H secretion proximal tubulethick ascending Henle Proximal tubule
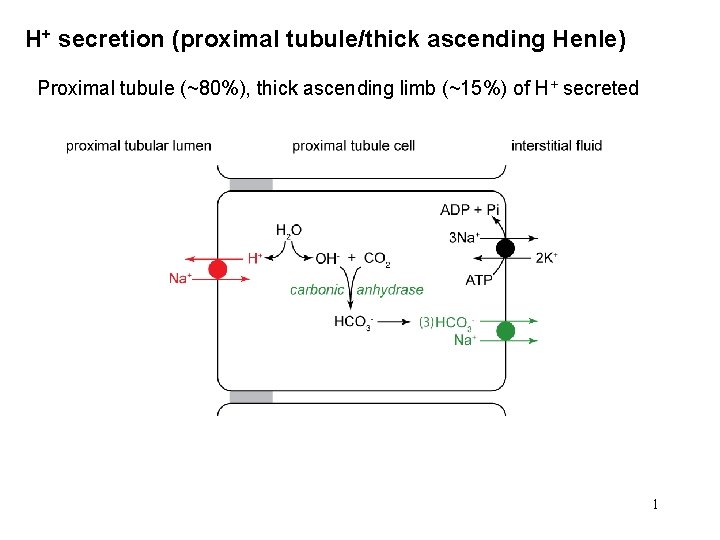
H+ secretion (proximal tubule/thick ascending Henle) Proximal tubule (~80%), thick ascending limb (~15%) of H+ secreted 1
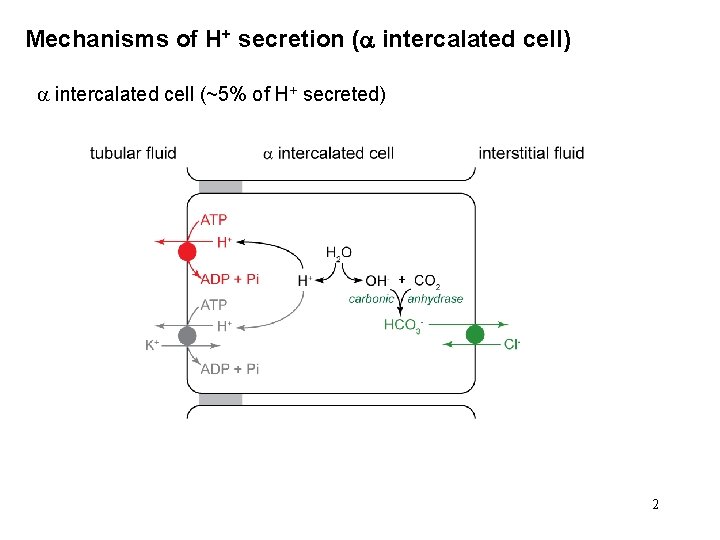
Mechanisms of H+ secretion ( intercalated cell) intercalated cell (~5% of H+ secreted) 2
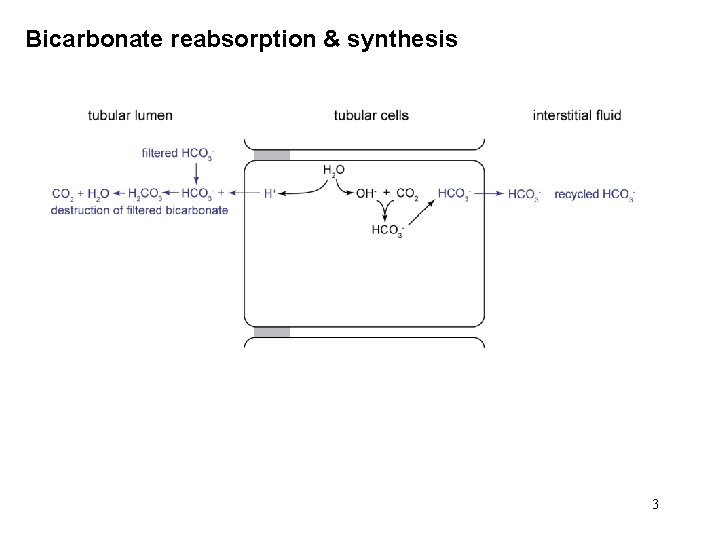
Bicarbonate reabsorption & synthesis 3
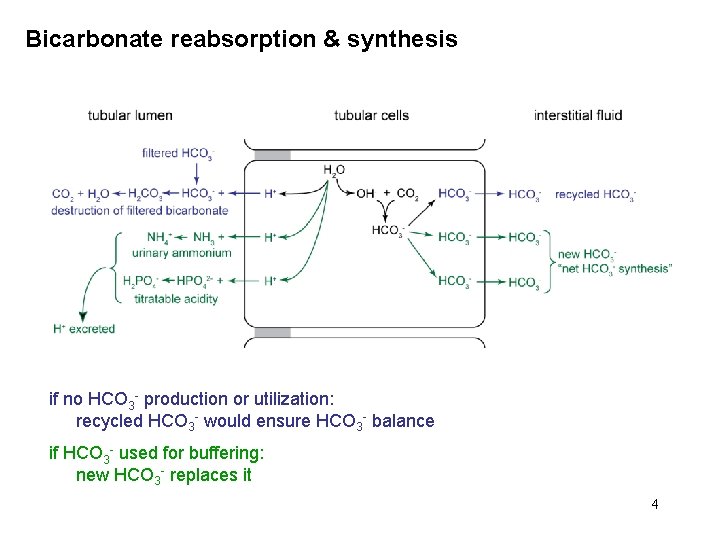
Bicarbonate reabsorption & synthesis if no HCO 3 - production or utilization: recycled HCO 3 - would ensure HCO 3 - balance if HCO 3 - used for buffering: new HCO 3 - replaces it 4

Quantitation of secreted H+ & reabsorbed HCO 3 - “Titratable acidity” = excreted H 2 PO 4 - & other urinary buffers “Urinary ammonium” = excreted NH 4+ 5

Important principles 1 H+ secreted = HCO 3 - reabsorbed H+ excreted = titratable acidity + urinary NH 4+ Most of the H+ secreted is used to “reclaim” filtered HCO 3 Filtered HCO 3 - is not reabsorbed as such; it is destroyed in tubule and resynthesized in tubular cell New HCO 3 - synthesized = H+ excreted = titratable acidity + urinary NH 4+ 6
![Important principles 2 Plasma [HCO 3 -] depends on the rate of renal H+ Important principles 2 Plasma [HCO 3 -] depends on the rate of renal H+](http://slidetodoc.com/presentation_image_h/ef86207284e4c733d98b3906e966db2e/image-7.jpg)
Important principles 2 Plasma [HCO 3 -] depends on the rate of renal H+ secretion plasma [HCO 3 -] (metabolic alkalosis) H+ secretion plasma [HCO 3 -] (metabolic acidosis) Healthy kidney maintains the constancy of plasma [HCO 3 -] by maintaining constancy of H+ secretion (irrespective of moderate acid or alkaline assaults) 7

Henderson Hasselbalch equation H 2 O + CO 2 H 2 CO 3 H+ + HCO 3 Looking at 2 nd half of equation: K (equilibrium constant) = [H+] x [HCO 3 -] [H 2 CO 3] Rearranging and taking negative logs: p. H = p. K + log 10 [HCO 3 -] [H 2 CO 3] Assuming the 1 st half of the equation is at equilibrium: [H 2 CO 3] = 0. 03 x Pa. CO 2 Then, the Henderson Hasselbalch equation is: p. H = p. K + log 10 [HCO 3 -] 0. 03 x Pa. CO 2 8

Why use the bicarbonate buffer system? Not ideal because: p. K 6. 1, and buffers are most effective around their p. Ks However: the body can regulate Pa. CO 2 & [HCO 3 -] independently and thus control p. H lungs regulate Pa. CO 2 & kidneys regulate [HCO 3 -] regulating p. H will alter all buffer systems (isohydric principle) clinically, we can measure p. H & Pa. CO 2, & calculate [HCO 3 -] 9
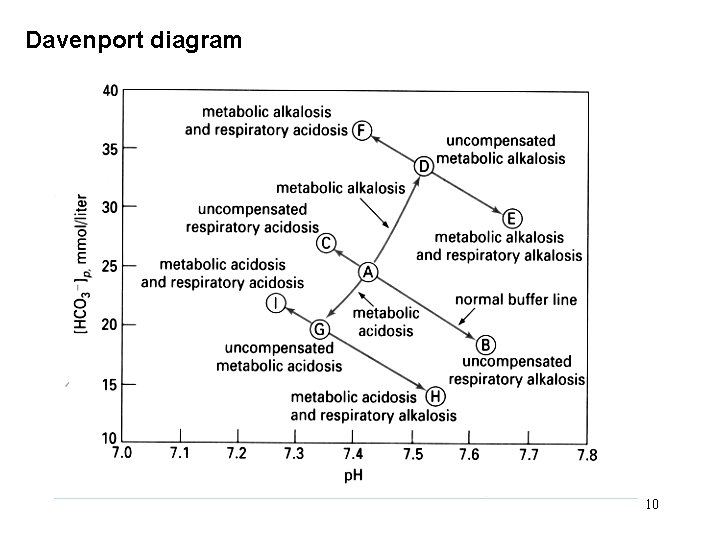
Davenport diagram 10

Interpreting acid base data (DRW AIM) Normal values: p. H 7. 35 – 7. 45, Pa. CO 2 33 – 44 mm Hg, HCO 3 - 22 – 28 m. Eq/L 1. Acidosis or alkalosis? p. H < 7. 35 = acidosis, p. H > 7. 45 = alkalosis you may have to revise that later, but start with this “overcompensation” does not occur 11

Metabolic or respiratory? 2 a. Is it metabolic acidosis (alkalosis)? Look at [HCO 3 -]; metabolic alkalosis, metabolic acidosis e. g. p. H 7. 25, [HCO 3 -] 14 m. Eq/L = Yes; metabolic acidosis e. g. p. H 7. 25, [HCO 3 -] 32 m. Eq/L = No; not metabolic acidosis 2 b. Is it respiratory acidosis (alkalosis)? Look at Pa. CO 2; respiratory acidosis, respiratory alkalosis e. g. p. H 7. 25, Pa. CO 2 64 mm Hg = Yes; respiratory acidosis e. g. p. H 7. 25, Pa. CO 2 26 mm Hg = No; not respiratory acidosis 12

Respiratory compensation 3. Respiratory compensation (metabolic disorders) Mechanism: p. H or p. H detected by carotid body & aortic arch chemoreceptors, ventilation rate Pa. CO 2; ventilation rate Pa. CO 2 Features: rapid response (minutes); no response suggests respiratory disorder relatively inadequate because Pa. CO 2 limits response i. e. metabolic acidosis AVR Pa. CO 2 AVR Pa. CO 2 > 55 mm Hg is respiratory acidosis (too high for compensation) 13

Metabolic compensation 3. Metabolic compensation (respiratory disorders) Mechanism: Pa. CO 2 or Pa. CO 2 causes or H+ secretion & HCO 3 reabsorption Features: buffering is not compensation [HCO 3 -] will by ~1 m. Eq/L for each 10 mm Hg Pa. CO 2 (buffering) slow response ~ 24 hrs for [HCO 3 -] (excretion) days for [HCO 3 -] (induction of glutaminase II) very effective (eventually) 14
![Terminology (compensation vs. mixed condition) p. H 7. 30, [HCO 3 -] 14 m. Terminology (compensation vs. mixed condition) p. H 7. 30, [HCO 3 -] 14 m.](http://slidetodoc.com/presentation_image_h/ef86207284e4c733d98b3906e966db2e/image-15.jpg)
Terminology (compensation vs. mixed condition) p. H 7. 30, [HCO 3 -] 14 m. Eq/L, Pa. CO 2 29 mm Hg is compensated metabolic acidosis, not (mixed) metabolic acidosis and respiratory alkalosis p. H 7. 34, [HCO 3 -] 32 m. Eq/L, Pa. CO 2 62 mm Hg is compensated respiratory acidosis, not (mixed) respiratory acidosis and metabolic alkalosis Compensation will disappear when the primary condition is treated; a mixed condition won’t 15
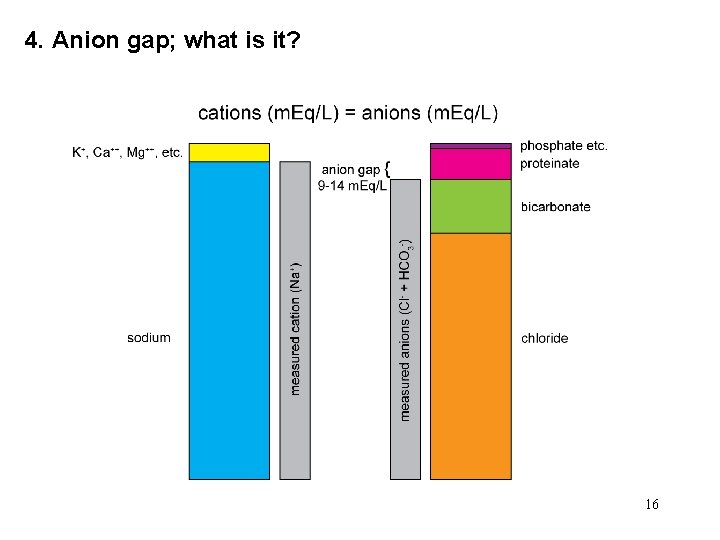
4. Anion gap; what is it? 16
![Anion gap; why does it change? The “gap” is [measured cations] - [measured anions] Anion gap; why does it change? The “gap” is [measured cations] - [measured anions]](http://slidetodoc.com/presentation_image_h/ef86207284e4c733d98b3906e966db2e/image-17.jpg)
Anion gap; why does it change? The “gap” is [measured cations] - [measured anions] 17

Significance of the anion gap Used for differential diagnosis of metabolic acidosis Hyperchloremic metabolic acidosis (normal anion gap) occurs when disorder is HCO 3 - loss, e. g. diarrhea, renal tubular acidosis, carbonic anhydrase inhibitors, Addison’s disease Normochloremic metabolic acidosis (increased anion gap) occurs when “non HCl” acids accumulate e. g. lactic acidosis, ketoacidosis, salicylate, methanol (formate), ethylene glycol (glycolate, oxalate), chronic renal failure (sulfate, phosphate, others) 18

Causes of respiratory acid base disorders Respiratory acidosis: opiates, sleep apnea, administration of O 2 to a “blue bloater” type of COPD, weakness of respiratory muscles, extreme obesity, pulmonary edema, asthma, pneumonia, pneumothorax Respiratory alkalosis: several respiratory diseases (pneumonia, interstitial fibrosis, pulmonary embolus), hyperventilation, liver failure, salicylate overdose, gram negative septicemia, mechanical ventilation 19

Causes of metabolic acid base disorders Metabolic acidosis (hyperchloremic, normal anion gap): diarrhea, carbonic anhydrase inhibitors, aldosterone deficiency, types I & II renal tubular acidosis Metabolic acidosis (normochloremic, increased anion gap): diabetic ketoacidosis, lactic acidosis, chronic renal failure, various poisonings (salicylate, methanol, ethylene glycol) Metabolic alkalosis: antacid ingestion, vomiting, nasogastric suction, loop or thiazide diuretics, aldosterone excess, vascular volume depletion, potassium depletion 20

Case F 1: acid base condition p. H 7. 58; Pa. CO 2 23 mm Hg; [HCO 3 -] 21 m. Eq/L 1. Look at p. H (is it acidosis or alkalosis? ) p. H = 7. 58 alkalosis 2. Look at HCO 3 - (is it metabolic alkalosis? ) HCO 3 - = 21 m. Eq/L (normal 22 -30) not metabolic alkalosis 3. Look at Pa. CO 2 (is it respiratory alkalosis? ) Pa. CO 2 = 23 mm. Hg (normal 35 -45) respiratory alkalosis 4. See if appropriate compensation has occurred: compensation for respiratory alkalosis is HCO 3 - excretion HCO 3 - = 21 mm. Hg (normal 22 -30) uncompensated i. e. acute respiratory alkalosis 21

Case F 1: questions Describe her acid base condition acute respiratory alkalosis Plausible causes for her acid base condition? hysterical hyperventilation Suggest causes of her dizziness Pa. CO 2 cerebral vasoconstriction Any evidence for ventilation/perfusion mismatch in pulmonary function? A-a O 2 difference = 123 -116 = 7 (i. e. OK) What is carpopedal spasm, and its possible cause? p. H Ca++ binding to albumin free [Ca++] more negative threshold for muscle cell depolarization ( Na+ channel sensitivity) hypocalcemic tetany 22

Case F 2: acid base condition p. H 7. 29; Pa. CO 2 26 mm Hg; [HCO 3 -] 12 m. Eq/L 1. Look at p. H (is it acidosis or alkalosis? ) p. H = 7. 29 acidosis 2. Look at HCO 3 - (is it metabolic acidosis? ) HCO 3 - = 12 m. Eq/L (normal 22 -30) metabolic acidosis 3. Look at Pa. CO 2 (is it respiratory acidosis? ) Pa. CO 2 = 26 mm. Hg (normal 35 -45) not resp. acidosis 4. See if appropriate compensation has occurred: compensation for metabolic acidosis is hyperventilation Pa. CO 2 = 26 mm. Hg (normal 35 -45); partial compensation 5. Anion gap = 142 - (100 + 12) = 30 (increased) i. e. partially compensated normochloremic metabolic acidosis 23

Case F 2: questions Total CO 2 (CO 2 content)? CO 2 released when venous plasma is acidified equals [HCO 3 -] + 0. 03 x P. CO 2 (units m. Eq/L) Describe his acid base condition partially compensated normochloremic metabolic acidosis (increased anion gap) Plausible causes for his acid base condition? normochloremic metabolic acidosis diabetic ketoacidosis: [glucose] 650 mg/d. L alcoholic ketoacidosis: starvation, GNG, lipolysis lactic acidosis: bp hypoperfusion Anion gap? ketoacids (diabetes, alcohol), lactate (shock, hypoxemia) 24

Case F 2: questions What precautions with his therapy? Aims: volume repletion, insulin, p. H correction HCO 3 -, insulin & osmolality causes K+ to enter cells (next slide) give supplemental intravenous K+ 25

Regulation of K+ distribution Normally, 98% of total body K+ is intracellular Factor Effect on [K+]extracellular metabolic, respiratory acidosis (except organic acidosis) H+ influx K+ efflux [K+]ec metabolic, respiratory alkalosis H+ efflux K+ influx [K+]ec insulin, aldosterone Na+/K+ ATP-ase [K+]ec exercise K+ efflux [K+]ec hyperosmolality (e. g. hyperglycemia) K+ efflux [K+]ec 26

Case F 3: acid base condition p. H 7. 05; Pa. CO 2 45 mm Hg; [HCO 3 -] 12 m. Eq/L 1. Look at p. H (is it acidosis or alkalosis? ) p. H = 7. 05 acidosis 2. Look at HCO 3 - (is it metabolic acidosis? ) HCO 3 - = 12 m. Eq/L (normal 22 -30) metabolic acidosis 3. Look at Pa. CO 2 (is it respiratory acidosis? ) Pa. CO 2 = 45 mm. Hg (normal 35 -45) poss. resp. acidosis 4. See if appropriate compensation has occurred: compensation for metabolic acidosis is hyperventilation Pa. CO 2 = 45 mm. Hg (normal 35 -45) respiratory acidosis 5. Anion gap = 140 - (116 + 12) = 12 (unchanged) i. e. mixed hyperchloremic metabolic acidosis (normal anion gap) and respiratory acidosis 27

Case F 3: questions 1 Describe her original acid base condition mixed hyperchloremic metabolic acidosis and respiratory acidosis Plausible causes for her acid base conditions hyperchloremic metabolic acidosis possibilities include: diarrhea, carbonic anhydrase (CA) inhibitors, aldosterone deficiency, & renal tubular acidosis (types I & II) aldosterone deficiency would have [K+], hers is 1. 4 m. Eq/L; history rules out diarrhea, CA inhibitors, RTA, urine p. H 7. 0 type 1 respiratory acidosis: hypokalemia (1. 4 m. Eq/L) decreases excitability of diaphragm 28

Renal tubular acidosis In general: proximal/thick ascending limb Na+/H+ exchager function is to destroy (& reabsorb) filtered HCO 3 intercalated H+ ATPase is to acidify urine urinary NH 4+ & titratable acidity thus synthesizing new HCO 3 Proximal RTA (type 2) activity of proximal/thick AL Na+/H+ exchanger serum [HCO 3 -] proximal HCO 3 - load; serum [HCO 3 -] stabilizes collecting duct H+ ATPase can acidify urine & synthesize “new” HCO 3 urine p. H within normal range Distal RTA (type 1) activity of collecting duct H+ ATPase, or back diffusion of H+ inability of synthesize “new” HCO 3 - by acidifying urine progressive depletion of serum [HCO 3 -] urine p. H at alkaline end of range although body p. H 29

Case F 3: questions 2 Urine p. H towards alkaline end of range (p. H 4. 5 -7. 5)? type 1 RTA is failure of collecting duct H+ secretion (acidifying region; generating new HCO 3 -) Increased [HCO 3 -] in emergency room ( = +13 m. Eq/L)? buffering (1 m. Eq/L per 10 mm Hg Pa. CO 2); Pa. CO 2 = +94 mm Hg, i. e. ~10 m. Eq/L [HCO 3 -] Role of hypokalemia? hyperpolarization muscle weakness (diaphragm) 30

Case F 3: questions 3 Cause of hypokalemia (1. 4 m. Eq/L)? “Holy Grail” of collecting duct function (Na+ for K+ and/or H+) next slide Urinary NH 4+ & titratable acidity? failure to acidify urine & synthesize “new” HCO 3 Kroger’s chemical? daily Na. HCO 3 (baking soda) would work she was given polycitra-K (K citrate & citric acid) citrate metabolized as citric acid, removing H+ then, OH- + CO 2 HCO 3 - 31
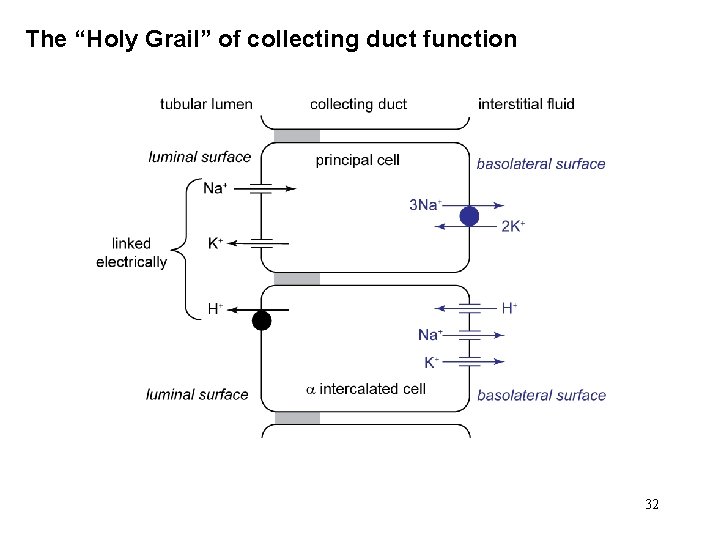
The “Holy Grail” of collecting duct function 32

Case F 4: acid base condition p. H 7. 44; Pa. CO 2 65 mm Hg: [HCO 3 -] 43 m. Eq/L 1. Look at p. H (is it acidosis or alkalosis? ) p. H = 7. 44 it’s close, but on alkalotic side of normal 2. Look at HCO 3 - (is it metabolic alkalosis? ) HCO 3 - = 43 m. Eq/L (normal 22 -30) metabolic alkalosis 3. Look at Pa. CO 2 (is it respiratory alkalosis? ) Pa. CO 2 = 65 mm. Hg (normal 35 -45) not resp. alkalosis 4. See if appropriate compensation has occurred: compensation for metabolic alkalosis would be hypoventilation Pa. CO 2 yes, but Pa. CO 2 too high for compensation respiratory acidosis i. e. mixed metabolic alkalosis and respiratory acidosis 33

Case F 4: questions Describe his acid base condition mixed metabolic alkalosis and respiratory acidosis Plausible causes for his acid base condition? metabolic alkalosis: diuretic excess, mild hypokalemia respiratory acidosis: COPD with hypoventilation (CO 2 insensitivity; “blue bloater”) Ventilation perfusion mismatch? A-a O 2 difference = 71 -45 = 26 mm Hg (not bad for age 56 & COPD); hypoventilation & V/Q mismatch hypoxemia Cause of mild hypokalemia (3. 1 m. Eq/L)? furosemide is K+ wasting diuretic Treatment with 100% O 2? hypercapnia; removal of O 2 drive 34
- Slides: 34