H 2 S Interactions with Palladium Alloy Membrane

H 2 S Interactions with Palladium Alloy Membrane Surfaces Bret Howard, Bryan Morreale US DOE NETL James Miller, Casey O’Brien NETL Research Associates, Carnegie Mellon University Dominic Alfonso, Michael Ciocco NETL Site Support Contractors, Parsons 2008 NHA Conference, Sacramento, CA

Global Energy · The US and other developed nations face several challenges: - Growing dependence on foreign oil · US imports >20 million barrels oil/day (>60%) - Environmental concerns (air and water quality) · Sources include: power plants, refineries, transportation - Greenhouse gas emissions (global warming) · CO 2, NO 2, CH 4 · To deal with these challenges, the US is implementing energy programs to promote the development of diverse energy resources - H 2, solar, wind, geothermal, biomass, etc.

Co-production of Hydrogen and Power from Coal Using Membrane Technologies CO 2 for sequestration Air H 2 Separation Membrane Candle Filter ITM Oxygen Syngas Compressor Combustor Air H 2 Expander HRSG Coal Transport Desulfurizer Dry Feed Gasifier CO 2 Electricity Sulfur By-Product Slag/Fines Solids · However, coal will remain a critical energy resource for the near future. Therefore, improved utilization methods must be developed and deployed.

Advantages of Membrane Reactor Integration · Enhanced plant efficiency - Eliminate the need to cool syngas stream - Lower steam/CO ratio (~1 – 2 vs. 10) - Lower cost of producing hydrogen · Eliminate energy intensive PSA - Membrane can produce ultra-high purity H 2 stream · Eliminate catalyst requirement - Membrane can be catalytically active for water-gas shift reaction · Maximum CO 2 Recovery - CO 2 captured at high pressure, eliminating/reducing the need for recompression · Increased Efficiency of Shift Conversion to H 2

Pd Membrane Purification of Hydrogen · Pd membranes have been extensively studied for hydrogen purification - Infinite H 2 selectivity - High H 2 permeability - Resistance to high T and corrosion · However, Pd exhibits certain characteristics which limit its use in a post gasifier membrane reactor/purifier - Poisoning (degradation of performance) - Lack of mechanical strength (failure) · Pd is a soft metal - thick walled or supported membranes are required, reducing H 2 flux

Membrane Requirements for Gasification Applications · A viable membrane candidate needs to be identified that is robust enough to operate at post-gasifier conditions: - In the presence of major and minor gasification constituents · CO, CO 2, H 2 O, H 2 S, NH 3, HCl, Hg, etc. - Extreme conditions · T up to 1000°C, P up to 7 MPa (1000 psig) · Pd-Cu alloys have received increased interest in recent years: - Minimum reduction in permeability with the addition of Cu - “Tolerance” of H 2 S Ag Knapton, Plat. Met. Rev. , 21 (1977) 44 -50 Mc. Kinley, Nitro, US Patent, 3, 350, 845 (1967). Au Ce Cu Y

Research Objectives · Study the H 2 permeability of palladium-copper alloys at post-gasifier conditions in order to: - Develop a thorough performance database both in the absence and presence of H 2 S - Develop a good understanding of the Pd-Cu-S system · Utilize surface chemistry and computational techniques to: - Understand atomic-level interactions Goal: Integrate macro-scale and nano-scale results to guide development of improved membrane technologies

Studies Performed · Test performance of Pd-Cu alloys - Clean H 2 and 1000 ppm H 2 S/H 2 · · Pd, 80 Pd-Cu, 60 Pd-Cu, 53 Pd-Cu 300, 450, 635°C – 120 hour duration · Designed and tested other Pd-based binary and ternary alloys - 14 other alloys investigated · Measure sulfide corrosion rates for Pd-Cu alloys - Coupon tests · HT WGS membrane reactor studies - H 2 S effects studied · Surface chemistry studies – 80 Pd-Cu - Understand atomic-level interactions · Computational studies - Pd 4 S – H 2 surface interactions
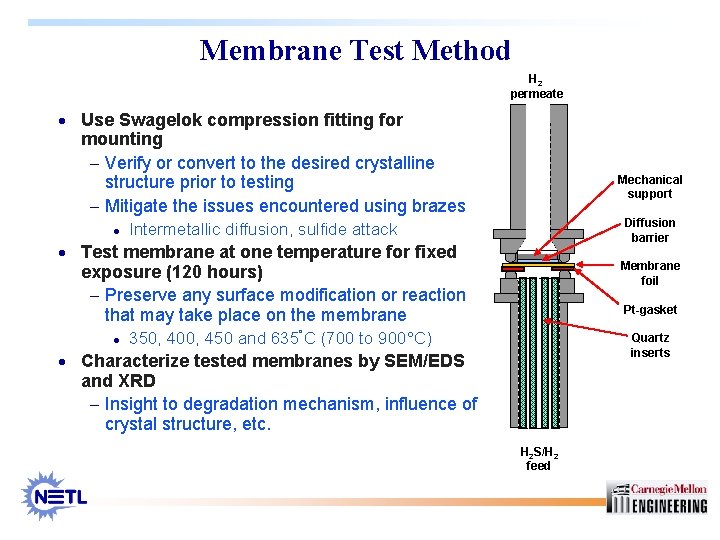
Membrane Test Method H 2 permeate · Use Swagelok compression fitting for mounting - Verify or convert to the desired crystalline structure prior to testing - Mitigate the issues encountered using brazes · Mechanical support Diffusion barrier Intermetallic diffusion, sulfide attack · Test membrane at one temperature for fixed Membrane foil exposure (120 hours) - Preserve any surface modification or reaction that may take place on the membrane · Pt-gasket 350, 400, 450 and 635°C (700 to 900°C) Quartz inserts · Characterize tested membranes by SEM/EDS and XRD - Insight to degradation mechanism, influence of crystal structure, etc. H 2 S/H 2 feed
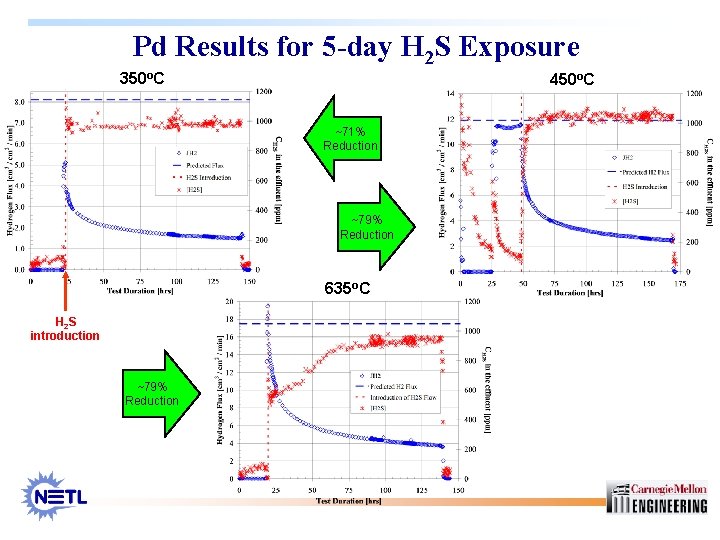
Pd Results for 5 -day H 2 S Exposure 350 o. C 450 o. C ~71% Reduction ~79% Reduction 635 o. C H 2 S introduction ~79% Reduction

SEM/EDS & XRD Analysis of Pd after H 2 S Exposure S Pd ~9 mm Pd 4 S 350 o. C Pd 450 o. C ~15 mm Pd 635 o. C S Pd 4 S S ~30 mm Pd 4 S
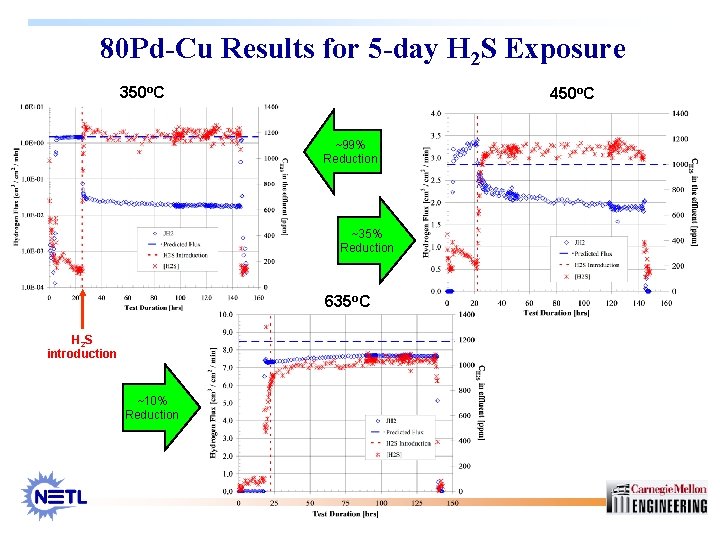
80 Pd-Cu Results for 5 -day H 2 S Exposure 350 o. C 450 o. C ~99% Reduction ~35% Reduction 635 o. C H 2 S introduction ~10% Reduction

SEM/EDS & XRD Analysis of 80 Pd-Cu after H 2 S Exposure Pd Cu S ~5 mm Pd 4 S Pd 13 Cu 3 S 7 B 2 alloy 350 o. C Pd Cu ~2 mm 450 o. C Pd 635 o. C S Cu Pd 4 S Pd 13 Cu 3 S 7 B 2 alloy fcc alloy
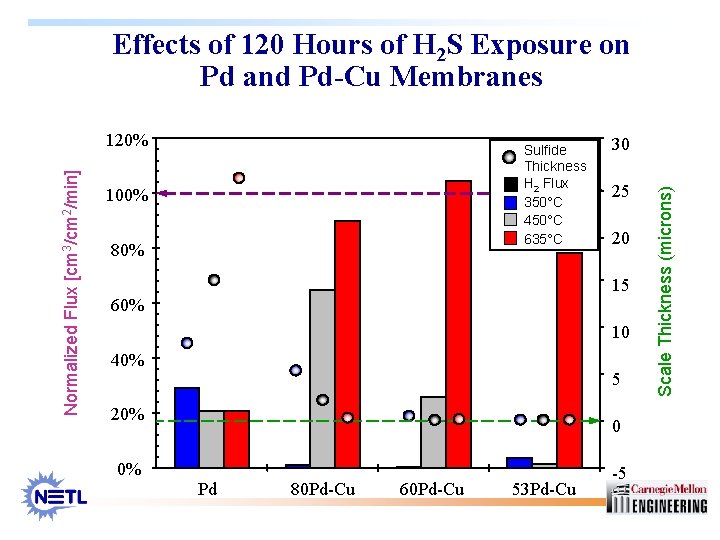
Effects of 120 Hours of H 2 S Exposure on Pd and Pd-Cu Membranes Sulfide Thickness H 2 Flux 350°C 450°C 635°C 100% 80% 30 25 20 15 60% 10 40% 5 20% 0% 0 Pd 80 Pd-Cu 60 Pd-Cu 53 Pd-Cu -5 Scale Thickness (microns) Normalized Flux [cm 3/cm 2/min] 120%
Impact of S on Pd-Cu Membrane Performance · Catalytic poisoning: - Immediate - Usually partially reversible - No significant Pd-S compound formation - Characteristic of alloy at low temperatures · Corrosive decay: - “Turns on” slowly - Associated with formation of Pd 4 S scale - Flux never = 0 - Characteristic of pure Pd
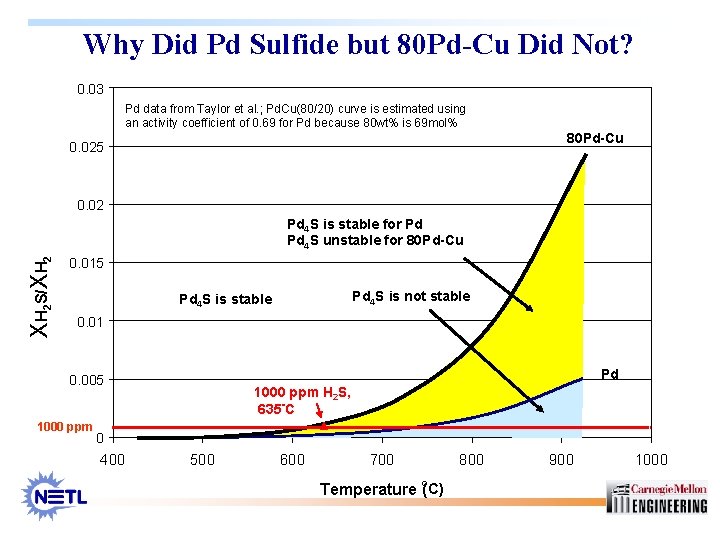
Why Did Pd Sulfide but 80 Pd-Cu Did Not? 0. 03 Pd data from Taylor et al. ; Pd. Cu(80/20) curve is estimated using an activity coefficient of 0. 69 for Pd because 80 wt% is 69 mol% 80 Pd-Cu 0. 025 0. 02 XH 2 S/XH 2 Pd 4 S is stable for Pd Pd 4 S unstable for 80 Pd-Cu 0. 015 Pd 4 S is not stable Pd 4 S is stable 0. 01 Pd 0. 005 1000 ppm H 2 S, 635°C 0 400 500 600 700 800 o Temperature ( C) 900 1000
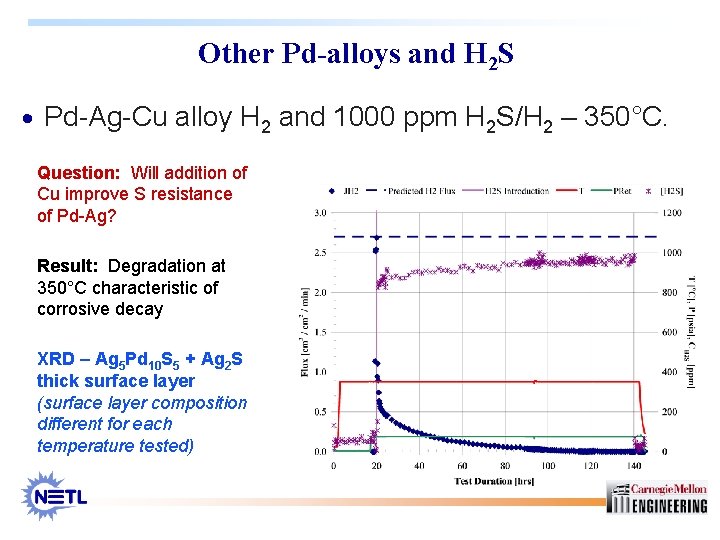
Other Pd-alloys and H 2 S · Pd-Ag-Cu alloy H 2 and 1000 ppm H 2 S/H 2 – 350°C. Question: Will addition of Cu improve S resistance of Pd-Ag? Result: Degradation at 350°C characteristic of corrosive decay XRD – Ag 5 Pd 10 S 5 + Ag 2 S thick surface layer (surface layer composition different for each temperature tested)

Surface Chemistry · The surface composition of an alloy is usually not the same as the bulk - Must be understood and controlled for design of practical devices Critical to catalytic activity Can change as a function of temperature Can be influenced by contaminants – bulk and surface Goals of surface study: • Quantify/understand surface segregation in the Pd-Cu system, both in clean H 2 and with a S-contaminant • Consider bridges between “surface chemistry” and “membrane performance”

Surface Analysis Approach · X-ray photoelectron spectroscopy (XPS) for composition of the “near-surface region” · Low-energy ion spectroscopy (LEIS) for composition of the top layer hv e He+ Pd-Cu composition used in this study: Polycrystalline 80 wt% Pd-Cu (~Pd 70 Cu 30)
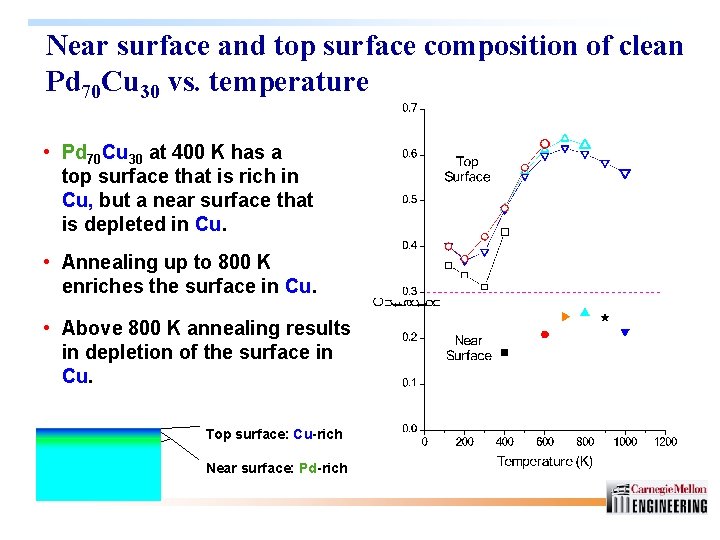
Near surface and top surface composition of clean Pd 70 Cu 30 vs. temperature • Pd 70 Cu 30 at 400 K has a top surface that is rich in Cu, but a near surface that is depleted in Cu. • Annealing up to 800 K enriches the surface in Cu. • Above 800 K annealing results in depletion of the surface in Cu. Top surface: Cu-rich Near surface: Pd-rich
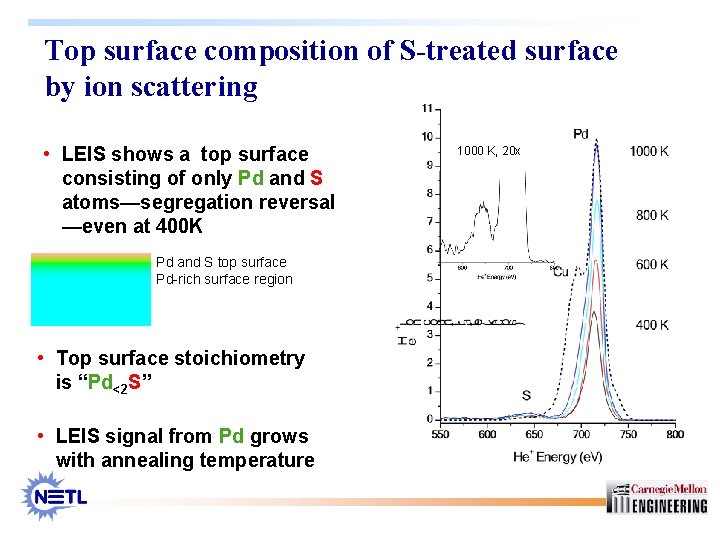
Top surface composition of S-treated surface by ion scattering • LEIS shows a top surface consisting of only Pd and S atoms—segregation reversal —even at 400 K Pd and S top surface Pd-rich surface region • Top surface stoichiometry is “Pd<2 S” • LEIS signal from Pd grows with annealing temperature 1000 K, 20 x
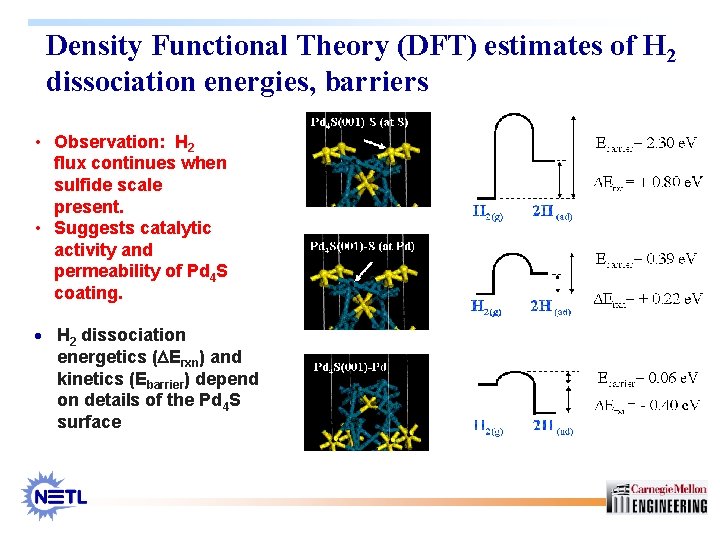
Density Functional Theory (DFT) estimates of H 2 dissociation energies, barriers • Observation: H 2 flux continues when sulfide scale present. • Suggests catalytic activity and permeability of Pd 4 S coating. · H 2 dissociation energetics (DErxn) and kinetics (Ebarrier) depend on details of the Pd 4 S surface

Summary · Pd-Cu alloys exhibit two general degradation modes in the presence of H 2 S - Catalytic poisoning - Corrosive decay · Gas species concentrations throughout a membrane system must be considered - As H 2 is removed [H 2 S] will increase · Surface chemistry and computational modeling can be used to probe atomic-level effects - Provide guidance in membrane design

Current Group Members Dr. Bryan Morreale Dr. Bret Howard U. S. DOE - NETL Dr. Michael Ciocco Dr. Dominic Alfonso Mr. Nicolas Means & technical staff Parsons, NETL Support Contractors Dr. Robert Enick Ms. Katie Barillas NETL Research Fellows, University of Pittsburgh Dr. Andrew Gelman Dr. James Miller Mr. Casey O’Brien Mr. William Michalak NETL Research Fellows, Carnegie-Mellon University Mr. Ian Wlodarczyk NETL Co-op Student Researchers, University of Pittsburgh
- Slides: 24