Half Life Calculation of Radioactive Decay Atomic Physics





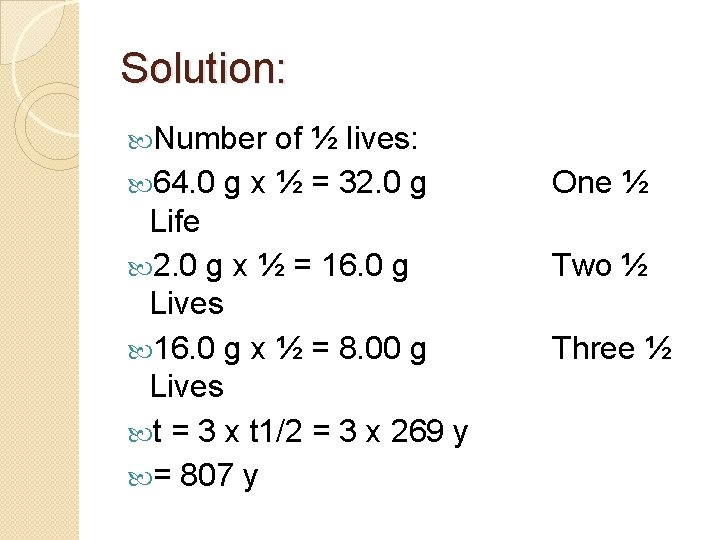

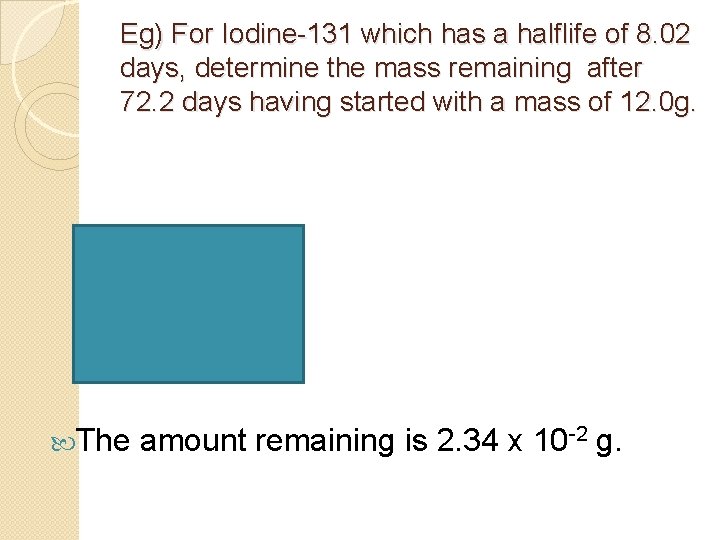







- Slides: 22

Half Life Calculation of Radioactive Decay Atomic Physics

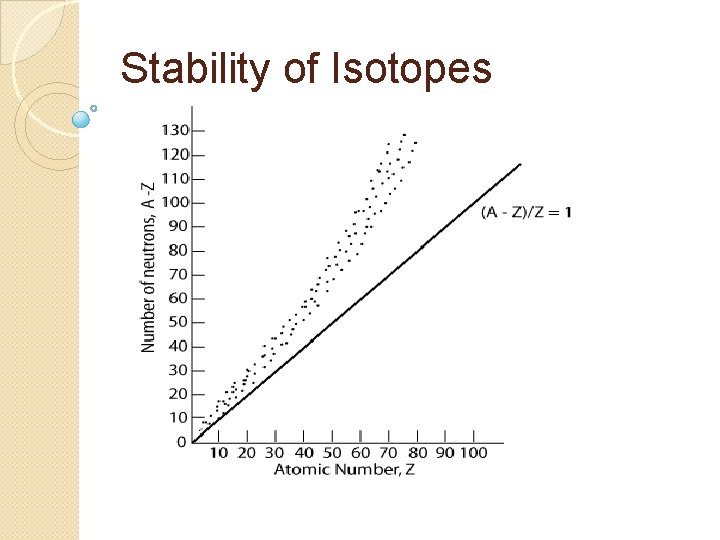
Stability of Isotopes

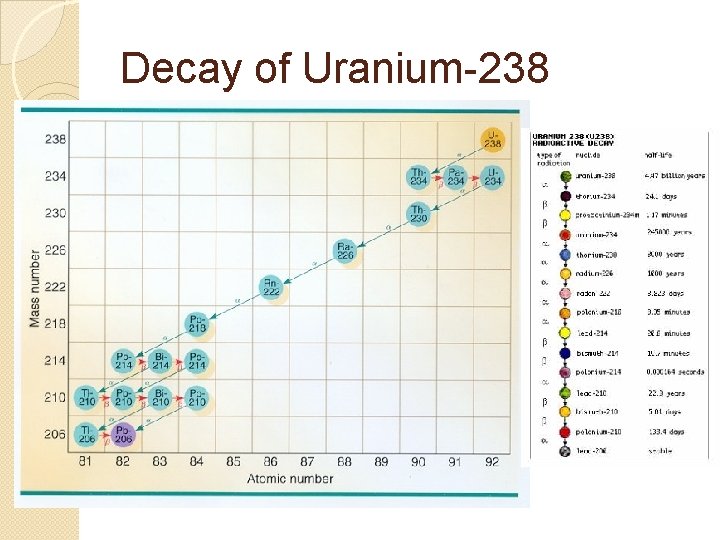
Decay of Uranium-238

Activity (A). . . is the number of nuclei in a given sample that will decay in a given time. Usually measured in decays/s, becquerels (Bq).

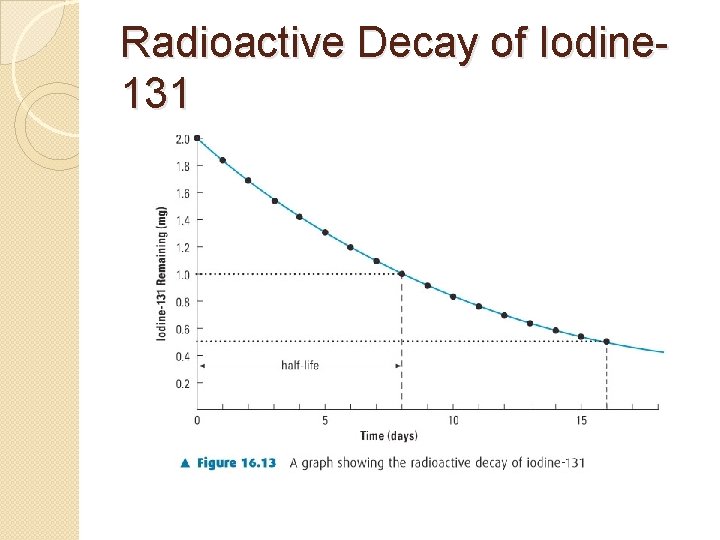
Half Life �Half-life of a radioactive isotope is the time taken for half of the atoms of an element to decay �Eg) each radioactive isotope has its own half life

Half Life http: //videos. howstuffworks. com/hsw/17819 -physicsthe-nature-of-radioactive-decay-video. htm . . . is the time required for half of the radioactive nuclei in a sample to decay. http: //www. youtube. com/watch? v=xh. Ot Example: Kur. Hayo Half-life for iodine-131 is 192 h. Initial mass of sample: 20 g After 192 h, 10 g of I-131 remains (the rest is decay products) After another 192 h, 5. 0 g of I-131 remains. http: //www. youtube. com/watch? v=6 Xzjm. EZO 4 o

Equation to determine the mass remaining after some time period

Example Argon-39 undergoes beta decay, with a half-life of 269 years. If a sample contains 64. 0 g of Ar-39, how many years will it take until only 8. 00 g of Ar 39 remain? Ignoring any other decays that may occur, what element is the rest of the sample transmuted into?
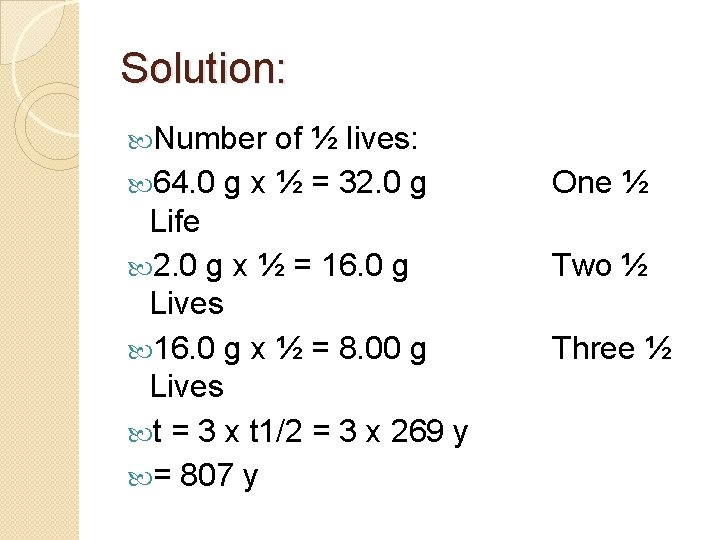
Solution: Number of ½ lives: 64. 0 g x ½ = 32. 0 g Life 2. 0 g x ½ = 16. 0 g Lives 16. 0 g x ½ = 8. 00 g Lives t = 3 x t 1/2 = 3 x 269 y = 807 y One ½ Two ½ Three ½

Solution: beta decay: Product: Potassium-39
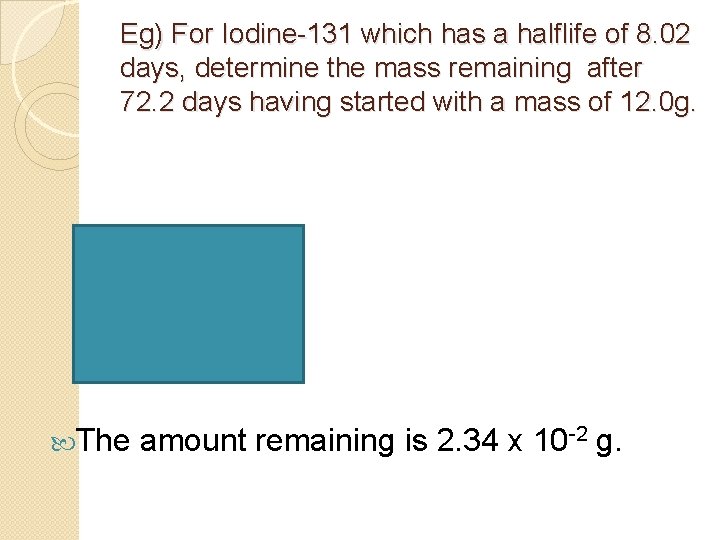
Eg) For Iodine-131 which has a halflife of 8. 02 days, determine the mass remaining after 72. 2 days having started with a mass of 12. 0 g. The amount remaining is 2. 34 x 10 -2 g.

Radioactive Decay of Iodine 131

Graph For Half. Life Calculation �Be able to interpret these graphs for half life time.

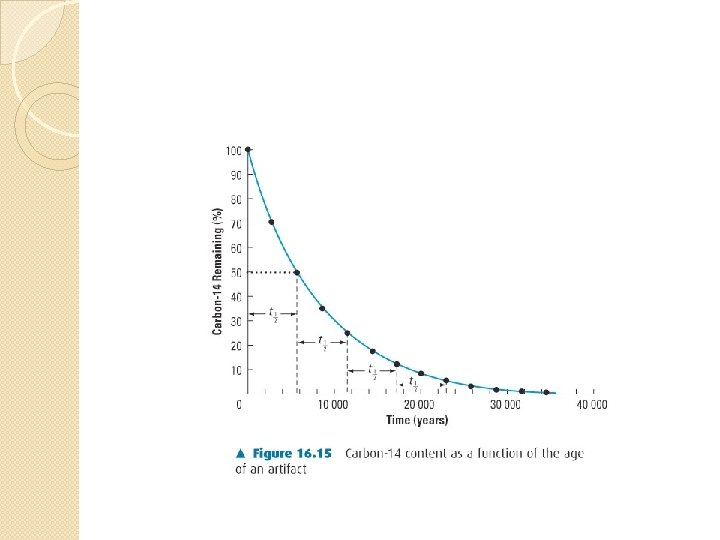
Radioactive Dating By measuring the relative amounts of different isotopes in a material, the age of the material can be determined. Carbon dating, using carbon-14, is the most well known example. Carbon-14 has a half-life of 5730 years.

Example A sample of bone contains one quarter of the C-14 normally found in bone. What is the bone’s approximate age?

Solution: The age of a sample with half the normal amount of C-14 would be approximately the same as the half life of C-14 (half the C-14 will have decayed). ¼ = ½ • ½ so ¼ is two half-lives. time = 2 • 5730 y = 11460 y


Why carbon dating works Carbon dating works for bone, and wood, etc. The proportion of C-14 to C-12 in the atmosphere is well known. A living tree will have the same proportion of C-14 to C-12 as it constantly absorbs carbon from the air.

Why carbon dating works When the tree dies (ie use the wood to make a tool) it no longer absorbs carbon. Decay of C-14 starts to reduce the amount of C-14 in the wood. Amount of stable C-12 remains constant.

Why carbon dating works When there is half the usual amount of C-14 remaining, the wood is about 5730 years old (one half life). Accurate measurements need to account for variations in proportion of C-14 to C-12 over the centuries. Carbon dating has been verified by comparing to known dates.

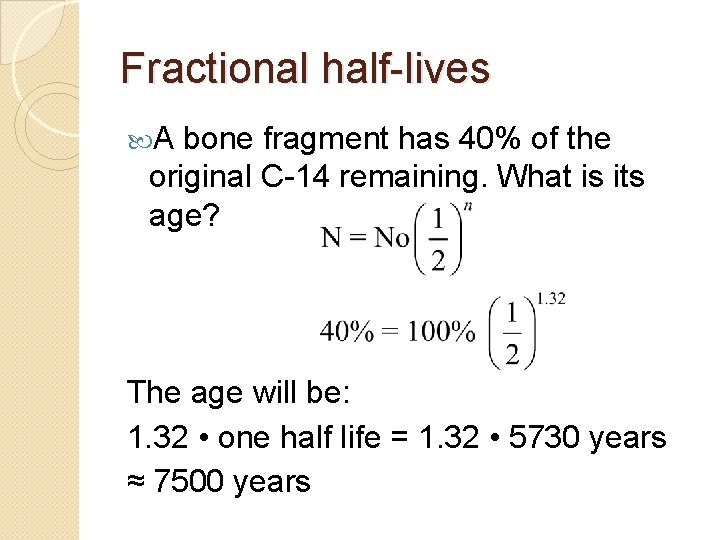
Fractional half-lives A bone fragment has 40% of the original C-14 remaining. What is its age? The age will be: 1. 32 • one half life = 1. 32 • 5730 years ≈ 7500 years

Why carbon dating works Carbon dating does not provide accurate results for materials older than about 50 000 years, or fairly recent materials. This is because there is either not enough C-14 left to accurately measure or not enough has decayed yet.