Half Life and Carbon Dating A Half Life

Half Life and Carbon Dating

A. Half Life � Half-life – the time it takes for ½ of a radioactive sample to decay � Half-life for a radioactive element is a constant rate of decay � Half-life differs for each radioactive substance
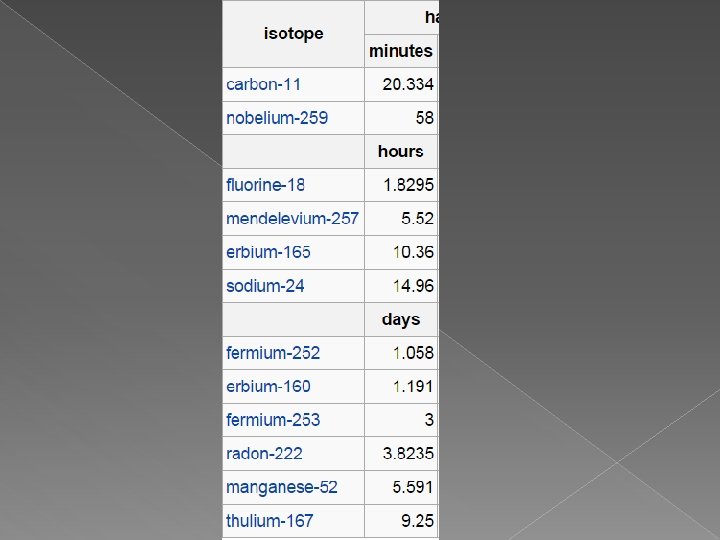

A. Half – Life Cont. � For example. . . strontium – 90 has a half-life of 29 years. If you start with 10 g of strontium – 90 today, how much will remain after 29 years?

B. Decay Curves � Decay curves show rate of decay for radioactive elements � Typically shows the relationship between half -life (y-axis) and percentage of original substance remaining (x-axis).
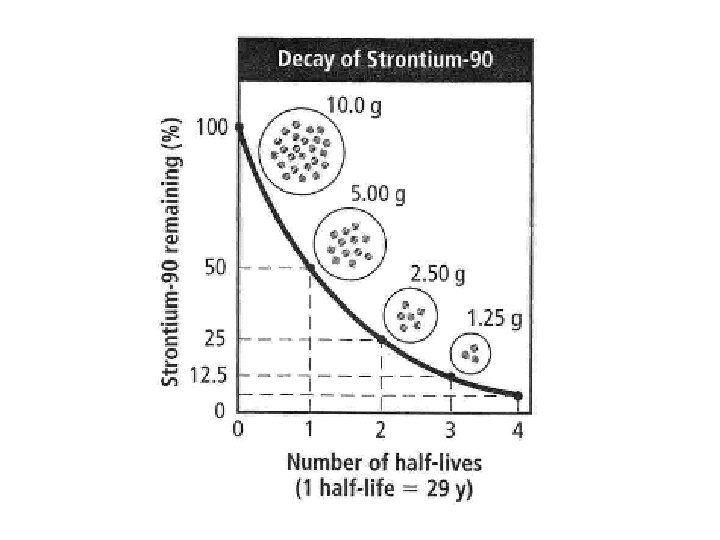

Half – Life Problems � If we start with 400 atoms of a radioactive substance, how many would remain after one half-life? _____ after two half-lives? _____ after three half-lives? ______ after four halflives? _____

The half-life of Zn-71 is 2. 4 minutes. If one had 100. 0 g at the beginning, how many grams would be left after 7. 2 minutes has elapsed?

Os-182 has a half-life of 21. 5 hours. How many grams of a 10. 0 gram sample would have decayed after exactly three half-lives?

How much time will be required for a sample of H-3 to lose 75% of its radioactivity? The halflife of tritium is 12. 26 years.

After 24. 0 days, 2. 00 milligrams of an original 128. 0 milligram sample remain. What is the half-life of the sample?

C. Carbon Dating � It can be difficult to determine the ages of objects by sight alone. › Radioactivity provides a method to determine age by measuring relative amounts of remaining radioactive material to stable products formed.
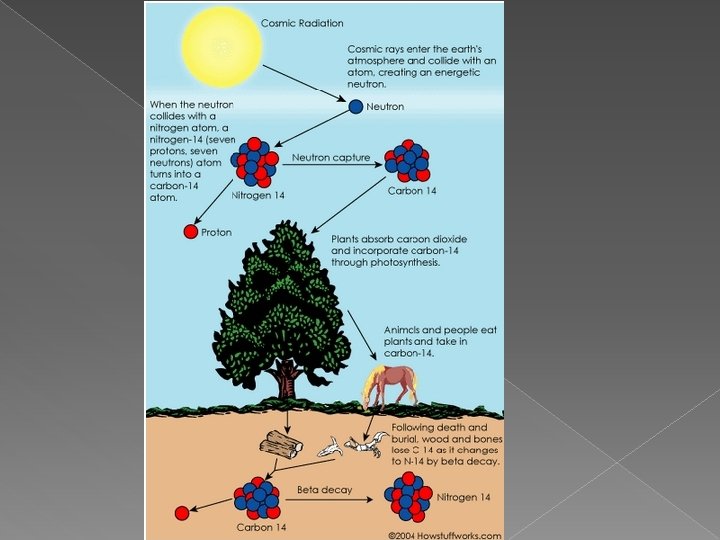
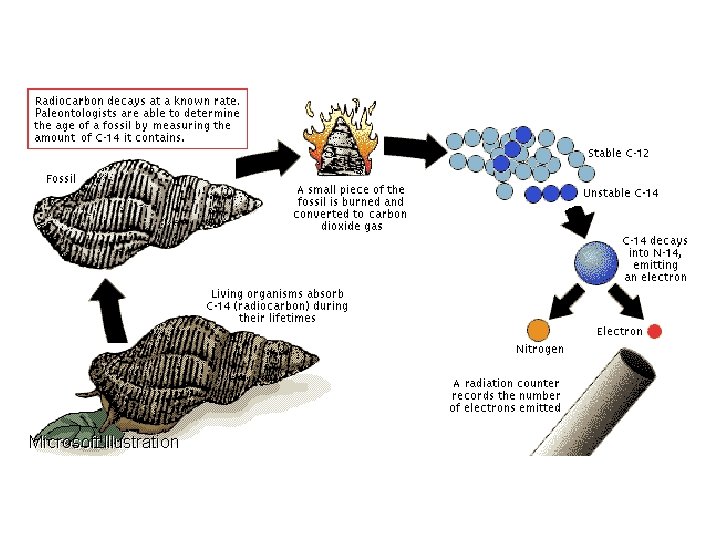

C. Carbon Dating Cont. • Carbon dating measures the ratio of carbon 12 and carbon-14. – Stable carbon-12 and radioactive carbon-14 exist naturally in a constant ratio. – When an organism dies, carbon-14 stops being created and slowly decays. • Carbon dating only works for organisms less than 50 000 years old.
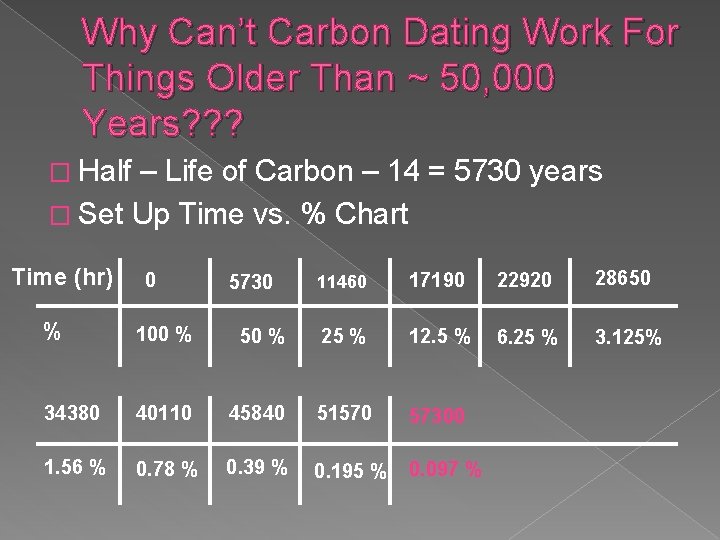
Why Can’t Carbon Dating Work For Things Older Than ~ 50, 000 Years? ? ? � Half – Life of Carbon – 14 = 5730 years � Set Up Time vs. % Chart Time (hr) 0 5730 11460 17190 22920 28650 6. 25 % 3. 125% % 100 % 50 % 25 % 12. 5 % 34380 40110 45840 51570 57300 1. 56 % 0. 78 % 0. 39 % 0. 195 % 0. 097 %

D. Applying Nuclear Reactions � Write the following Carbon Dating nuclear reactions: › Nitrogen – 14 undergoes neutron capture to become carbon – 14. › Carbon – 14 undergoes beta decay.
- Slides: 17