Gynecological infections Gebre K Tseggay M D Normal

Gynecological infections Gebre K. Tseggay, M. D.

Normal Vaginal Flora n n Dominated by lactobacilli Lactobacilli convert glucose to lactic acid, to maintain an acidic vaginal p. H of 3. 8 to 4. 2. This acidic environment inhibits the overgrowth of bacteria and other organisms with pathogenic potential. Some lactobacilli also produce hydrogen peroxide (H 2 O 2), a potential microbicide. After onset of sexual activity, increase in Gardnerella vaginalis, lactobacilli, mycoplasmas, ureaplasmas is seen.

BACTERIA ENDOGENOUS TO THE LOWER GENITALTRACT GRAM POSITIVE GRAM NEGATIVE Lactobacillus acidophilus Escherichia coli Corynebacterium Enterobacter cloacae spp Gardnerella vaginalis Staphylococcus epidermidis Klebsiella Streptococci Morganella Enterococcus faecalis Proteus Peptococcus Bacteroides Peptostreptococcus Fusobacterium Prevotella modified from Schlossberg, CTID 2001

Vaginitis Most common causes include: n Vulvovaginal Candidiasis (VVC) n Bacterial Vaginosis (BV) n Trichomoniasis *In some cases the etiology may be mixed

VAGINITIS SYMPTOMS n Often non-specific: n Abnormal discharge n Vulvovaginal irritation n Vulvar itching n Odor

VAGINITIS DIAGNOSIS n n n History Visual inspection Appearance of vaginal discharge: color, viscosity, adherence to vaginal walls, odor Collection of specimen Diagnostic tests: n n Vaginal p. H: determine vaginal p. H with narrow-range p. H paper Whiff test: assessment of a fishy odor after application of 10% KOH to wet mount KOH (wet mount): wet mount of discharge with 10% KOH Na. Cl (wet mount): wet mount of discharge with 0. 9% normal saline

VAGINITIS DIAGNOSIS Other tests: n Cultures: not used routinely, but are available for both T. vaginalis and Candida spp. n New tests for BV (commercially available) : n n n Fem Exam Test Card™: p. H and amines Fem Exam vaginalis PIP Activity Test Card™: detects enzyme breakdown from G. vaginalis DNA probe for 3 organisms (T. vaginalis, C. albicans, and G. vaginalis): sensitivity, specificity, and clinical utility are under investigation.

VULVOVAGINAL CANDIDIASIS Not considered to be STD n Caused by overgrowth of Candida species (Candida species are normal flora of vagina) n 80 -90% caused by C. albicans. n Non-albicans candida play increasing role n

VULVOVAGINAL CANDIDIASIS RISK FACTORS n n n n n Uncontrolled DM Corticosteroid therapy Antimicrobial therapy (oral, parental, topical) Poor hygiene Estrogen therapy High-dose estrogen contraceptives Pregnancy IUD HIV infection § § § n Sponge Nonoxynol-9 (? ) Diaphragm (? ) Increased frequency of coitus "Candy binge“ Women frequenting STD clinics Tight-fitting synthetic underclothing But, most episodes of vulvovaginal candidiasis occur in the absence of a recognizable precipitating factors

VULVOVAGINAL CANDIDIASIS CLASSIFICATION Uncomplicated Complicated Sporadic, infrequent Mild-to-moderate Likely C albicans Non-immunocomprised Recurrent Severe Non-albicans Diabetes, pregnancy, immunosuppression
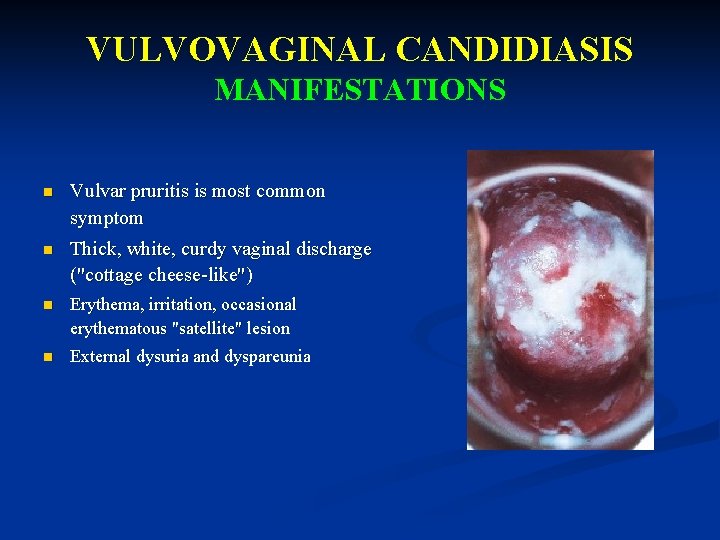
VULVOVAGINAL CANDIDIASIS MANIFESTATIONS n Vulvar pruritis is most common symptom n Thick, white, curdy vaginal discharge ("cottage cheese-like") n Erythema, irritation, occasional erythematous "satellite" lesion n External dysuria and dyspareunia

VULVOVAGINAL CANDIDIASIS DIAGNOSIS Clinical n p. H normal (<4. 5) n Whiff test negative n Fungal stain positive n n 30% may have a negative fungal stain n Severity does not depend on No. yeasts present

Regimens for the Treatment of Vulvovaginal Candidiasis n n Intravaginal agents: n Butoconazole 2% cream, 5 g intravaginally for 3 days† n Butoconazole 2% sustained release cream, 5 g single intravaginally application n Clotrimazole 1% cream 5 g intravaginally for 7 -14 days† n Clotrimazole 100 mg vaginal tablet for 7 days n Clotrimazole 100 mg vaginal tablet, 2 tablets for 3 days n Clotrimazole 500 mg vaginal tablet, 1 tablet in a single application n Miconazole 2% cream 5 g intravaginally for 7 days† n Miconazole 100 mg vaginal suppository, 1 suppository for 7 days† n Miconazole 200 mg vaginal suppository, 1 suppository for 3 days† n Nystatin 100, 000 -unit vaginal tablet, 1 tablet for 14 days n Tioconazole 6. 5% ointment 5 g intravaginally in a single application† n Terconazole 0. 4% cream 5 g intravaginally for 7 days n Terconazole 0. 8% cream 5 g intravaginally for 3 days n Terconazole 80 mg vaginal suppository, 1 suppository for 3 days Oral agent: n Fluconazole 150 mg oral tablet, 1 tablet in a single dose Note: The creams and suppositories in this regimen are oil-based and may weaken latex condoms and diaphragms. Refer to condom product labeling for further information. † Over-the-counter (OTC) preparations

RECURRENT VULVOVAGINAL CANDIDIASIS Four or more symptomatic episodes/year n Usually NOT from resistance to antifungals n Diabetes mellitus or immunosuppression should be considered in refractory/ recurrent cases n Simultaneous Rx of sex partners has no effect on recurrence (but 3 -10% of sex partners may have balanitis) n Vaginal culture useful to confirm diagnosis and identify unusual species Treatment n Initial regimen of 7 -14 days topical therapy n Fluconazole 150 mg (repeat 72 hrs) n Maintenance regimens- clotrimazole, ketoconazole, fluconazole, itraconazole n For Non-albicans VVC: n Longer duration of therapy n Non-azole regimen may even be needed n 600 mg boric acid in gelatin capsule vaginally once a day for 14 days n

VULVOVAGINAL CANDIDIASIS Treatment in Pregnancy n Only topical intravaginal regimens recommended (usually for 7 days)

VULVOVAGINAL CANDIDIASIS Management of Sex Partners n Treatment not recommended n Treatment of male partners does not reduce frequency of recurrences in the female n But, male partners with balanitis may benefit from treatment

BACTERIAL VAGINOSIS n Not a classical STD n Overgrowth of vaginal normal flora with anaerobic bacteria and decrease or loss of protective lactobacilli (Disturbed vaginal ecosystem) Gardrenella vaginalis (GV) & other microrganisms in high titers But, GV found in 50% of vaginal cultures in asymptomatic women too. BV linked to: premature rupture of membranes, premature delivery and low birth-weight delivery, acquisition of HIV, development of PID, and post-operative infections after gynecological procedures Male sex partners may be colonized but asymptomatic n n
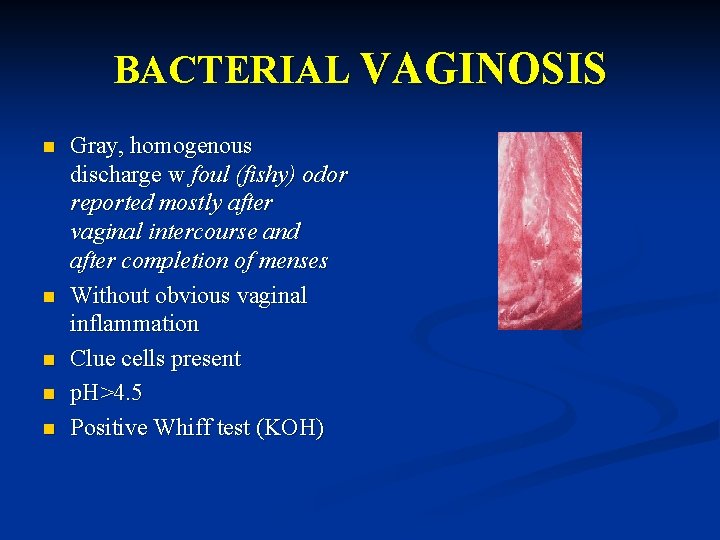
BACTERIAL VAGINOSIS n n n Gray, homogenous discharge w foul (fishy) odor reported mostly after vaginal intercourse and after completion of menses Without obvious vaginal inflammation Clue cells present p. H>4. 5 Positive Whiff test (KOH)
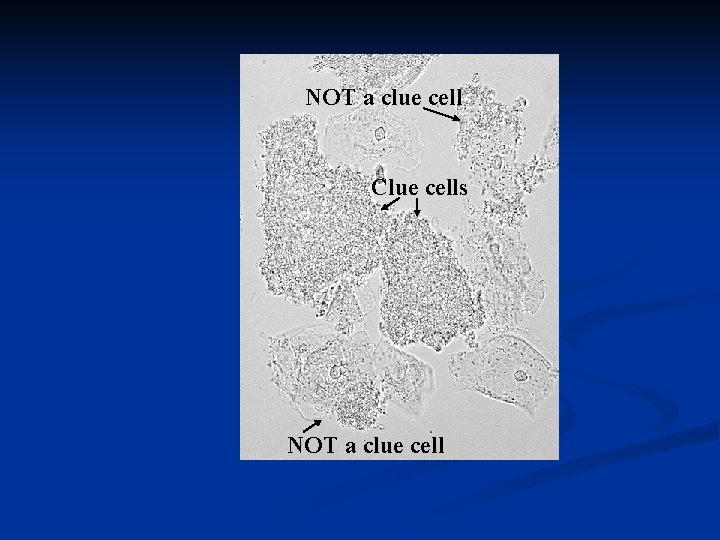
NOT a clue cell Clue cells NOT a clue cell

BV Diagnosis: Amsel Criteria Ø Vaginal p. H >4. 5 Ø Presence of >20% per HPF of "clue cells" on wet mount examination Amsel Criteria: Must have at least three of the following Ø findings: Ø Positive amine or "whiff" test Homogeneous, non-viscous, milky-white discharge adherent to the vaginal walls

BACTERIAL VAGINOSIS Other Diagnostic Tools n Culture not recommended; Do not Rx a positive GV vaginal culture in asymptomatic women n n Newer diagnostic modalities include: n Fem. Exam™ n PIP Activity Test. Card™ DNA probe

BACTERIAL VAGINOSIS TREATMENT n. Metronidazole 500 mg twice daily x 7 days n. Metronidazole gel 0. 75%, 5 g intravaginally once daily x 5 days n. Clindamycin cream 5%, 5 g intravaginally qhs x 7 days Alternative regimens n. Metronidazole 2 gm in a single dose n. Clindamycin 300 mg twice daily x 7 days n. Clindamycin ovules 100 g intravaginally qhs x 3 days

BACTERIAL VAGINOSIS Treatment in Pregnancy n Symptomatic pregnant women should be treated due to association with adverse pregnancy outcomes n Do not use of topical agents in pregnancy n Some experts recommend screening and treatment of asymptomatic women at high risk for preterm delivery (previous preterm birth) at the first prenatal visit; optimal regimen not established

BACTERIAL VAGINOSIS Treatment in Pregnancy Metronidazole 250 mg three times daily for 7 days or Clindamycin 300 mg twice daily for 7 days

BACTERIAL VAGINOSIS Management of Sex Partners n. Not recommended n Woman’s response to therapy and the likelihood of relapse or recurrence not affected by treatment of sex partner
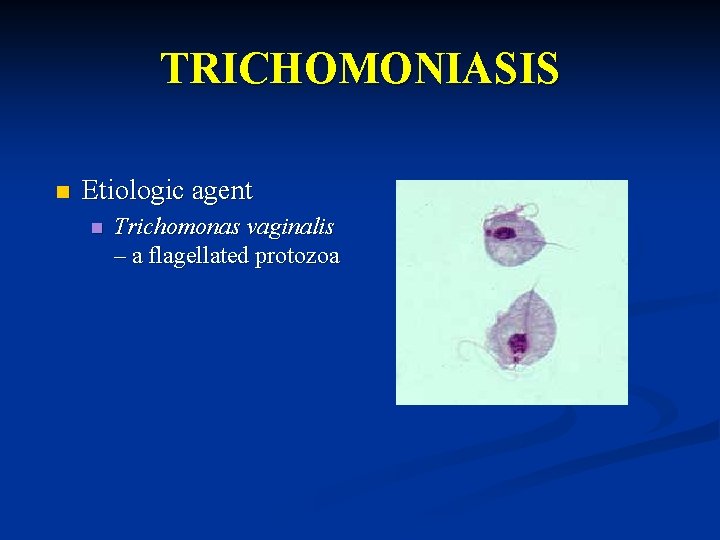
TRICHOMONIASIS n Etiologic agent n Trichomonas vaginalis – a flagellated protozoa

Trichomoniasis and other vaginal infections — Initial visits to physicians’ offices: United States, 1966– 2003 SOURCE: National Disease and Therapeutic Index (IMS Health)

TRICHOMONIASIS n n n Estimated 7. 4 million cases annually in the U. S. Almost always sexually transmitted Causes urethritis in men (usu. asymptomatic) Transmission between female sex partners has been documented Fomite transmission rare Possible association with n n Pre-term rupture of membranes and pre-term delivery Increased risk of HIV acquisition
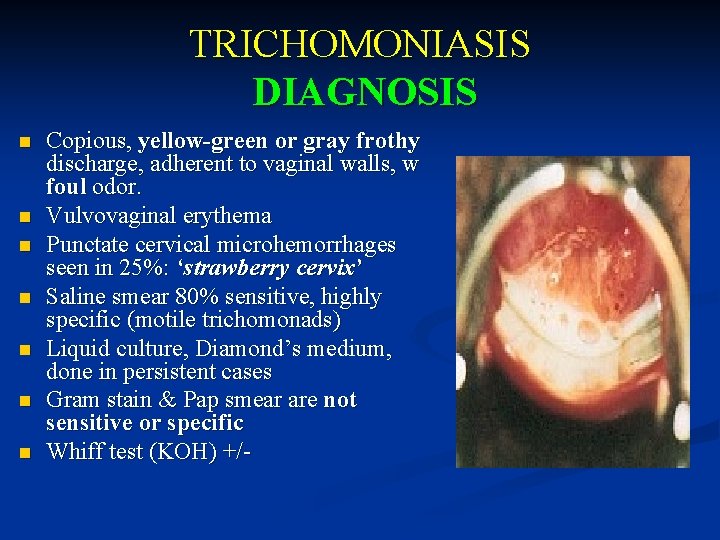
TRICHOMONIASIS DIAGNOSIS n n n n Copious, yellow-green or gray frothy discharge, adherent to vaginal walls, w foul odor. Vulvovaginal erythema Punctate cervical microhemorrhages seen in 25%: ‘strawberry cervix’ Saline smear 80% sensitive, highly specific (motile trichomonads) Liquid culture, Diamond’s medium, done in persistent cases Gram stain & Pap smear are not sensitive or specific Whiff test (KOH) +/-

TRICHOMONIASIS TREATMENT Recommended regimen Metronidazole 2 gm orally in a single dose Alternative regimen Metronidazole 500 mg twice a day for 7 days Pregnancy Metronidazole 2 gm orally in a single dose

TRICHOMONIASIS TREATMENT FAILURE n Re-treat with metronidazole 500 mg twice daily for 7 days n If above fails, Rx with metronidazole 2 gm single dose x 3 -5 days n In repeated failure: n Confirm diagnosis with culture n consider metronidazole susceptibility testing through the CDC n Trial of tinidazole

TRICHOMONIASIS Other management issues No alcohol for the duration of treatment and for at least 24 h after the last dose. n Trich is an STD, so: n GC and Chlamydia testing should be done, & n Syphilis, HIV, and hepatitis B serologic testing should be considered n

TRICHOMONIASIS Management of Sex Partners n Sex partners should be treated, even if asymptomatic n Avoid intercourse until therapy is completed and patient and partner are asymptomatic .
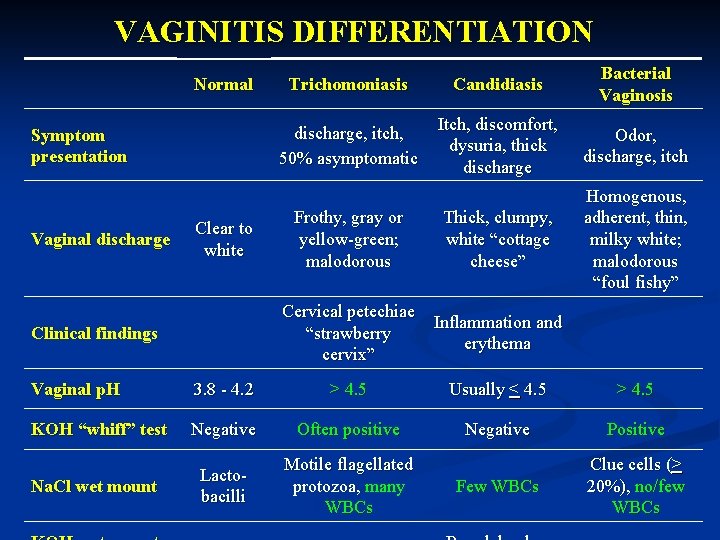
VAGINITIS DIFFERENTIATION Normal Symptom presentation Vaginal discharge Clear to white Trichomoniasis Candidiasis Bacterial Vaginosis discharge, itch, 50% asymptomatic Itch, discomfort, dysuria, thick discharge Odor, discharge, itch Thick, clumpy, white “cottage cheese” Homogenous, adherent, thin, milky white; malodorous “foul fishy” Frothy, gray or yellow-green; malodorous Cervical petechiae Inflammation and “strawberry erythema cervix” Clinical findings Vaginal p. H 3. 8 - 4. 2 > 4. 5 Usually < 4. 5 > 4. 5 KOH “whiff” test Negative Often positive Negative Positive Na. Cl wet mount Lactobacilli Motile flagellated protozoa, many WBCs Few WBCs Clue cells (> 20%), no/few WBCs

NON-INFECTIOUS VAGINITIS n n n Vaginal foreign bodies, especially in prepubescent girls, may present with a heavy white discharge but would be unaccompanied by vulvar erythema or the microscopic appearance of hyphae. Atrophic vaginitis is commonly found in postmenopausal women and is distinguished from candidal vaginitis by mucosal dryness, atrophy, dyspareunia, minimal discharge, and itching. Contact dermatitis, local irritation secondary to tight -fitting underwear, and contact dermatitis from toiletry items, latex condoms, diaphragms, spermicides
MUCOPURULENT CERVICITIS n Largely caused by Chlamydia trachomatis and Neiserria Gonorrheae

Chlamydia trachomatis

Chlamydia — Rates: United States, 1984– 2003

Chlamydia — Rates by sex: United States, 1984– 2003 CDC

Chlamydia trachomatis n n Estimated 3 million cases in the U. S. annually Women: bartholinitis, cervicitis, urethritis, PID, perihepatitis, conjunctivitis n n n Men: urethritis, epididymitis M&W: LGV Infants: conjunctivitis, pneumonia Complications: PID, perihepatitis, Reiter’s syndrome, infertility, ectopic pregnancy, chronic pelvic pain, increased risk for HIV Incubation period is 7 -21 days.

Chlamydia trachomatis Risk factors n Adolescence n n n n Cervical epithelial cells are developmentally immature (ectopy) making them more susceptible to infection. Risky behaviors also contribute to susceptibility. New or multiple sex partners History of past STD infection Presence of another STD Oral contraceptive use (contributes to cervical ectopy, & OCP users less likely to use barrier protection) Lack of barrier contraception

Chlamydia trachomatis Cervicitis n Majority of cervical infections are asymtpomatic-70% to 80%. n When symptomatic, S+S may be non-specific: n spotting, or mucopurulent cervicitis, with mucopurulent endocervical discharge, edema, erythema, and friability w easily induced bleeding within the endocervix or any zones of ectopy. Urethritis n n n 50% of infected women yield chlamydia from both urethra and cervical sites Usually asymptomatic May cause the “dysuria-pyuria” syndrome mimicking acute cystitis. On urinalysis, pyuria present but few bacteria.

Chlamydia trachomatis DIAGNOSIS Culture: high specificity BUT n labor-intensive, expensive, n variable sensitivity (50%-80%), n not suitable for widespread screening Non-culture methods: n Serology: not very useful n EIA, DFA, DNA probe : less sensitive(50 -75%), nonspecific n Nucleic acid amplification tests (NAAT): PCR, LCR: n more sensitive than culture (>80%-90%) n highly specific (>99%) n can use first void urine n can use self-obtained vaginal swab

Chlamydia trachomatis Treatment Azithromycin 1 gm single dose or Doxycycline 100 mg bid x 7 d

Chlamydia trachomatis Alternative regimens Erythromycin base 500 mg qid for 7 days or Erythromycin ethylsuccinate 800 mg qid for 7 days or Ofloxacin 300 mg twice daily for 7 days or Levofloxacin 500 mg for 7 days

Chlamydia trachomatis Treatment in Pregnancy Recommended regimens Erythromycin base 500 mg qid for 7 days or Amoxicillin 500 mg three times daily for 7 days Alternative regimens Erythromycin base 250 mg qid for 14 days or Erythromycin ethylsuccinate 800 mg qid for 14 days or Erythromycin ethylsuccinate 400 mg qid for 14 days or Azithromycin 1 gm in a single dose

Chlamydia trachomatis Screening n n n Annual screening of sexually active women < 25 yrs Annual screening of sexually active women > 25 yrs with risk factors Sexual risk assessment may indicate need for more frequent screening for some women Screen pregnant women in the first trimester Re-screen women 3 -4 months after treatment due to high prevalence of repeat infection

GONORRHEA

Gonorrhea — Rates: United States, 1970– 2003 and the Healthy People 2010 target Note: The Healthy People 2010 target for gonorrhea is 19. 0 cases per 100, 000 population.

GONORRHEA n n n Caused by Neisseria gonorrhoeae, a gram-neg intracellular diplococcus. Estimated 700, 00 -800, 000 persons infected annually in the US. Manifestations in women may include: n cervicitis, PID, urethritis, pharyngitis, proctitis, disseminated (bacteremia, arthritis, tenosynovitis) n Accessory gland infection (Bartholin’s glands, Skene’s glands) n Fitz-Hugh-Curtis Syndrome (Perihepatitis)

Gonorrhea Cervicitis Clinical Manifestations Symptoms are non-specific : abnormal vaginal discharge, intermenstrual bleeding, dysuria, lower abdominal pain, or dyspareunia n Clinical findings: mucopurulent or purulent cervical discharge, easily induced cervical bleeding n 50% of women with clinical cervicitis are asymptomatic n Incubation period unclear, but symptoms may occur within 10 days of infection n
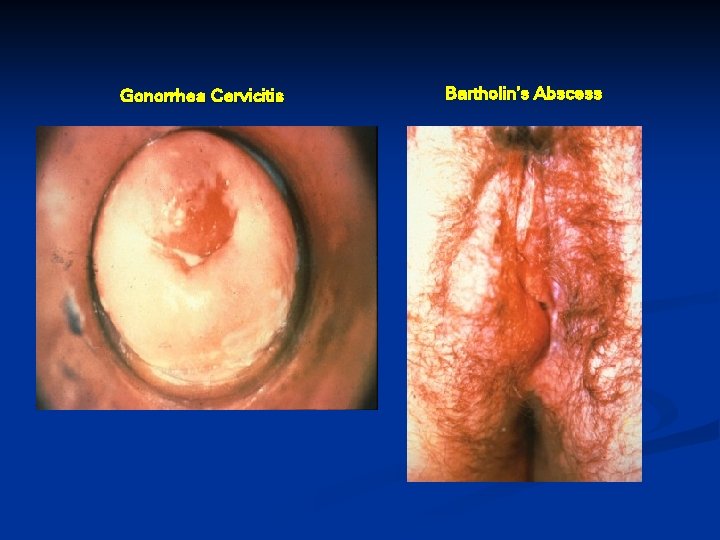
Gonorrhea Cervicitis Bartholin’s Abscess

GONORRHEA LAB DIAGNOSIS n Culture (selective media-Thayer Martin, needs CO 2) n Non-culture tests: DNA probe, nucleic acid amplification n Gram-stain, less sensitive in cervicitis (most sensitive for symptomatic urethritis in men)
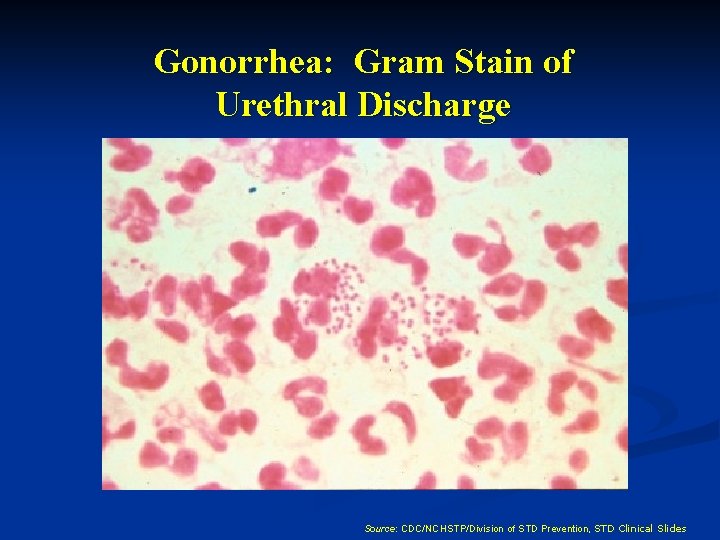
Gonorrhea: Gram Stain of Urethral Discharge Source: CDC/NCHSTP/Division of STD Prevention, STD Clinical Slides

Neisseria gonorrhoeae (Cervix, Urethra, Rectum) Cefixime 400 mg or Ceftriaxone 125 IM or 1 Ciprofloxacin 500 mg or 1 Ofloxacin 400 mg or 1 Levofloxacin 250 mg PLUS Chlamydial therapy if infection not ruled out 1 Contraindicated in pregnancy and children. Not recommended for infections acquired in California, Asia, or the Pacific, including Hawaii.

Neisseria gonorrhoeae (Cervix, Urethra, Rectum) Alternative regimens Spectinomycin 2 grams IM in a single dose or Single dose cephalosporin (cefotaxime 500 mg) or Single dose quinolone (gatifloxacin 400 mg, lomefloxacin 400 mg, norfloxacin 800 mg) PLUS Chlamydial therapy if infection not ruled out

Neisseria gonorrhoeae Treatment in Pregnancy n Cephalosporin regimen n Women who can’t tolerate cephalosporin regimen may receive 2 g spectinomycin IM n No quinolone or tetracycline regimen PLUS Erythromycin or amoxicillin for presumptive or diagnosed chlamydial infection

Gonococcal Isolate Surveillance Project (GISP) — Percent of Neisseria gonorrhoeae isolates with resistance or intermediate resistance to ciprofloxacin, 1990– 2003 Note: Resistant isolates have ciprofloxacin MICs ≥ µg/ml. Isolates with intermediate resistance have ciprofloxacin MICs of 0. 125 - 0. 5 µg/ml. Susceptibility to ciprofloxacin was first measured in GISP in 1990.

Neisseria gonorrhoeae Antimicrobial Resistance n Surveillance is crucial for guiding therapy recommendations n No significant resistance to ceftriaxone n Fluoroquinolone resistance in SE Asia, Pacific, Hawaii, California, Washington. n FQ resistance 15% in MSM.

GONORRHEA TREATMENT ISSUES n n n Fluoroquinolones are no longer recommended for therapy for gonorrhea acquired in Asia, the Pacific Islands (including Hawaii), and California. CDC no longer recommends fluoroquinolones as a first-line therapy for gonorrhea in MSM If symptoms persist, perform culture for N. gonorrhoeae. Any gonococci isolated should be tested for antimicrobial susceptibility Co-infection with Chlamydiae in up to 50% of pts, hence anti-Chlmydia Rx added. Note : A test of cure is not recommended, if a recommended regimen is administered.

GONORRHEA Partner Management n n n Evaluate and treat all sex partners for N. gonorrhoeae and C. trachomatis infections if contact was within 60 days of symptoms or diagnosis. If a patient’s last sexual intercourse was >60 days before onset of symptoms or diagnosis, the patient’s most recent sex partner should be treated. Avoid sexual intercourse until therapy is completed and both partners no longer have symptoms.

PELVIC INFLAMMATORY DISEASE (PID)

PELVIC INFLAMMATORY DISEASE n n n Estimated about 1 million annual cases in the US Endometritis, salpingitis, tuboovarian abscess, & pelvic peritonitis. Ascending infection from or via cervix Most cases of PID are polymicrobial: Chlamydia, GC, vaginal organisms, anaerobes, enteric GNR, GPC). May be unrelated to STD. Most common pathogens: n N. gonorrhoeae: recovered from cervix in 30%-80% of women with PID n C. trachomatis: recovered from cervix in 20%-40% of women with PID n N. gonorrhoeae and C. trachomatis are present in combination in approximately 25%-75% of patients

PELVIC INFLAMMATORY DISEASE RISK FACTORS n n n n n Adolescence (in sexually active teens 3 x more than 25 -29 yr olds) History of PID GC or chlamydia, or a history of GC or chlamydia Male partners with GC or chlamydia Multiple partners Current douching Insertion of IUD (especially within 4 mos after insertion) Bacterial vaginosis Demographics (lower socioeconomic status) Oral contraceptive use, in some cases (by avoidance of barrier precautions? )
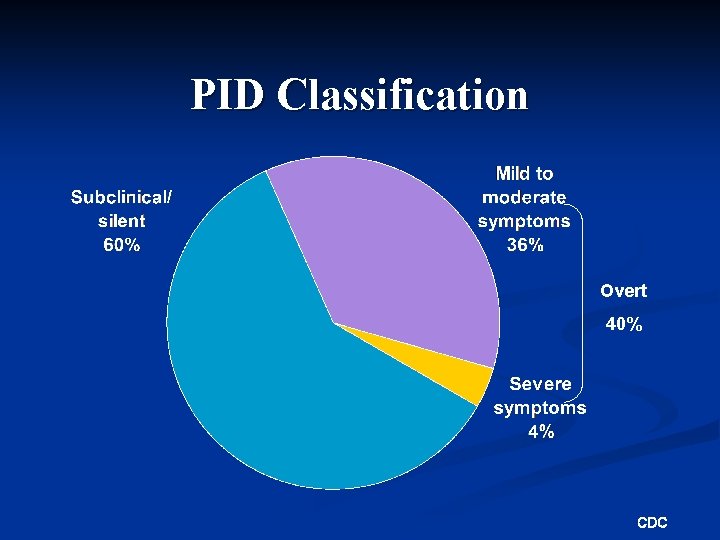
PID Classification Overt 40% CDC

PELVIC INFLAMMATORY DISEASE Minimum Diagnostic Criteria Uterine/adnexal tenderness or cervical motion tenderness Additional Diagnostic Criteria Oral temperature >38. 3 C Elevated ESR Cervical Chlamydia or GC Elevated CRP WBCs/saline microscopy Cervical Discharge

Pelvic Inflammatory Disease More Specific Criteria n Endometrial biopsy: histopathologic evidence of endometritis n Imaging Studies: Transvaginal sonography or MRI (showing thickened fluid-filled tubes) n Laparoscopy: abnormalities consistent with PID

PELVIC INFLAMMATORY DISEASE MANAGEMENT n n Antibiotics Bed rest Reevaluation within 72 hrs of treatment All male sex partners should be evaluated for STD and empirically treated with regimen effective for GC/Chlmydia

PELVIC INFLAMMATORY DISEASE MANAGEMENT Hospitalize, if: n Surgical emergencies not excluded (e. g. , appendicitis, ectopic pregnancy. . ) n Pregnant patient n Pelvic abscess is suspected n Adolescent n Severe illness n If unable to tolerate outpt regimen n If f/up within 72 hrs after starting abx cannot be arranged n Non-response to oral therapy n HIV infection with low CD 4 count

Pelvic Inflammatory Disease Parenteral Regimen A Cefotetan 2 g IV q 12 hours or Cefoxitin 2 g IV q 6 hours PLUS Doxycycline 100 mg orally/IV q 12 hrs

PELVIC INFLAMMATORY DISEASE Parenteral Regimen B Clindamycin 900 mg IV q 8 hours PLUS Gentamicin loading dose IV/IM (2 mg/kg) followed by maintenance dose (1. 5 mg/kg) q 8 hours. Single daily dosing may be substituted.

PELVIC INFLAMMATORY DISEASE Alternative Parenteral Regimens Ofloxacin 400 mg IV q 12 hours or Levofloxacin 500 mg IV once daily WITH OR WITHOUT Metronidazole 500 mg IV q 8 hours or Ampicillin/Sulbactam 3 g IV q 6 hrs PLUS Doxycycline 100 mg orally/IV q 12 hrs

PELVIC INFLAMMATORY DISEASE Oral Regimen A Ofloxacin 400 mg twice daily for 14 days or Levofloxacin 500 mg once daily for 14 days WITH OR WITHOUT Metronidazole 500 mg twice daily for 14 days

PELVIC INFLAMMATORY DISEASE Oral Regimen B Ceftriaxone 250 mg IM in a single dose or Cefoxitin 2 g IM in a single dose and Probenecid 1 g administered concurrently PLUS Doxycycline 100 mg twice daily for 14 days WITH or WITHOUT Metronidazole 500 mg twice daily for 14 days

SUSPECTED TUBOOVARIAN ABSCESS Cultures n Broad spectrum antibiotics n n n 85% of abscesses w a diameter of 4 -6 cm (& only 40% of those >10 cm) respond to abx alone Surgery for failure to respond to abx.

PELVIC INFLAMMATORY DISEASE SEQUELAE n Ectopic pregnancy n n 7 -fold increase in risk after a single episode of PID Infertility: n n n 13% of women after one episode of PID 25 -35% after 2 episodes, 50 -75% after 3 or more episodes 2/3 unable to conceive after Rx for TOA Dyspareunia n Pelvic adhesions n Chronic pelvic pain n

PELVIC INFLAMMATORY DISEASE Management of Sex Partners n Male sex partners of women with PID should be examined and treated for sexual contact 60 days preceding pt’s onset of symptoms n Sex partners should be treated empirically with regimens effective against CT and GC


Genital herpes — Initial visits to physicians’ offices: United States, 1966– 2003 SOURCE: National Disease and Therapeutic Index (IMS Health)

Genital HSV Infection n n n More than one in five Americans (45 million people)-are estimated infected with genital herpes more common in women than men, infecting approximately one out of four women, versus one out of five men. In a national house-hold survey, less than 10 percent of people who tested positive with herpes knew they were infected (Fleming, 1997). ---Silent epidemic--Genital herpes is a recurrent, lifelong viral infection. Asymptomatic shedding occurs (Most sexual transmission occurs while source case is asymptomatic). Incubation period is 2 -12 days (average is 4 days). Can be transmitted between sex partners, from mothers to newborns, and can increase a person's risk of becoming infected with HIV

Estimated Annual Incidence of Selected STDs in the U. S. , 2000 Trichomoniasis 7. 4 million n Human Papillomavirus (HPV) 6. 2 million n Chlamydia 2. 8 million n Herpes Simplex Virus (HSV) Type 2 : 1. 6 million n Gonorrhea 718, 000 n Syphilis 37, 000 n


HSV Serologic Tests Type-Specific n HSV-specific glycoprotein G 2 for HSV 2 infection and glycoprotein G 1 for HSV 1 n Available g. G type-specific assays- POCkit HSV-2, Herpe. Select HSV 1/2 Ig. G ELISA and Herpe. Select 1/2 immunoblot Ig. G n Sensitivity 80 -98%, Specificity > 96% n Confirmatory testing may be indicated in some settings

Genital Herpes First Clinical Episode Acyclovir 400 mg tid or Famciclovir 250 mg tid or Valacyclovir 1000 mg bid Duration of Therapy 7 -10 days

Genital Herpes Episodic Therapy Acyclovir 400 mg three times daily x 5 days or Acyclovir 800 mg twice daily x 5 days or Famciclovir 125 mg twice daily x 5 days or Valacyclovir 500 mg twice daily x 3 -5 days or Valacyclovir 1 gm orally daily x 5 days

Genital Herpes Daily Suppression Acyclovir 400 mg bid or Famciclovir 250 mg bid or Valacyclovir 500 -1000 mg daily

Genital Herpes in HIV Infection n May have prolonged or severe episodes with extensive genital or perianal disease n Episodic or suppressive antiviral therapy often beneficial n For severe cases, acyclovir 5 -10 mg/kg IV q 8 hours may be necessary

Genital Herpes HIV Infection/Episodic Therapy Acyclovir 400 mg three times daily or Famciclovir 500 mg twice daily or Valacyclovir 1 gm twice daily Duration of Therapy 5 -10 days

Genital Herpes HIV Infection/Daily Suppression Acyclovir 400 -800 mg twice to three times daily or Famciclovir 500 mg twice daily or Valacyclovir 500 mg twice daily

Genital Herpes Antiviral Resistance n Persistent or recurrent lesions on antivirals n Obtain viral isolate for viral susceptability n 5% immunocomprised patients n Acyclovir resistant isolates-resistant to valacyclovir, most resistant to famciclovir n Alternatives: Foscarnet 40 mg/kg IV q 8 or topical cidofovir gel 1% (daily x 5 days)

Herpes in Pregnancy Risk for transmission to neonate from infected mother is : n high (30%-50%) among women who acquire genital herpes near the time of delivery, but low (<1%) in women with histories of recurrent herpes at term or who acquire genital HSV during the first half of pregnancy. n Prevention of neonatal herpes depends on avoiding acquisition of HSV during late pregnancy and avoiding exposure of the infant to herpetic lesions during delivery. n Women without symptoms or signs of genital herpes or its prodrome can deliver vaginally

Genital Herpes Treatment in Pregnancy n Acyclovir may be used with first episode or severe recurrent disease n Available data do not indicate an increased risk of major birth defects (first trimester) n The safety of acyclovir, valacyclovir, and famciclovir therapy in pregnant women has not been established.

Genital Herpes Counseling n Natural history of infection, recurrences, asymptomatic shedding, transmission risk n Individualize use of episodic or suppressive therapy n Abstain from sexual activity when lesions or prodromal symptoms present n Risk of neonatal infection



HUMAN PAPILLOMAVIRUS n n n . 6. 2 million Americans get a new genital HPV infection each year May cause cancer of cervix, vulva, vagina, or anus n the most common sources of genital warts--HPV types 6 and 11 --are rarely associated with malignancy n the high-risk HPV types 16 and 18 have been found in more than 90% of cervical cancers They appear an average of 3 months after exposure, the latency period can be much longer. Infection can be clinically apparent, subclinical, or latent Frequency of spontaneous regression is unclear. A few studies indicate a regression rate of 10%-30% within 3 months. Persistence of infection occurs, but frequency and duration is unknown. Recurrences after treatment are common (20%-50% recurrence rate at 36 months). Symptoms Genital warts usually cause no symptoms other than the warts themselves. Vulvar warts can cause dyspareunia, pruritis, and burning discomfort. Urethral meatal warts occasionally cause hematuria or impairment of urinary stream. Vaginal warts occasionally cause discharge, bleeding, or obstruction of

HUMAN PAPILLOMAVIRUS Risk for Malignancy n n Externa genital warts n HPV types 6, 11. n Minimal risk for malignancy Flat warts n HPV 16, 18, 31, 45… n Associated with cancer of cervix, vagina, vulva, anus, penis n Most women with persistent HPV infection do not develop cervical cancer precursors or cervical cancer. n Over 99% of cervical cancers have HPV DNA detected within the tumor. n Persistent infection with a high-risk HPV type is necessary but not sufficient for the development of cervical cancer.

HUMAN PAPILLOMAVIRUS DIAGNOSIS Inspection usually diagnostic of external warts: , biopsy if in doubt n Pap smear, biopsy for flat warts of cervix n HPV-DNA studies, PCR, hybrid capture n HPV cannot be cultured, and serologic tests are not available to test for HPV antibodies n Subclinical infections may be detected by applying 3% to 5% acetic acid solution for 5 to 10 minutes. The lesions then become visible, and can be further visualized via colposcopy. n

HUMAN PAPILLOMAVIRUS Treatment n Primary goal for treatment of visible warts is the removal of symptomatic warts n Therapy may reduce but probably does not eradicate infectivity n Difficult to determine if treatment reduces transmission n. No laboratory marker of infectivity n. Variable results utilizing viral DNA

HUMAN PAPILLOMAVIRUS n Choice of therapy guided by preference of patient, experience of provider, resources n No evidence that any regimen is superior n Locally developed/monitored treatment algorithms associated with improved clinical outcomes n Acceptable alternative may be to observe; possible regression/uncertain transmission

PAPILLOMAVIRUS Patient-applied n n Podofilox 0. 5% solution or gel Imiquimod 5% cream Provider-administered n n Cryotherapy Podophyllin resin 10 -25% Trichloroacetic or Bichloroacetic acid 80 -90% Surgical removal

HUMAN PAPILLOMAVIRUS Treatment in Pregnancy n n Imiquimod, podophyllin, podofilox should not be used in pregnancy Many specialists advocate wart removal due to possible proliferation and friability HPV types 6 and 11 can cause respiratory papillomatosis in infants and children Preventative value of cesarean section is unknown; may be indicated for pelvic outlet obstruction



CHORIOAMNIONITIS RISK FACTORS n n n n Nulliparity Length of labor Preterm labor PROM Meconium stained amniotic fluid Internal fetal or uterine monitoring Presence of GU pathogens (GBS, GC, BV) No of vag exams in women w ruptured membrane Underlying Host Factors n No. of lactobacilli, n Ig. A, n Chronic diseases n Immunosuppression n Nutritional disorders n Drug abuse.

CHORIOAMNIONITIS DIAGNOSIS n n Maternal fever >38 C(>100. 4) AND at least 2 of the following: n Maternal leukocytosis (>15, 000 cells/cubic mm) n Maternal tachycardia (>100 beats/min) n Fetal tachycardia (>160 beats/min) n Uterine tenderness n Foul odor of the amniotic fluid AMNIOTIC FLUID ANALYSIS: n Gram stain: bacteria & leukocytes (> 6 leukocytes/hpf) n Glucose (<15 mg/dl abnormal) n WBC (Abnormal >30 cells/cc) n Leukocyte esterase (strips) + Abnormal glu + wbc + Leuk/esterase= sensitivity 90%, specificty 80% for pos culture MICROBIOLOGY: n Organisms from vaginal flora, anaerobes, mycoplasma, GBS, E. coli. n Usually polymicrobial

CHORIOAMNIONITIS MANAGEMENT 1. Antibiotics n n 2. Amp/gent/clinda. Other broad-spectrum regimen Delivery (Note: C-section should be performed only for accepted obstetric indications)


POSTPARTUM ENDOMETRITIS DIAGNOSIS n Fever, usually on 1 st or 2 nd postpartum day. Lower abdominal pain Uterine tenderness Leukocytosis n Bimanual exam should be done n n n Microbiologic diagnosis: n Transvaginally obtained cultures are controversial (contaminants) n Blood cultures should be done (10 -20% have bacteremia) n Chlamydia testing (culture, antigen, PCR) should be done for high risk pts & with late-onset PPE.

POSTPARTUM ENDOMETRITIS PREDISPOSING FACTORS n n C-section, especially after labor or rupture of membranes is the main predictor n Incidence after vaginal delivery 0. 9 -3. 9 % n Incidence after C-section 10 -50% Other predictors: n Duration of labor n Rupture of membranes n Presence of BV n Number of vag. Exams during labor n Use of internal fetal monitoring.

POSTPARTUM ENDOMETRITIS (PPE) MICROBIOLOGY n n n Polymicrobial (GBS, enterococci, G. vaginalis, E. coli, Prevotella bivia, Bacteroides spp, peptostreptococci, Ureoplasma urealyticum, Mycoplasma hominis) Chlamydia trachomatis may cause a late form of PPE (>2 days to 6 wks postpartum, after vag delivery) Group A Strep PPE is rare n n of exogenous source, usually caregiver. Major epidemiologic significance: HCW screening (all at the delivery & those who did vag exam before delivery should be screened w cultures of nares, throat, vagina, rectum, skin. If culture + should refrain from patient care for the 1 st 24 h of abx therapy)

POSTPARTUM ENDOMETRITIS MANAGEMENT n Antibiotics (broad-spectrum) n until pt is afebrile, pain-free, & with normal wbc count. FAILURE TO RESPOND may indicate: n multi-drug resistant bacteria, n inadequate regimen, n abscess, n puerperal ovarian vein thrombosis, n non-infectious fever (e. g. , drug-fever, breast engorgement) PROPHYLAXIS: n Abx prophylaxis for any c-section after labor or rupture of membranes of any duration

PUERPERAL OVARIAN VEIN THROMBOSIS n n n Acute postpartum thrombosis of ovarian veins Rare, incidence 1/2000 deliveries or 1 -2/100 pts w postpartum infection Can occur after c-section or vaginal delivery. Usually associated with post-c-section endometritis. Previously diagnosed w “PPE failing to respond to abx” Onset mostly 2 -4 days after delivery. Acute onset, pt appears ill, febrile/chills, lower abd pain (usually rt sided), tachycardia disproportionately elevated c/w temp. EXAM: tenderness, tender sausage-shaped mass may be palpable (1/22/3). If PE has occurred may have respiratory complaints too. Usually a diagnosis of exclusion. Sono, CT, or MRI may confirm diagnosis Rx: Abx, anticoagulation ( usually x 7 -10 d, in absence of PE)
- Slides: 113