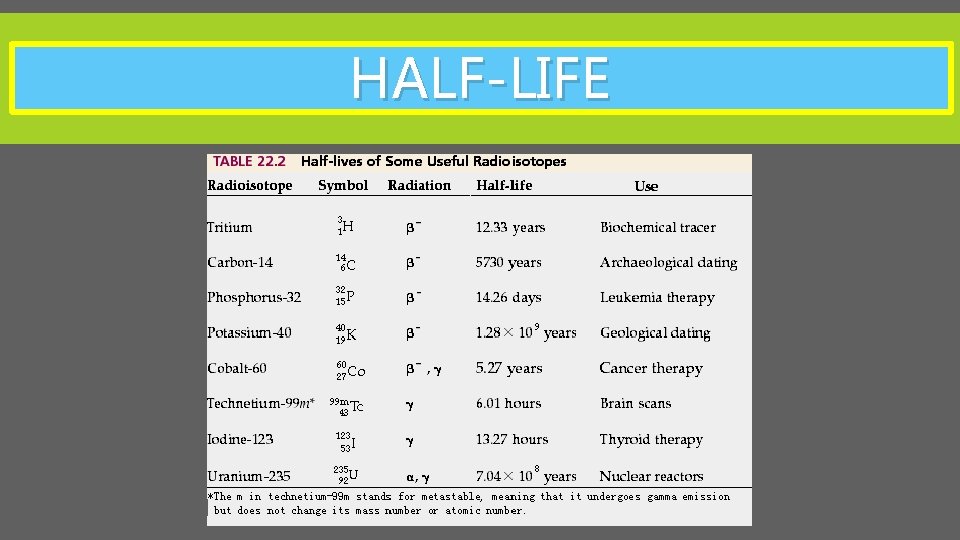
HALFLIFE Its impossible to know exactly when an

HALF-LIFE It’s impossible to know exactly when an unstable atom will decay. We can however predict how many will decay in a period of time. A half-life is the time taken for half a group of unstable nuclei to decay. Half-lives vary according to the isotope that is decaying – these can range from microseconds, to thousands of millions of years.
HALF-LIFE
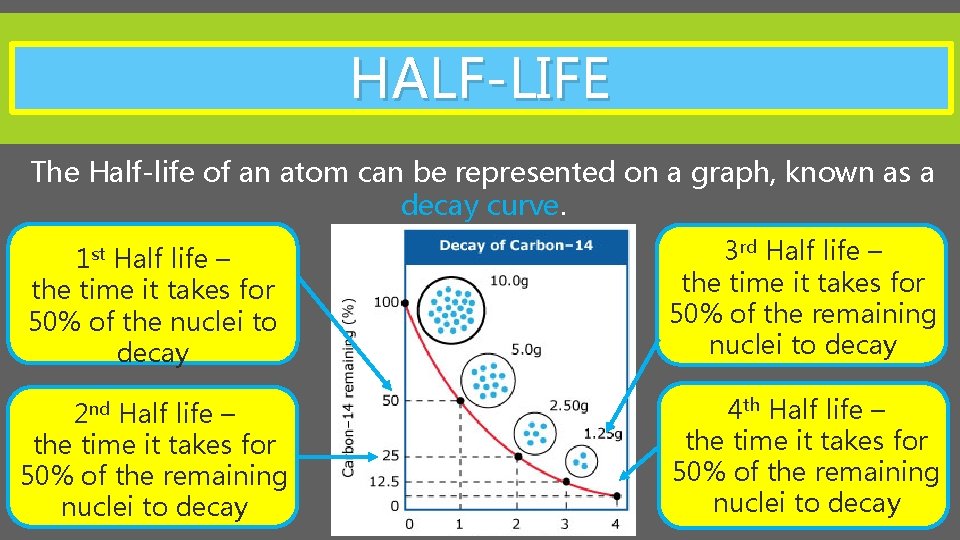
HALF-LIFE The Half-life of an atom can be represented on a graph, known as a decay curve. Half life – the time it takes for 50% of the nuclei to decay 3 rd Half life – the time it takes for 50% of the remaining nuclei to decay 2 nd Half life – the time it takes for 50% of the remaining nuclei to decay 4 th Half life – the time it takes for 50% of the remaining nuclei to decay 1 st
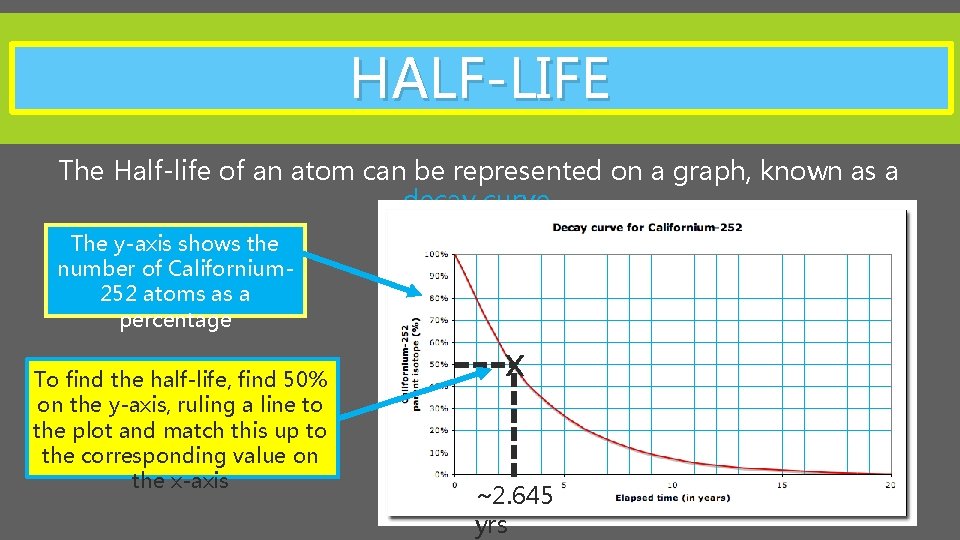
HALF-LIFE The Half-life of an atom can be represented on a graph, known as a decay curve. The y-axis shows the number of Californium 252 atoms as a percentage X To find the half-life, find 50% on the y-axis, ruling a line to the plot and match this up to the corresponding value on the x-axis ~2. 645 yrs
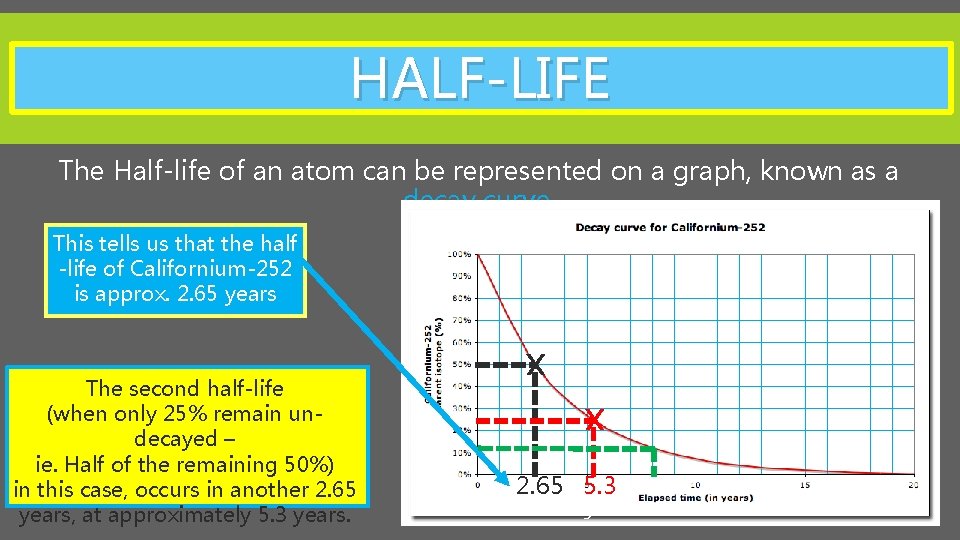
HALF-LIFE The Half-life of an atom can be represented on a graph, known as a decay curve. This tells us that the half -life of Californium-252 is approx. 2. 65 years X X The second half-life (when only 25% remain undecayed – ie. Half of the remaining 50%) in this case, occurs in another 2. 65 years, at approximately 5. 3 years. 2. 65 5. 3 3
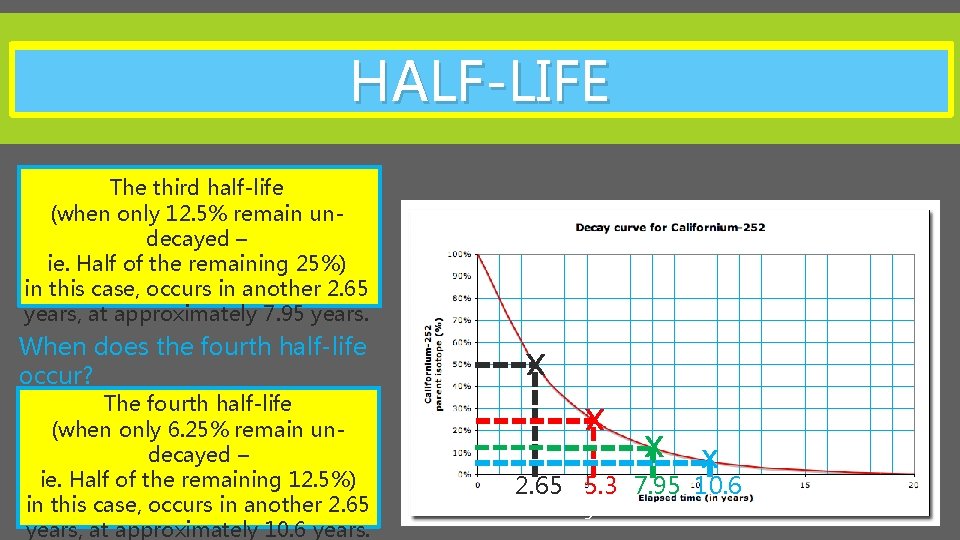
HALF-LIFE The third half-life (when only 12. 5% remain undecayed – ie. Half of the remaining 25%) in this case, occurs in another 2. 65 years, at approximately 7. 95 years. X The fourth half-life (when only 6. 25% remain undecayed – ie. Half of the remaining 12. 5%) in this case, occurs in another 2. 65 years, at approximately 10. 6 years. X When does the fourth half-life occur? X X 2. 65 5. 3 7. 95 10. 6 3
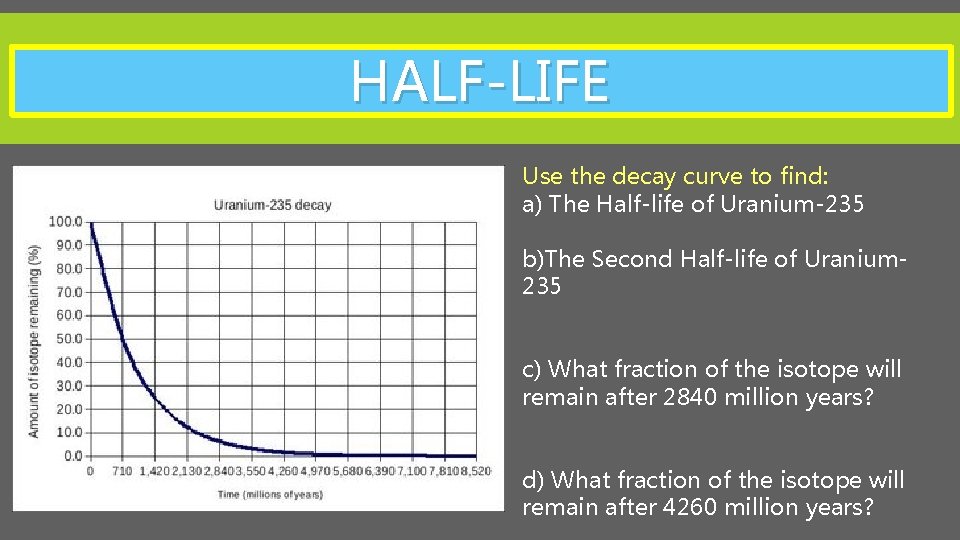
HALF-LIFE Use the decay curve to find: a) The Half-life of Uranium-235 b)The Second Half-life of Uranium 235 c) What fraction of the isotope will remain after 2840 million years? d) What fraction of the isotope will remain after 4260 million years?
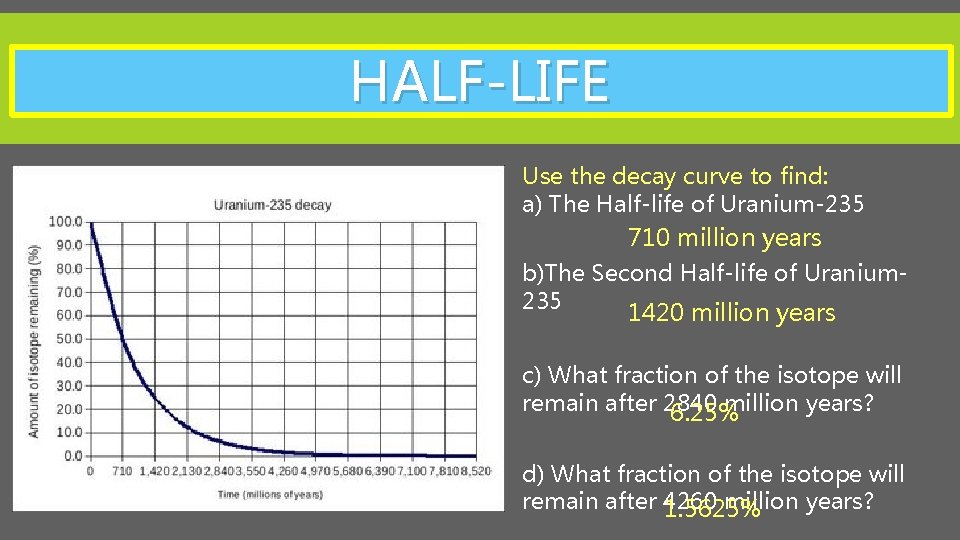
HALF-LIFE Use the decay curve to find: a) The Half-life of Uranium-235 710 million years b)The Second Half-life of Uranium 235 1420 million years c) What fraction of the isotope will remain after 2840 million years? 6. 25% d) What fraction of the isotope will remain after 4260 million years? 1. 5625%

NOW DO HALF-LIFE ~ SIMULATION TASK

MEASURING DECAY We can measure the ionising radiation of a radioactive source using a Geiger counter. • A Geiger counter detects Alpha, Beta and Gamma radiation. • The common unit for measuring radioactive decay is Becquerel (Bq). • Bq = number of decay’s per second. http: //atomic. lindahall. org/what-is-a-geiger-counter. html

MEASURING DECAY Refer to the graph below, showing the decay curve of Thorium-234. At the beginning when the decay is at large, the Geiger counter would of course be the most active, recording a high count rate Gradually decreasing over time So, if we measured the decay of a radioactive source as graphed it, it would be the same as the decay curve

MEASURING DECAY eg. A radioactive material is measured to have 600, 000 decays per second. 600, 000 Bq a) What is this equivalent to in Bq? b) After 3 half-lives, what will the activity be in Bq?

NOW TRY CHAPTER ONE - Q 17; 23 -29
- Slides: 13