GuillainBarre Syndrome William Woodfin MD K F 40

Guillain-Barre Syndrome William Woodfin MD

K. F. 40 y. o. r/h woman 3/17 Nausea, diarrhea & severe myalgias Son dxed c rotavirus 1 wk. Previously 4/21 “Creepy-crawlies” legs>arms 4/25 Weakness legs progressing 4/26 Handwriting looks like “hen scratch”

K. F. 40 y. o. woman 4/28 Admitted to outside hospital. L. P. wnl EMG positive waves in some leg muscles NCVs absent H-reflexes F responses & motor latencies wnl

K. F. 40 y. o. woman 4/29 Transferred to PHD Hx. : diabetic x 10 yrs. hypothyroid- treatedx yrs. no sphincter disrubance aching pain low back & buttocks mild postural light headedness no SOB or palpatations

Exam BP 150/90 P 80 Wt. 250 lbs. Mild weakness neck flexors 4/5 biceps, grip & interossei- symmetric 2/5 iliopsoas & quadriceps 3/5 hamstrings & adductors 4/5 abductors 4/5 ankles & toes- extensors & flexors

Exam Sensory- intact DTRs- biceps, BR, knees are trace c reinforcement. Triceps & ankles unobtainable Plantars- flexor F to N- intact Gait- not testable

Lab H/H 10. 3/33. 5 c microcytic indices A 1 c Hgb 10. 1 TSH 0. 97 LDL 182 Serum immunofixation- wnl. No Ig. A def. FVCs- consistently 4+ liters

MRI LS spine s & c contrast- no nerve root enhancement

Course in hospital Treated c IVIG 0. 4 gms/kgm daily x 5 Strength fluctuated only mildly Blood sugars ok in AM, high in afternoons Repeated NCVs show mild dispersion of F waves Transferred back to referring hospital 5/6

Telephone FU Ambulating fairly well c walker. Strength clearly improving. Still bothered by “creepy-crawlies”

What is the GBS? • Due to the breadth of clinical presentation it is of limited help to try to define rigid diagnostic criteria. • Thomas Munsat 1965: “…The GBS is easy to diagnose but difficult to define

The typical illness evolves over weeks usually following an infectious disease and involves: • 1. Paresthesiaes usually hearld the disease • 2. Fairly symmetric weakness in the legs, later the arms and, often, respiratory and facial muscles • 3. Dimunition and loss of the DTRs • 4. Albuminocytologic dissociation • 5. Recovery over weeks to months

History Waldrop 1834 Olliver 1837 Landry 1859 Graves 1884 Ross & Bury 1893 Guillain, Barre & Strohl 1916 -1920 Brussel’s Conf. 1937 Haymaker & Kernohan 1949 Waksman & Adams 1955 Miller Fisher 1956 Asbury, Aranson & Adams 1969


Note sur la paralysie ascendante aigue 1859 • March 16 - a febrile illness • May 11 - mild sensory symptoms in the fingers and toes • June 13 - knees buckle • June 16 - unable to walk • Subsequent respiratory failure and death. • Autopsy unrevealing. Peripheral nerves probably not examined

Late 19 th century • Westphal 1876 - “Landry’s Ascending Paralysis” • Graves 1884 - localized neurologic disease to the peripheral nerves, “the nervous cords” • Ross & Bury 1893 - 90 cases. A disease of the peripheral nerves • Numerous reports emphasizing various aspects of the disease with most authors crediting Landry

Georges Guillain

Revue Neurologique 1916

Guillain, Barre & Strohl 1916 Revue Neurologique • Two soldiers in Amiens developing paralysis and loss of DTRs. • A new diagnostic feature: albuminocytologic dissociation in the CSF • No mention of Landry

Foundations • Quincke- CSF observations 25 years earlier • Siccard & Foix- “albuminocytologic dissociation” in Pott’s disease Late 19 th century: examination of the reflexes had become a part of the neurologic exam with appreciated as a sign of neuropathy based on observations in tabes dorsalis areflexia

Haymaker & Kernohan 1949 • Landmark in pathological description c 50 fatal cases & detailed review of clinical findings • Emphasized prominent damage to proximal nerves often at junction of ventral & dorsal roots. Little study of more distal nerves • Unified findings of Landry & Guillain, Barre & Strohl

Waksman & Adams 1955 • Experimental Allergic Neuritis • First animal model of a noninfectious inflammatory neuritis • Rabbit nerve and Freund’s adjuvant injected intradermally • Target of activated T cells uncertain

Asbury, Aranson & Adams 1969 • 19 pts. All with well developed mononuclear infiltrates in spinal roots and nerves within days of clinical onset • Pathological hallmark: perivascular mononuclear inflammatory infiltrates to adjacent to the areas of demyelination

Overview of Adaptive Immunity • Lymphocytes: “command & control, ” identify antigen components, respond specifically, mobilize other elements and direct the attack c memory for each antigenic assault • Antibodies: specialized immunoglobulin molecules directly neutralize and remove antigen

T lymphocytes • CD 8 - recognize epitopes paired c MHC-I • CD 4 - activate and control the immune response • Scavenger cells break down antigen into small peptide fragments (T cell epitopes), MHC-II epitope complexes are expressed on the surface & the scavenger become an APC which docks on a CD 4 c a compatible TCR. CD 4 proliferates releasing cytokines.

Antibodies • Cytokines activate other lymphocytes including B cells that differentiate into plasma cells and serve as immunoglobulin factories. • Abs are Ig molecules that recognize, bind, neutralize and opsonize Ag for phagocytosis. They activate complement(membrane attack complex) & induce target cells to activate the inflammatory response
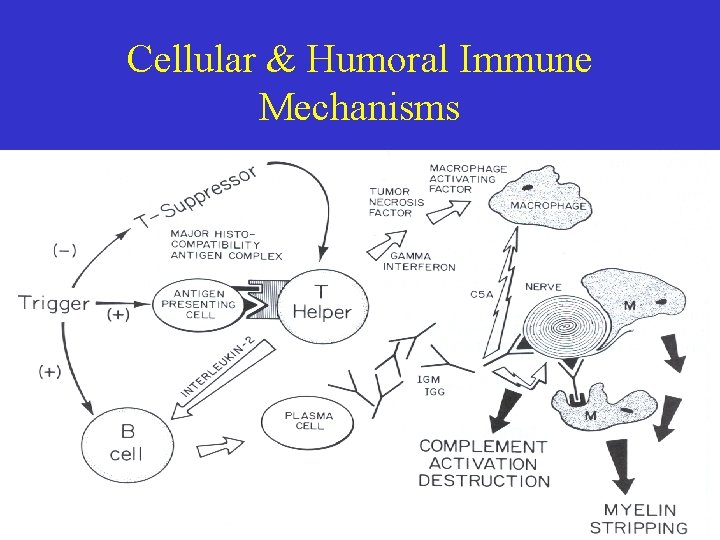
Cellular & Humoral Immune Mechanisms

Self-tolerance • The process of self recognition • T & B cells learn self tolerance during maturation • Autoimmunity occurs when the mechanisms of self protection are defective

Mechanisms of Autoimmunity • Molecular mimicry- microbe cell surface Ag resembles self protein. Damage results from “friendly fire” The inciting Ag is usually unidentified & may not exist as a single stimulus. • Excessive cytokine release due to profound immune stimulus may awaken self tolerant T cells or may cause expression of MHC complexes. • Self Ags bound to drugs may lose tolerated status

Antecedent Events: Infectious • Viral: Influenza, Coxsackie, EBV, Herpes, HIV, Hepatitis, CMV, WNV Bacterial: Campylobacter jejuni, Mycoplasma, E. coli Parasitic: Malaria, Toxoplasmosis

Antecedent Events: Systemic disease • • • Hodgkins CLL Hyperthyroidism Sarcoidosis Collagen Vascular d. Renal d.

Other antecedent events • • • Surgery Immunization Pregnancy Envenomization Bone marrow transplantation Drug ingestion

Features of AIDP • 2/3 s have identifiable preceding event • 50% begin with paresthesias followed by weakness in legs; 10% begin with arm weakness; rarely begins in face • Ophthalmoplegia: partial 15%, total 5% • Autonomic dysfunction in 65%, arrhythmias, hypotension, urinary retention in 10 -15%, pupillary inequality

AIDP • • Progresses for days to 4 weeks 15% with severe disability Mortality 3 -5% CSF: protein may be normal early, elevated in 90% by clinical nadir, cells< 10 in 95%, >50 suggests HIV • EDX: prolonged F & distal motor latencies, conduction block 30 -40% in routine studies

AIDP • Pathology: immune attack directed at schwann cell plasmalemma esp. at nerve roots with Ig. G & complement deposits preceding demyelination
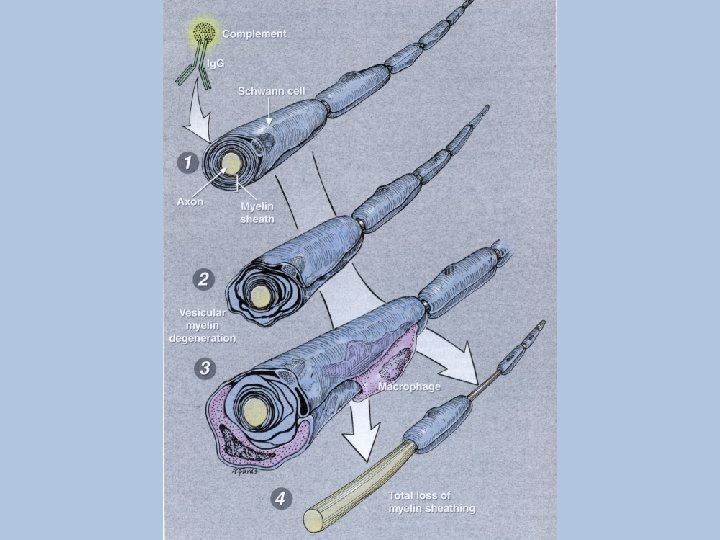

CIDP • Evolves over months • Fluctuates • Respiratory failure, dysautonomia, facial weakness, ophthalmoplegia- all are rare • CSF protein often highly elevated • Marked slowing of motor nerve conduction • Steroid responsive

Features of AMSAN • Commonly preceded by diarrhea esp. c. jejuni • Abrupt onset of weakness c rapid progression to quadriplegia & respiratory insufficiency • Other features as c AIDP • Longer recovery, more residual & mortality 10 -15%

AMSAN • CSF as in AIDP • EDX: no response in some motor nerves, decreased amplitude of the CMAPs, fibrillations on needle study, absent SNAPs • Immune attack directed at axon plasmalemma at nodes of Ranvier. Wallerian degeneration

Features of AMAN • Often preceded by diarrhea affecting younger population in China. Sporadic in USA • Prognosis similair to AIDP • Mortality <5% • EDX: reduced CMAPs c normal F & distal motor latencies and sensory studies. Fibrillations in 2 -3 weeks

AMAN • Pathology: again axonal plasmalemma at nodes of Ranvier sometimes limited to physiologic dysfunction c nodal lengthening. May go on to extension through axonal basal lamina. Most axons recover s Wallerian degeneration
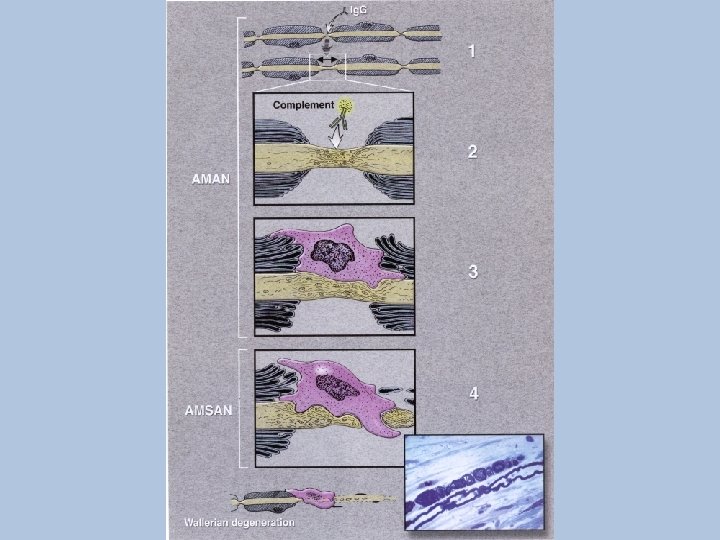

Miller Fisher Syndrome • Ophthalmoplegia, Ataxia, Areflexia • May be heterogonous: 1. Related to other patterns of GBS 2. Related to brainstem encephalitis, Bickerstaff 1952 3. CNS demyelination in association with GBS

Miller Fisher Syndrome • 95% have serum Ig. G Ab to ganglioside GQ 1 b • Studies show preferential location of anti-GQ 1 b to cerebellar molecular layer & Cranial Nerves 3, 4 & 6 • May act at N-M junction depleting acetylcholine from nerve terminals

Acute Panautonomic Neuropathy • Manifests over 1 -2 weeks but may be of subacute onset • Frequent preceding infection • DTRs lost in 1/3, distal sensory changes 1/4 • Albuminocytologic dissociation • EDX: NCVs usually normal • Recovery is gradual and incomplete

Differential Diagnosis • Consider the possibility of an upper motor neuron lesion • Other considerations are rare. Diphtheritic neuritis & poliomyelitis belong more to the history section of this presentation. A new possibility is West Nile Virus.

Differential • N-M: MG, LES, Antibiotics • Toxic: Cigutera (ciguatoxin), Pufferfish (tetrodotoxin), Shellfish (saxitioxin), Botulism, Tick paralysis (Lone Star tick, Gulf Coast tick), Glue sniffing, Buckthorn • Mononeuritis multiplex assoc. c Wegner’s. PAN, SLE, RA, Sjogren’s, Cryoglobulinemia etc.

Differential • Metabolic: Periodic paralyses, Hypokalemia, Hypermagnesemia, Hypophoshatemia c parenteral hyperailimentation, Thyrotoxicosis, ICU myoneuropathy (CIP) • Heavy metal: Lead, Arsenic, Thallium, Barium c hypokalemia

Differential: Miller Fisher Syn. • • Multiple sclerosis Encephalitis Posterior circulation ischemia or infarct Other: Botulism, MG, Tick

Treatment Respiratory failure Autonomic dysfunction DVT & PE Pain Positioning & Skin care Physical therapy Nutrition

Respiratory Failure • Oropharyngeal weakness in ~25% with impaired swallowing of secretions & aspiration • Mechanical respiratory failure- mainly due to diaphragmatic weakness (Phrenic nerves. ) Inspiratory c MIF (Max. Inspir. Force) a good supplement measure to FVC

Respiratory Failure • ~33% require intubation • Avg. time to intubation is 1 week & these pts. have substantially longer recovery time • Need is unlikely if patient does well for 2 wks. post onset of paresthesiaes • Guidelines: FVC <15 m. L/kgm MIF < 25 cm water

Psychological • • Fear Helplessness Communication Pain Sleep deprivation & hallucinosis Depression Visits from other GBS patients

Personal Experience • Bowes, Denise; The doctor as patient: an encounter with Guillain-Barre syndrome. Can Med Assoc J 131: 1343 -1348

Corticosteroids • Lancet 1993 242 pts. IV Methylprednisilone 500 mgm/day x 5. Ineffective May cause relapse

Plasma Exchange • Removal of the blood’s liquid soluble components including complement, immunoglobulin, immune complexes, cytokines and interleukins • A typical session removes about 60% of the body mass of plasma proteins which is replaced c saline, albumin & FFP • Done qod for 3 -5 sessions

Plasma Exchange • Various studies since 1985 • Time on ventilator reduced by ½ • Full strength regained at 1 year: Exchange 71%, Untreated 52% • Limitations: Limited availability Avoid with autonomic instability

Intravenous Immune Globulin • Originally used for immune insufficiency • Use as an immunosuppresant “seems to defy reason” • 1981 Rx for ITP • 5, 000 -10, 000 donors/batch. Diversity of Abs from large donor pool maximizes effect

IVIG • Mechanism of action- unknown ? Antiidiotypic antibody action ? Inhibition of cytokines ? “Sponging” of complement ? Binding to Fc receptors so macrophages can’t bind

IVIG • Dosage: 0. 4 gms/kgm/day x 5 c each dose given over 3 -4 hours preceded by IV diphenhydramine &/or po ibuprofen • Caution c renal insufficiency or Ig. A deficiency • 38 Center trial in 1997 • Equal to plasma exchange

J. H. C. 48 yo welder • June ’ 02 H. A. followed in 2 wks by Lt. Facial weakness • June ’ 03 Rhinorrhea & cough • August 6 Pain lt. Hip spreading over a few days to back 7 legs • August 15 Legs buckle c lt. facial weakness 1 wk later. LP c protein of 70. NCVs c prolonged F waves

• 1 Week post discharge, elevated titers to West Nile Virus • Follow up at 1 month- continued improvement
- Slides: 62