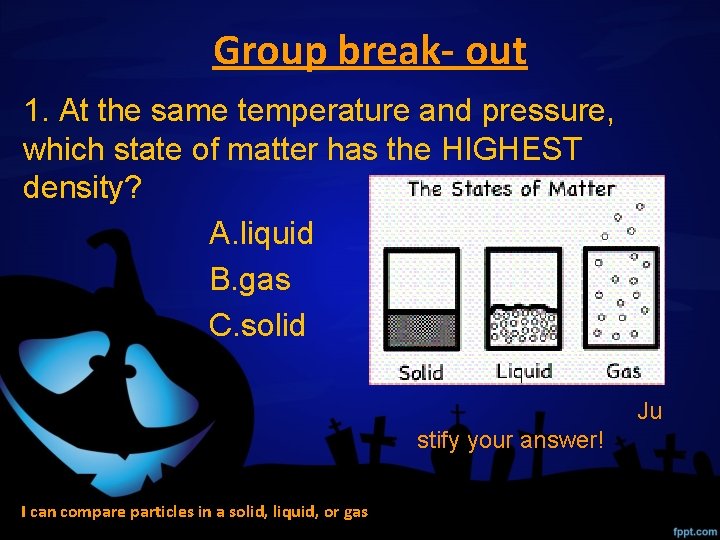
Group break- out 1. At the same temperature and pressure, which state of matter has the HIGHEST density? A. liquid B. gas C. solid Ju stify your answer! I can compare particles in a solid, liquid, or gas
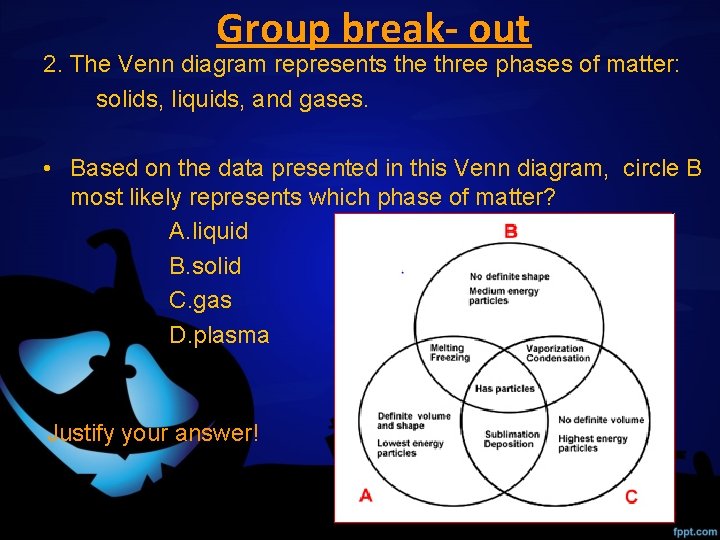
Group break- out 2. The Venn diagram represents the three phases of matter: solids, liquids, and gases. • Based on the data presented in this Venn diagram, circle B most likely represents which phase of matter? A. liquid B. solid C. gas D. plasma Justify your answer!

Group break- out 3. Which diagram best shows the type of particle arrangement associated with a gas? a. B. C. D. Justify your answer! I can compare particles in a solid, liquid, or gas

Dry Ice Demonstration Dry Ice is Solid Carbon Dioxide (CO 2). Caution: Dry ice is very dangerous and you should always use proper safety when handling it. Always wear gloves and never keep in a closed container. Sublimation- state in which a solid changes directly to a gas. Let's experiment! Analyze what happened! I can compare particles in a solid, liquid, or gas

Steve Spangler Halloween I can compare particles in a solid, liquid, or gas

Root Beer Floats: States of Matter • Measure ½ cup of root beer and pour into cup. • Next add 1 scoop of ice cream. • Slowly add more root beer to the cup and observe what happens. • What is produced? Is this a physical or chemical change? • On you chart, draw and label the solid, liquid, and gas produced when making the root beer float (ice cream, root beer, air bubbles) I can compare particles in a solid, liquid, or gas

Disappearing Ice! • Share your conclusions on your lab page. I can compare particles in a solid, liquid, or gas

Exit You get to be a molecule in a solid, a liquid, or a gas. Which would you rather be? Why? Explain with evidence about the state of matter you chose. I can compare particles in a solid, liquid, or gas
- Slides: 15