Grudgeball Rules 1 Each team starts out with
Grudgeball

Rules 1. Each team starts out with 10 X’s (points). 2. Objective: Be the last team with points. Knock other teams out of the game by taking away their points. 3. Earn the ability to knock off points by answering questions correctly as a team.

4. Answering correctly = knock 2 points off any team (or 1 point off 2 teams) 5. If you answer correctly, you get a chance to take a 2 or 3 pointer. • 2 pointer = knock off 4 X’s • 3 pointer= knock off 5 X’s • If you miss, you can still take off 2 X’s.

When a team loses all of their X’s, they are out. To get back in, they have to answer a question correctly to regain 3 X’s. Last team standing wins.

Any disrespectful or obnoxious behavior loses your team an X at the teacher’s discretion. If anyone on your team has their phone out, your team also loses an X. Repeat offenders taken out of game and have to write vocab definitions from textbook.

Enzymes are what type of macromolecule? §carbohydrates §lipids §proteins §nucleotides

Lipid functions: • Reserve energy • Cushion • Cell membranes • All of the above

The _____ of an enzyme is the area where the substrate binds • Product • Active site • Inhibitor • Substrate competitor

The monomer of carbohydrates: • Polysaccharide • Glycerol • Monosaccharide • Hydrocarbon

The number that designates the # of protons & neutrons: • Mass number • Electron number • Atomic weight

The number that designates the # of electrons: • Mass number • Electron number • Atomic weight

When water is removed to form a polymer: • Hydrolysis • Dehydration synthesis • Polymerization • Catalysis
How many fatty acids does a phospholipid contain? • One • Two • Three • Four

Water molecules sticking to other water molecules: • Adhesion • Cohesion • Solubility • Heat capacity

How many neutrons does Li have? • 6 • 3 • 7 • 4

The monomer of proteins: • Amino acids • Nitrogenous bases • Amino groups • Polypeptides
How many fatty acids does a triglyceride contain? • One • Two • Three • Four

How many valence electrons does N have? 5

___ is the unequal sharing of electrons. • Electronegativity • Productivity • Polarity • Electron affinity

Substance that donates H+ ions: • Acid • Base • Neutral

When a solution has a high H+ concentration, it is considered: • Acidic • Basic • Neutral

Elements with different mass numbers: • Isomers • Compounds • Isotopes • Molecules

Bond where electrons are shared: • Oxygen bond • Water bond • Ionic bond • Covalent bond

Particles in the outermost shell of an atom: • Electrons • Protons • Neutrons • Valence electrons

A mixture where 2 substances dissolve into 1 cohesive mixture • Suspension • Solution

How many protons does Li have? • 6. 9 • 3 • 7 • 4
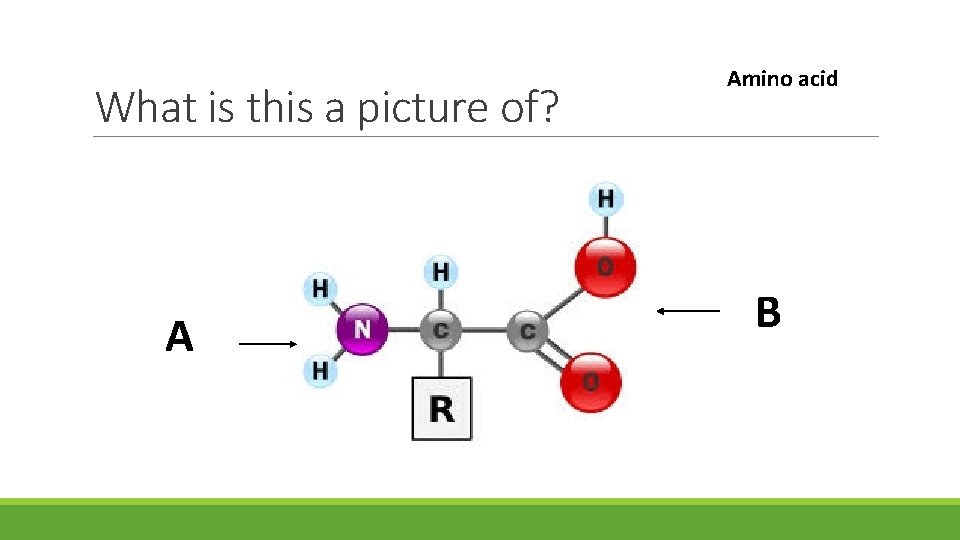
What is this a picture of? A Amino acid B

How many valence electrons does Li have? 1

Amino acids are held together by: • Covalent bonds • Hydrogen bonds • Ionic bonds • Peptide bonds
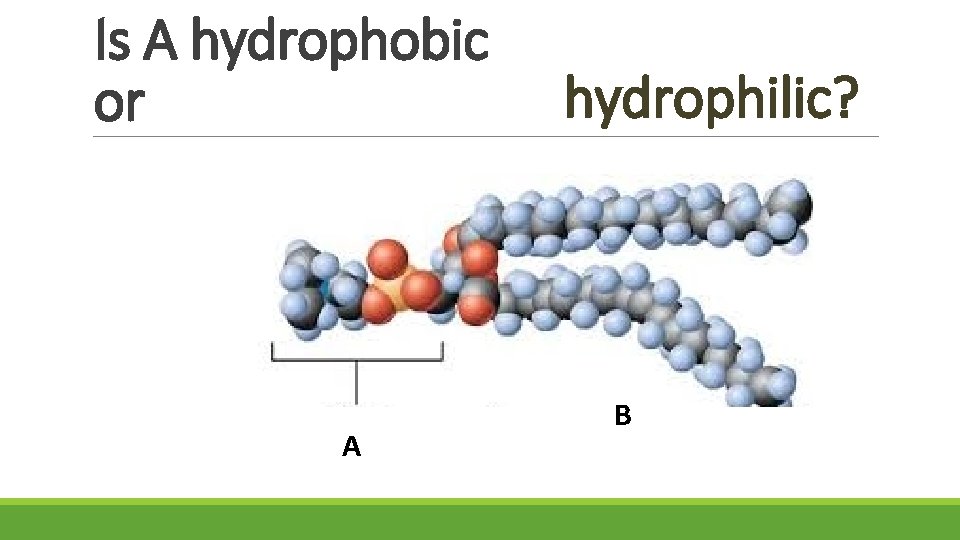
Is A hydrophobic or A hydrophilic? B

Water molecules sticking to anything other than water molecules: • Adhesion • Cohesion • Solubility • Heat capacity

When a solution has a high OH- concentration, it is considered: • Acidic • Basic • Neutral

The ___ in water is partially negative: • Hydrogens • Hydrogen and oxygen • Oxygen • Polarity

Dissolving agent: • Solution • Solvent • Solute

Functions of carbs: • Energy storage • Cell structure • Both of the answers above • Cushion

Polymer of carbs: • Monosaccharide • Disaccharide • Polysaccharide • Glucose

Contains all single bonds: • Trans fat • Unsaturated fat • Cholesterol • Saturated fat

Type of lipid found in cell membranes: • Phospholipid • Saturated fat • Unsaturated fat • Cholesterol

A substance is considered organic if it: • Contains C & H • Contains C & O • Contains C, H & O
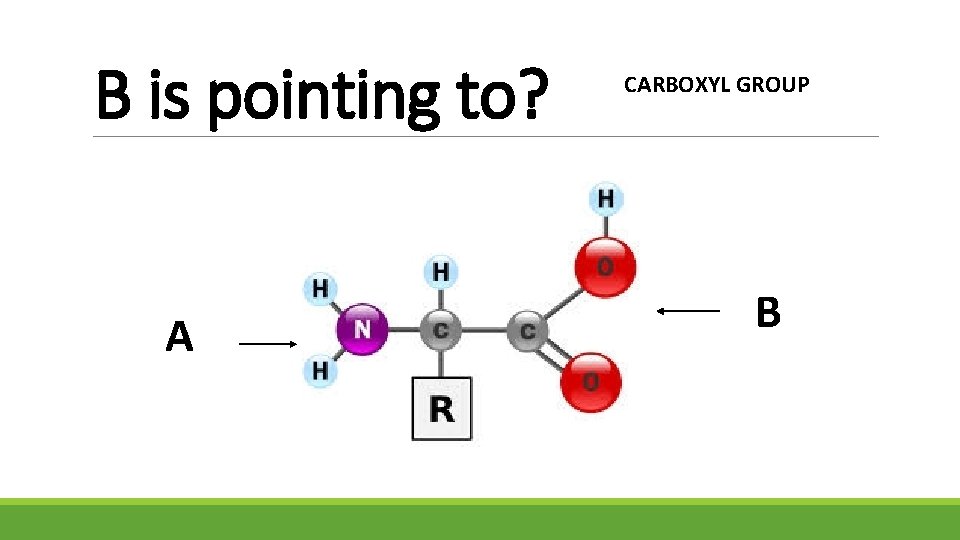
B is pointing to? A CARBOXYL GROUP B

Carries cholesterol away from cells & tissues • Steroids • HDL • LDL • Saturated fats
- Slides: 41