HalfLife HalfLife u Time required for of the













- Slides: 24

Half-Life

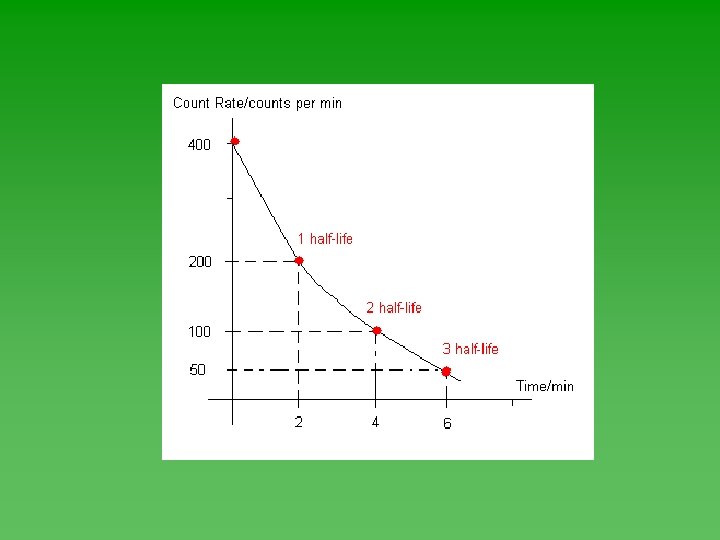
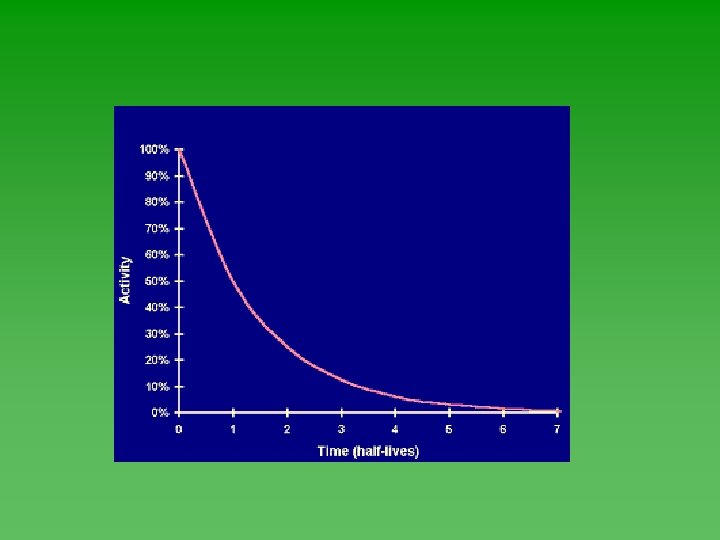
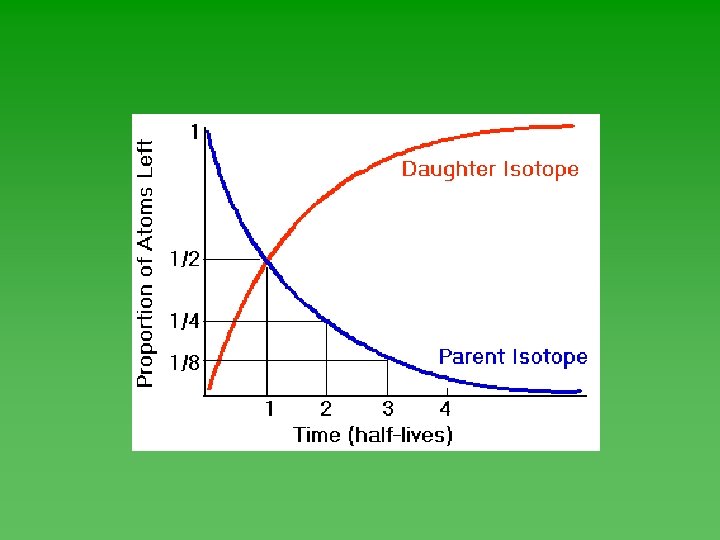
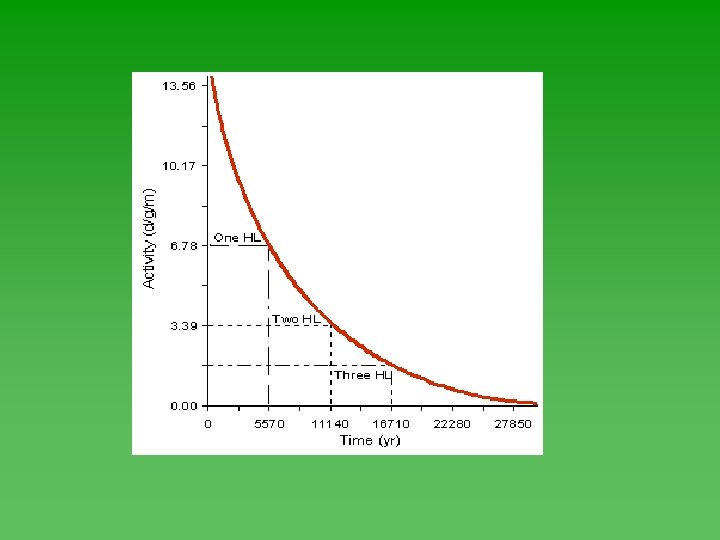
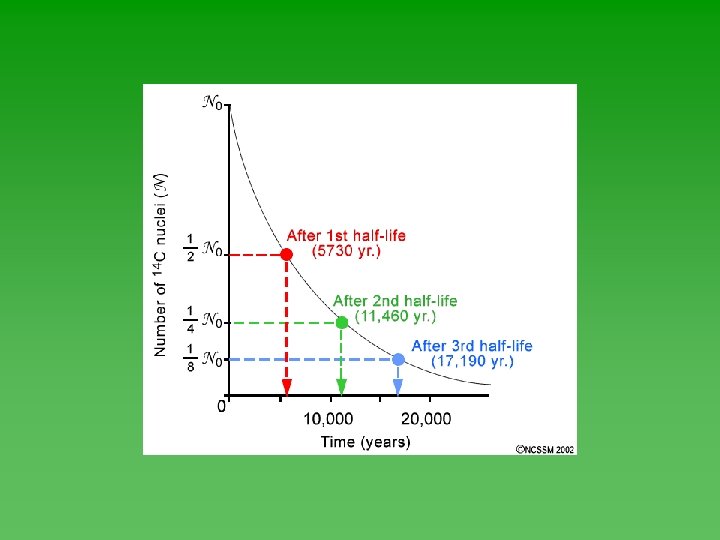
Half-Life u Time required for ½ of the atoms in a sample of radioactive nuclei to decay or change into something else. u Each isotope has its own half-life. u Range from fractions of a second to billions of years.

Half-Life u Half-life of each isotope - always the same. u Not affected by the chemical or physical environment of the nuclide. Not affected by changes in T, P, or concentration. u Doesn’t depend on how much you have or how long it’s been sitting around.

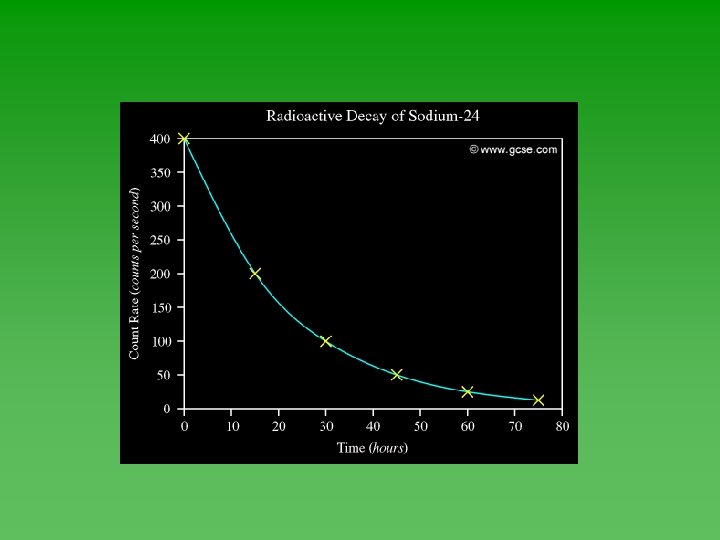
Geiger Counter

Radioactive Decay Another animation Compare C-10: half-life = 20 sec C-15: half-life = 2. 5 sec

Graphing Half-Life Data u Use Geiger counter to record decay events u Plot the data – always has the same shape. u Determine half-life from graph







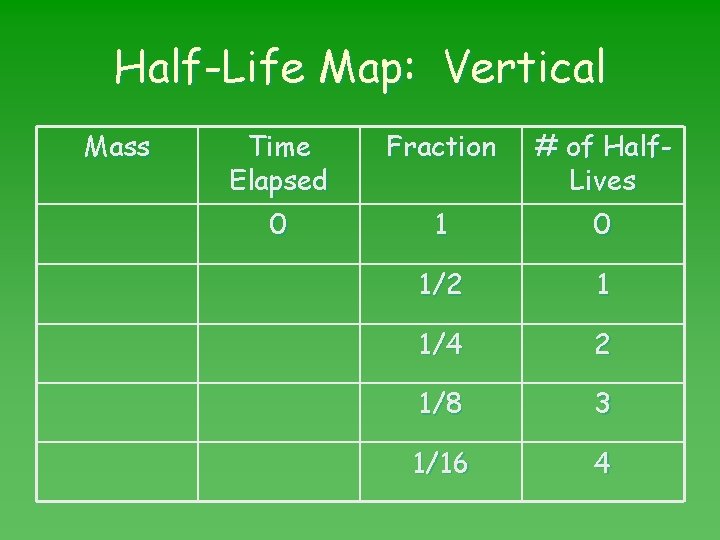
Half-Life Map: Vertical Mass Time Elapsed Fraction # of Half. Lives 0 1/2 1 1/4 2 1/8 3 1/16 4

No. of half-lives Number of half-lives = time elapsed length of 1 half-life Look up the half-life in Table N.

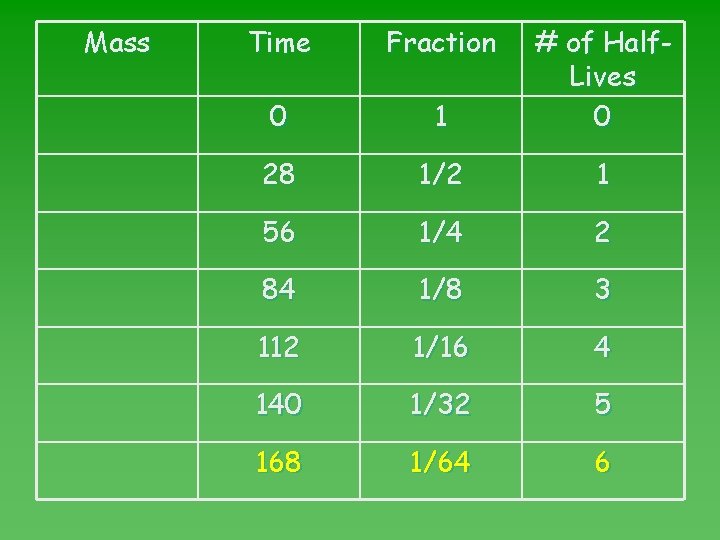
Problem #1 u Cr-51 has a half-life of 28 days. u What fraction will remain after 168 days? u Find number of half-lives in 168 days. Time elapsed Length of HL = 168 days = 6 HL’s 28 days

Problem #1 u Go to half-life map. u Read fraction remaining after 6 HL

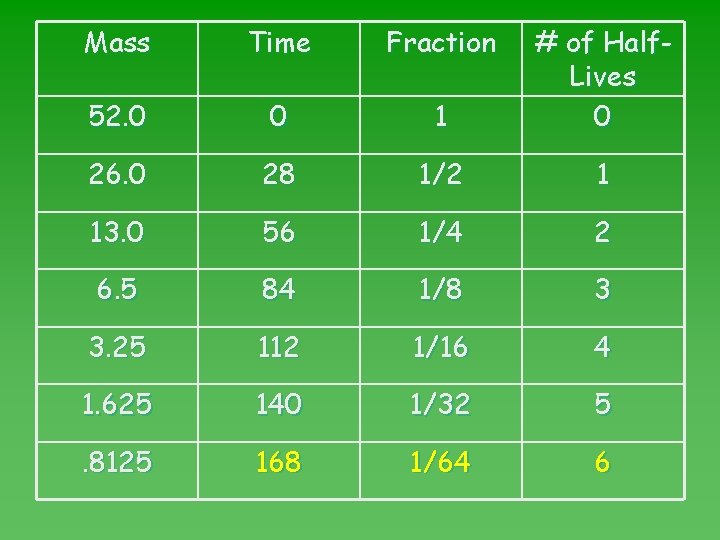
Mass Time Fraction 0 1 # of Half. Lives 0 28 1/2 1 56 1/4 2 84 1/8 3 112 1/16 4 140 1/32 5 168 1/64 6

Problem #2 u If a sample of Cr-51 has an original mass of 52. 0 g, how much will remain after 168 days? u Already u Go calculated 168 days = 6 HL. to half-life map. To calculate remaining mass, divide original by 2, SIX times! OR Multiply orignial by 1/64.

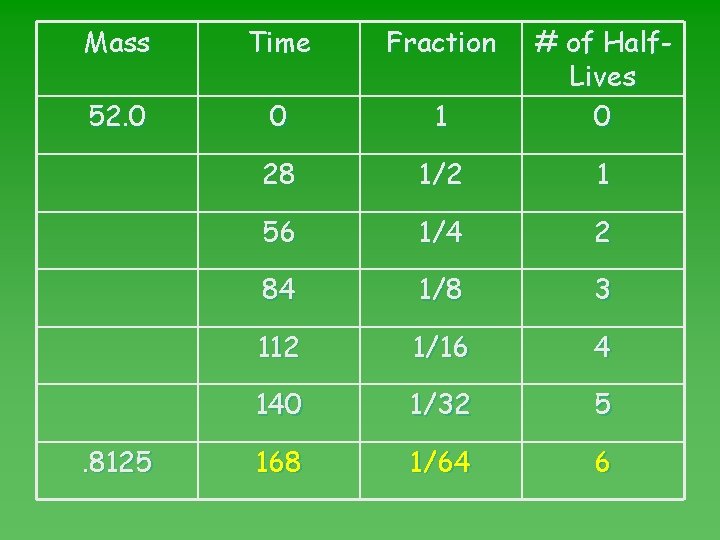
Mass Time Fraction 52. 0 0 1 # of Half. Lives 0 26. 0 28 1/2 1 13. 0 56 1/4 2 6. 5 84 1/8 3 3. 25 112 1/16 4 1. 625 140 1/32 5 . 8125 168 1/64 6

Mass Time Fraction 52. 0 0 1 # of Half. Lives 0 28 1/2 1 56 1/4 2 84 1/8 3 112 1/16 4 140 1/32 5 168 1/64 6 . 8125

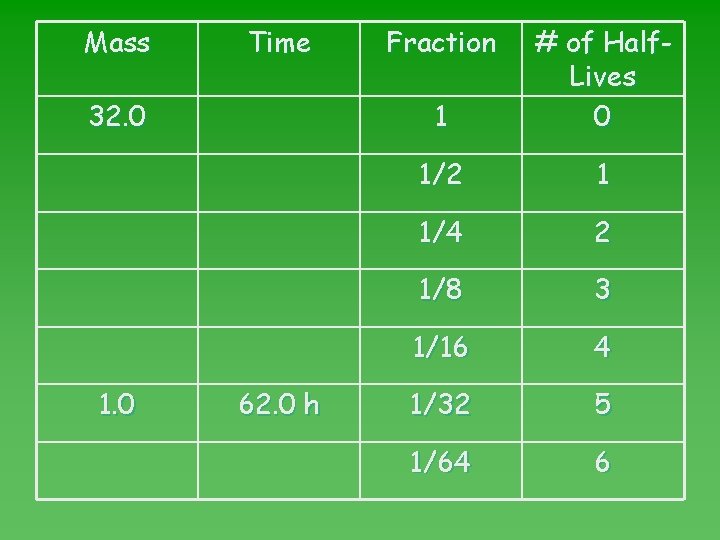
Problem 3 u After 62 hours, 1. 0 g remains unchanged from a sample of K-42. How much K-42 was in the original sample? u Look up HL of K-42 in table N: 12. 4 h u Calculate number of HL: 62 hours = 5 HL 12. 4 h/HL

Mass Time 32. 0 1. 0 62. 0 h Fraction 1 # of Half. Lives 0 1/2 1 1/4 2 1/8 3 1/16 4 1/32 5 1/64 6

Problem 4 u 80 milligrams of a radioactive substance decays to 10 milligrams in 30 minutes. u Calculate the HL. u NOTICE: They gave BOTH masses. 80 mg 40 mg 20 mg 10 mg Took 3 HL to decay to 10 mg

Problem 4 u 3 HL = 30 minutes so 1 HL = 10 minutes