Half Life Half Life Chart for Carbon14 Half

Half Life
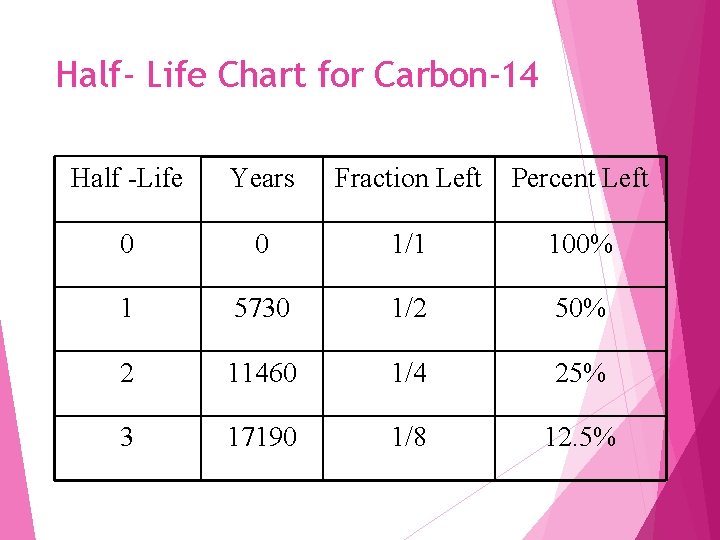
Half- Life Chart for Carbon-14 Half -Life Years Fraction Left Percent Left 0 0 1/1 100% 1 5730 1/2 50% 2 11460 1/4 25% 3 17190 1/8 12. 5%

Detecting Radiation Geiger Counter Alpha Gas or beta particles hit the tube in the tube becomes charged Creates electrical pulse and makes a “click” sound

Scintillation Counter Fluoresces when exposed to radiation The intensity of light can measure the amount of radiation Radon in basements can be detected with this. Radon gas builds up from uranium decaying in soil.

Half Life the time it takes for one half of a sample of a radioisotope to decay Every radioisotope has a different half-life length The longer a half life is the longer it takes to decay

The half life of C-14 is 5730 years. If a sample originally contained 3. 36 g of C-14 how much is present after 22, 920 years? Half -Life Time years sample 0 0 3. 36 g 1 5730 2 11460 3 17190 4 22920

algebra ln no nf 0. 693 t t 1/2 ln = natural log no = mass original sample nf= final sample mass t = total time t 1/2 = half life time

There are 3. 29 g of Iodine-126 remaining in the sample originally containing 26. 3 g of I-126. The half life of Iodine- 126 is 13 days. How old is the sample? Half -Life Time hours sample 0 0 26. 3 1 13 days 2 3 4 3. 29

Lets try the same problem with the algebra formula. There are 3. 29 g of Iodine-126 remaining in the sample originally containing 26. 3 g of I-126. The half life of Iodine- 126 is 13 days. How old is the sample? Ln 26. 3 = 0. 693 (x) 3. 29 2. 078 * 13 =. 693 ln (7. 99) =. 693 x 13 days x = 38. 98 days This is a more exact method. 13

Gold-191 has a half life of 12. 4 hours. After one day and 13. 2 hours, 10. 6 g of Au-191 remains in a sample. How much Au-191 was originally present? Half -Life Time hours 0 0 1 12. 4 sample 2 3 4 37. 2 10. 6 g

Gold-191 has a half life of 12. 4 hours. After one day and 13. 2 hours, 10. 6 g of Au-191 remains in a sample. How much Au-191 was originally present? Half -Life Time hours sample 0 0 84. 8 g 1 12. 4 42. 4 2 3 4 21. 2 37. 2 10. 6 g

Algebraic Gold-191 has a half life of 12. 4 hours. After one day and 13. 2 hours, 10. 6 g of Au-191 remains in a sample. How much Au-191 was originally present? Ln x = 0. 693(37. 2 hr) 10. 6 g x = 12. 4 hr 2 nd ln(2. 079) 10. 6 x = 7. 996 x = 84. 76 grams 10. 6 This is a more exact method. so take the antilog
- Slides: 12