Guard Wire Plus Guard Wire Plus Export Aspiration
























- Slides: 37





Guard. Wire Plus™ Guard. Wire Plus Export Aspiration Catheter Inflation System: Micro. Seal® Adapter & EZ Flator™





Accu. Net Polyurethane 115 microns 3. 5 -3. 7 F

Angioguard™ XP / RX Cordis Polyurethane Crossing profile: 3. 2 -3. 9 Fr 100μm



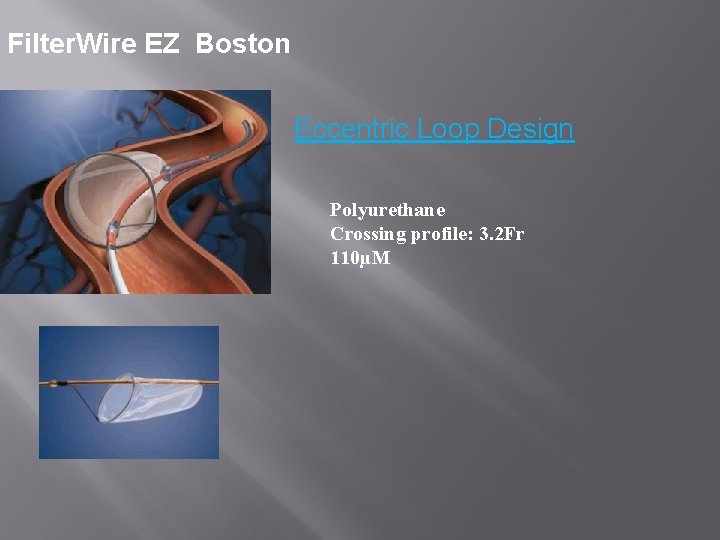
Filter. Wire EZ Boston Eccentric Loop Design Polyurethane Crossing profile: 3. 2 Fr 110µM

Spider. FX COVIDIEN ΑΝΕΞΑΡΤΗΤΟ ΣΥΡΜΑ Eccentric design heparin coated nitinol 50 mm – 3. 2 F

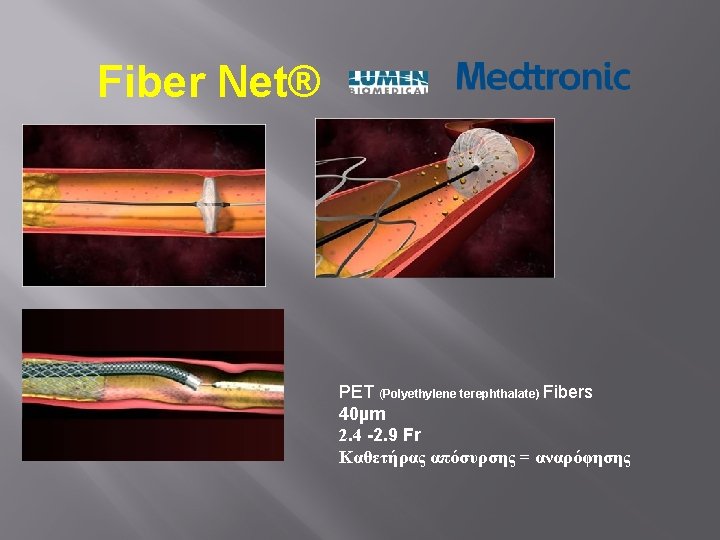
Fiber Net® PET (Polyethylene terephthalate) Fibers 40µm 2. 4 -2. 9 Fr Καθετήρας απόσυρσης = αναρόφησης

ΦΙΛΤΡΟ ΚΑΙ ΜΠΑΛΟΝΙ 40 μm Πρόσθετη προστασία κατά την μετα-διαστολή του νάρθηκα as compared with CAS performed with 1 distal EPD, double filtration with the Paladin System is effective in reducing the number of microemboli reaching the brain during CAS Double Filtration During Carotid Artery Stenting Using a Novel Post-Dilation Balloon With Integrated Embolic Protection JACC: Cardiovascular Interventions, 2019 -02 -25, Volume 12, Issue 4, Pages 395 -403

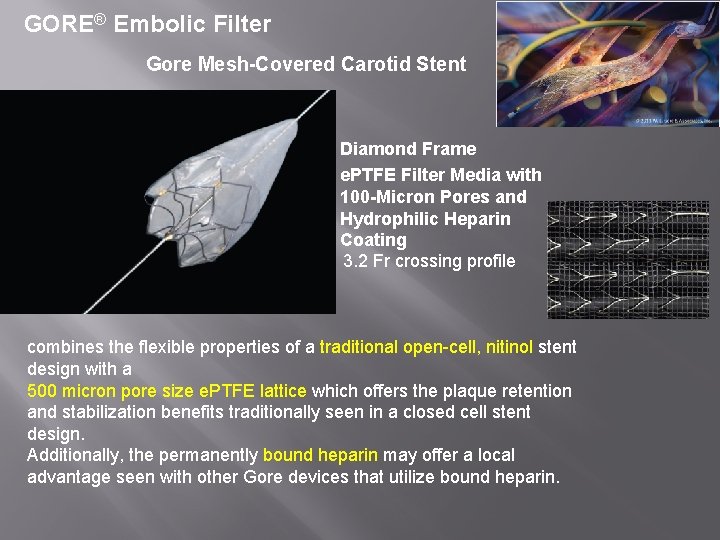
GORE® Embolic Filter Gore Mesh-Covered Carotid Stent Diamond Frame e. PTFE Filter Media with 100 -Micron Pores and Hydrophilic Heparin Coating 3. 2 Fr crossing profile combines the flexible properties of a traditional open-cell, nitinol stent design with a 500 micron pore size e. PTFE lattice which offers the plaque retention and stabilization benefits traditionally seen in a closed cell stent design. Additionally, the permanently bound heparin may offer a local advantage seen with other Gore devices that utilize bound heparin.


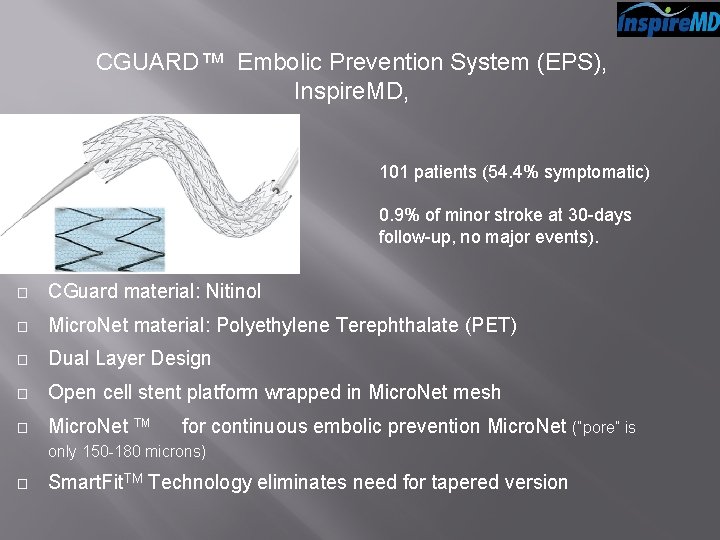
CGUARD™ Embolic Prevention System (EPS), Inspire. MD, 101 patients (54. 4% symptomatic) 0. 9% of minor stroke at 30 -days follow-up, no major events). � CGuard material: Nitinol � Micro. Net material: Polyethylene Terephthalate (PET) � Dual Layer Design � Open cell stent platform wrapped in Micro. Net mesh � Micro. Net TM for continuous embolic prevention Micro. Net (“pore” is only 150 -180 microns) � Smart. Fit. TM Technology eliminates need for tapered version


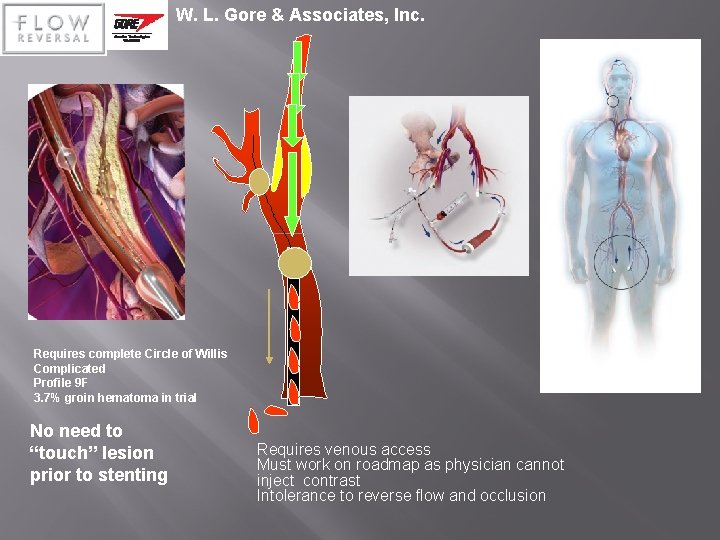
W. L. Gore & Associates, Inc. Requires complete Circle of Willis Complicated Profile 9 F 3. 7% groin hematoma in trial No need to “touch” lesion prior to stenting Requires venous access Must work on roadmap as physician cannot inject contrast Intolerance to reverse flow and occlusion

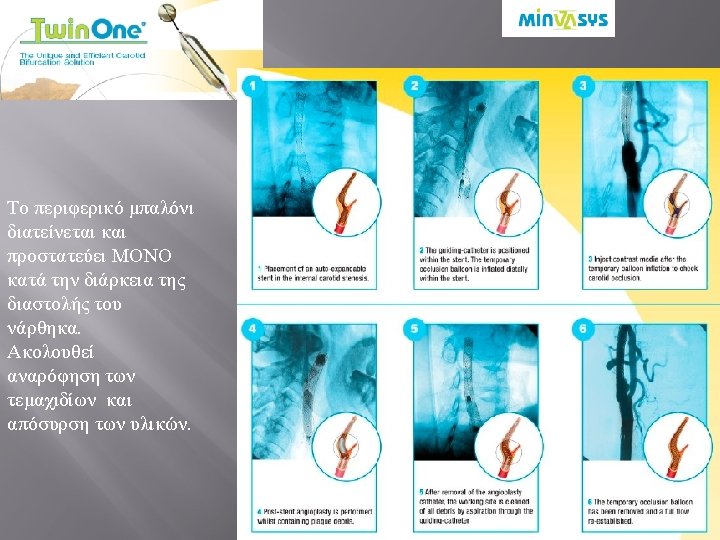
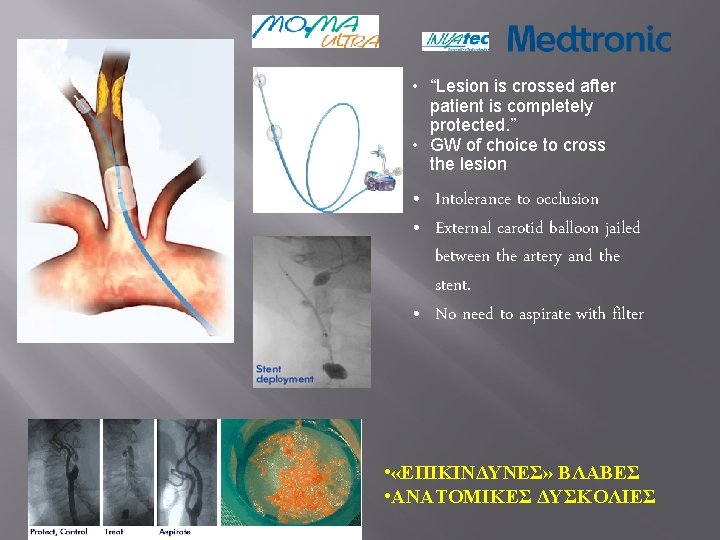
• “Lesion is crossed after patient is completely protected. ” • GW of choice to cross the lesion • Intolerance to occlusion • External carotid balloon jailed between the artery and the stent. • No need to aspirate with filter • «ΕΠΙΚΙΝΔΥΝΕΣ» ΒΛΑΒΕΣ • ΑΝΑΤΟΜΙΚΕΣ ΔΥΣΚΟΛΙΕΣ

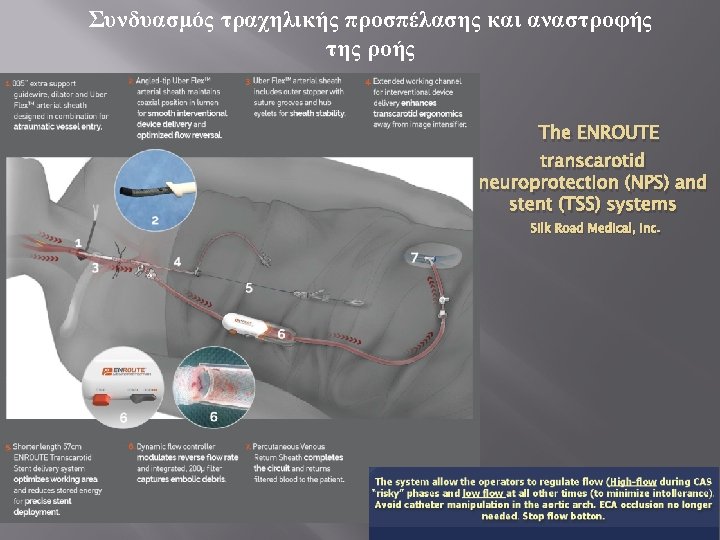
Συνδυασμός τραχηλικής προσπέλασης και αναστροφής της ροής The ENROUTE transcarotid neuroprotection (NPS) and stent (TSS) systems Silk Road Medical, Inc.

΄΄With the Enroute transcarotid NPS, we now have CEA-like neuroprotection and a simplified procedure that can fulfill the promise of CAS. ”









Evidence overview: benefit of cerebral protection devices during carotid artery stenting In conclusion, all currently available EPDs still result in some degree of cerebral embolization. No solid recommendation for a particular type of EPDs, if any, can be derived from literature J Cardiovasc Surg (Torino). 2017 Apr; 58(2): 170 -177. doi: 10. 23736/S 0021 -9509. 16. 09848 -7. Epub 2016 Dec 22 In patients undergoing CAS, both D-EPD and P-EPD provide similar levels of protection from peri-procedural stroke and 30 days mortality. Catheter Cardiovasc Interv. 2017 Apr; 89(5): 923 -931. doi: 10. 1002/ccd. 26842. Epub 2016 Nov 12. CAS with current carotid stent-EPD combinations is associated with low rates of inpatient stroke/TIA. There is no statistically significant difference in rates of inpatient stroke/TIA across device combinations Catheter Cardiovasc Interv. 2018 Oct 1; 92(4): 743 -749.

A New Milestone in Carotid Stenting? Reports show the use of meshcovered stents to be very safe with a 30 -day stroke rate of 1. 1% (Schneider et al. ) and 1. 3% (Stabile et al. ), respectively. Due to their small pore sizes, treatment of unstable plaques in high-risk lesions is a potential target for this stent type, widening the indication for carotid stenting. JA C C : C A R D I O V A S C U L A R I N T E R V E N T IO N S V O L. 1 1 , N O. 2 3 , 2 0 1 8 Mudra and Ledwoch. D E C EMB E R 1 0 , 2 0 1 8 : 2 4 1 2 – 3

A multi-institutional analysis of transcarotid artery revascularization compared to carotid endarterectomy Stroke (1. 0% TCAR vs 0. 3% CEA; P =. 62) and death (0. 3% TCAR vs 0. 7% CEA; P = NS) rates were similar at 30 days and comparable at 1 year (stroke, 2. 8% vs 2. 2% [P =. 79]; death 1. 8% vs 4. 5% [P =. 09]). The composite end point of stroke/death/myocardial infarction at 1 month postoperatively was 2. 1% vs 1. 7% (P = NS). TCAR was associated with a decreased rate of cranial nerve injury (0. 3% vs 3. 8%; P =. 01). These early data suggest that patients undergoing TCAR, even those with high-risk comorbidities, achieve broadly similar outcomes compared with patients undergoing CEA while mitigating cranial nerve injury Journal of Vascular Surgery, 2019, ISSN 0741 -5214, https: //doi. org/10. 1016/j. jvs. 2018. 09. 060.
