GROUP II CATIONS As3 As5 Sb3 Sb5 Sn2

GROUP II CATIONS As+3, As+5, Sb+3, Sb+5, Sn+2, Cu+2, Cd+2, Bi+3 , Hg+2, Pb+2

GROUP II CATIONS As+3, As+5, Sb+3, Sb+5, Sn+2, Cu+2, Cd+2, Bi+3 The specific reactions of Group II cations when they are alone = The experiments you will perform with known samples of group II cations.

Cu +2 1 - The solutions of Cu salts have blue or bluish-green color. You can predict the presence of Cu in your sample just by looking at the color of your sample. 2 - Reaction with K 4[Fe(CN)6] (potassium ferrocyanide) : Red-brown precipitates are formed and they are soluble in diluted ammonia 2 Cu+2 + [Fe(CN)6] – 4 → Cu 2[Fe(CN)6] ↓ 3 - Reaction with KCN (potassium cyanide): Firstly greenish-yellow Cu(CN)2 ↓ precipitates are formed and upon excess adition of KCN, it will be dissolved by formation of K 3[Cu(CN)4] (potassium cyanocuprate)

Cd+2 1 - Reaction with H 2 S: colloidal and yellow colored Cd. S is formed and it is soluble in HNO 3. Cd+2 +H 2 S → Cd. S ↓ (yellow) + 2 H+ 3 Cd. S + 8 H+ + 2 NO 3– → 3 Cd+2 + 3 S + 2 NO + 4 H 2 O 2 - Reaction with KCN: In the beginning, white colored Cd(CN)2 precipitates are formed and upon addition of excess KCN it is dissolved by formation of K 2[Cd(CN)4] (potassium tetracyanocadmate(II)) complex. This complex can react with H 2 S to produce Cd. SO 4 + 2 KCN → Cd(CN)2 + K 2 SO 4 Cd(CN)2 + 2 KCN → K 2[Cd(CN)4] + H 2 S → Cd. S ↓ + 2 KCN +2 HCN Yellow The last reaction is the difference between Cd+2 and Cu+2 , K 3[Cu(CN)4] complex does not react with H 2 S while K 2[Cd(CN)4] reacts.

Bi+3 ** Main oxidation states of bismuth are +3, – 3, +5. Bi-3 behaves strong reducing agent while Bi+5 is strong oxidizing agent. 1 - Reaction with Na 2 Sn. O 2 (Sodium stannite ): Bismuth is reduced to black colored metallic Bi 0 2 Bi(OH)3 + 3 Na 2 Sn. O 2 → 2 Bi 0↓+ 3 Na 2 Sn. O 3 + 3 H 2 O black 2 - Reaction with KI: Bi+3 + KI→ Bi. I 3 ↓ black precipitate Bi. I 3 + KI ↔ [Bi. I 4]– Soluble complex

Sn +2 (Tin) The hydrolysis of Sn+2 salts produce white precipitates of Sn(OH)Cl. 1 - Reaction with H 2 S: Brown Sn. S precipitates. Brown precipitate might turn to black later 2 - Tin reduce Bi+3 to black Bi 0 in basic medium. Add Na. Bi. O 3 (sodium bismuthate) prepared in basic medium.

Sn +2 (Tin) 3 - Luminescence test: The sample is directly used without any pretreatment. • Fill a test tube with cold tap water. • Put 5 m. L sample into a small beaker. • Add 3 m. L HCl into the beaker. • Add Zn granules (2 microspatula) into the beaker. • Immerse the test tube into the beaker. • Hold the bottom of the test tube directly into the reducing zone of the flame) • Blue shining (luminescence) indicates the presence of Sn+2 • Luminescence is seen outside of the test tube.

As+3, As+5 (Arsenic) They both form strong covalent bonds with oxygen in water As+3 : As. O 3– 3, As. O 2– (arsenite) As+5 : As. O 4– 3 (arsenate) 1 -Reaction with Ag. NO 3 yields yellow colored Ag 3 As. O 4 (silver arsenate). 3 Ag+ + As. O 4 - → Ag 3 As. O 4↓ yellow

Sb+3, Sb+5 (Antimony) It can produce white colored Sb. OCl precipitates in water. 1 - Reaction with H 2 S: Orange Sb 2 S 3 (antimony sulfide) precipitates 2 Sb+3 + 3 H 2 S → Sb 2 S 3 ↓ + 6 H+ orange. . . brown 2 - Reaction with Na. OH: White colored Sb. OCl precipitates. Sb. Cl 3 + Na. OH → Sb. OCl ↓ + HCl + Na. Cl white

Pb 2+ (Lead) Reaction with potassium chromate (K 2 Cr. O 4): yellow lead chromate precipitates Pb 2+ + K 2 Cr. O 4 → Pb. Cr. O 4↓(s) + 2 K+ yellow precipitates Hg+2 (Mercury) Copper strip (Amalgam test): A piece of copper wire is cleaned with 8 -10 drops of concentrated HNO 3 solution in the watch glass, then it is dried by filter paper and mercury sample solution is added to the copper strip. Mercury ions reduce and coat the copper strip (bright gray colored).
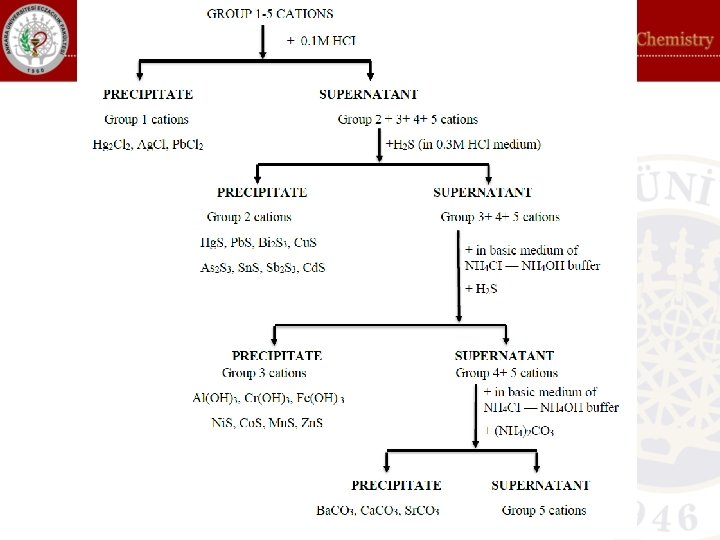
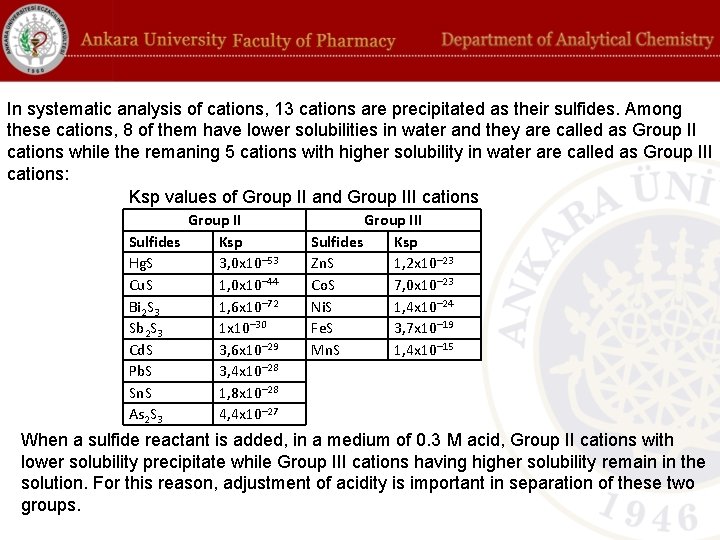
In systematic analysis of cations, 13 cations are precipitated as their sulfides. Among these cations, 8 of them have lower solubilities in water and they are called as Group II cations while the remaning 5 cations with higher solubility in water are called as Group III cations: Ksp values of Group II and Group III cations Group II Sulfides Ksp Hg. S 3, 0 x 10– 53 Cu. S 1, 0 x 10– 44 Bi 2 S 3 1, 6 x 10– 72 Sb 2 S 3 1 x 10– 30 Cd. S 3, 6 x 10– 29 Pb. S 3, 4 x 10– 28 Sn. S 1, 8 x 10– 28 As 2 S 3 4, 4 x 10– 27 Group III Sulfides Ksp Zn. S 1, 2 x 10– 23 Co. S 7, 0 x 10– 23 Ni. S 1, 4 x 10– 24 Fe. S 3, 7 x 10– 19 Mn. S 1, 4 x 10– 15 When a sulfide reactant is added, in a medium of 0. 3 M acid, Group II cations with lower solubility precipitate while Group III cations having higher solubility remain in the solution. For this reason, adjustment of acidity is important in separation of these two groups.


Thioacetamide can be used as sulfide source. Hydrogen sulfide will be produced when thioacetamide solution is heated. For this reason, the test tube containing your sample will be heated in a water bath after addition of required amount of thioacetamide. Since the produced hydrogen sulfide will immediately react with cations in the sample, hazardous effects of hydrogen sulfide such as rotten egg smell will be reduced. In the analysis, the flow chart will be followed. Sulfides of Group II cations have different colors so you may have preliminary information at the beginning of the analysis: Bi 2 S 3, Hg. S, Cu. S, Pb. S As 2 S 3, As 2 S 5 Cd. S Sb 2 S 3, Sb 2 S 5 Sn. S = Black = Yellow = Pale yellow = Orange / red / brown = Brown
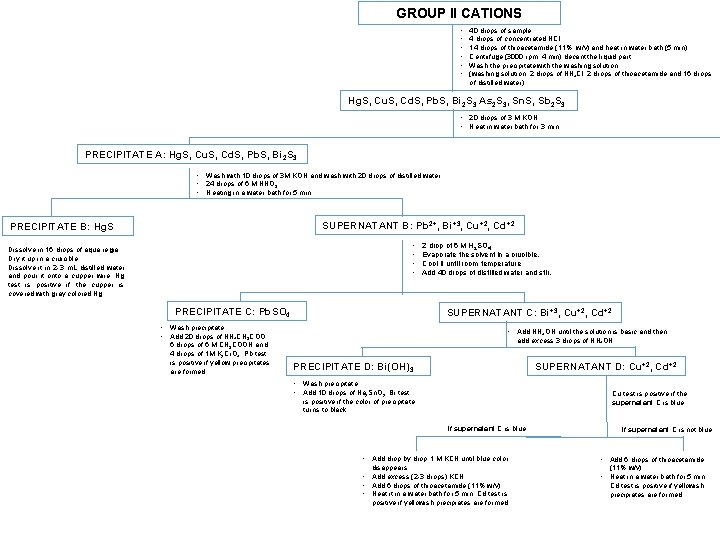
GROUP II CATIONS • • • 40 drops of sample 4 drops of concentrated HCl 14 drops of thioacetamide (11%, w/v) and heat in water bath (5 min) Centrifuge (3000 rpm, 4 min) decant the liquid part Wash the precipitate with the washing solution (washing solution: 2 drops of NH 4 Cl, 2 drops of thioacetamide and 16 drops of distilled water) Hg. S, Cu. S, Cd. S, Pb. S, Bi 2 S 3 As 2 S 3, Sn. S, Sb 2 S 3 • 20 drops of 3 M KOH • Heat in water bath for 3 min PRECIPITATE A: Hg. S, Cu. S, Cd. S, Pb. S, Bi 2 S 3 • Wash with 10 drops of 3 M KOH and wash with 20 drops of distilled water • 24 drops of 6 M HNO 3 • Heating in a water bath for 5 min SUPERNATANT B: Pb 2+, Bi+3, Cu+2, Cd+2 PRECIPITATE B: Hg. S • • • Dissolve in 16 drops of aqua regia. • Dry it up in a cruicible • Dissolve it in 2 -3 m. L distilled water and pour it onto a cupper wire. Hg test is positive if the cupper is covered with gray colored Hg. PRECIPITATE C: Pb. SO 4 SUPERNATANT C: Bi+3, Cu+2, Cd+2 • Wash precipitate • Add 20 drops of NH 4 CH 3 COO, 6 drops of 6 M CH 3 COOH and 4 drops of 1 M K 2 Cr. O 4. Pb test is positive if yellow precipitates are formed. 2 drop of 6 M H 2 SO 4 Evaporate the solvent in a crucible. Cool it until room temperature Add 40 drops of distilled water and stir. • Add NH 4 OH until the solution is basic and then add excess 3 drops of NH 4 OH SUPERNATANT D: Cu+2, Cd+2 PRECIPITATE D: Bi(OH)3 • Wash precipitate • Add 10 drops of Na 2 Sn. O 2. Bi test Cu test is positive if the supernatant C is blue. is positive if the color of precipitate turns to black. If supernatant C is blue • Add drop by drop 1 M KCN until blue color disappears. • Add excess (2 -3 drops) KCN • Add 6 drops of thioacetamide (11% w/v) • Heat it in a water bath for 5 min. Cd test is positive if yellowish precipiates are formed. If supernatant C is not blue • Add 6 drops of thioacetamide (11% w/v) • Heat in a water bath for 5 min. Cd test is positive if yellowish precipiates are formed.
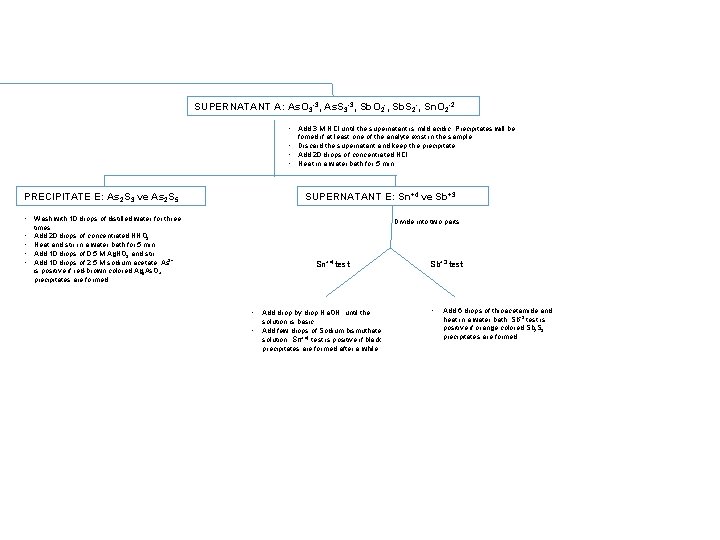
SUPERNATANT A: As. O 3 -3, As. S 3 -3, Sb. O 2 -, Sb. S 2 -, Sn. O 2 -2 • Add 3 M HCl until the supernatant is mild acidic. Precipitates will be fomed if at least one of the analyte exist in the sample. • Discard the supernatant and keep the precipitate. • Add 20 drops of concentrated HCl. • Heat in a water bath for 5 min. SUPERNATANT E: Sn+4 ve Sb+3 PRECIPITATE E: As 2 S 3 ve As 2 S 5 • Wash with 10 drops of distilled water for three • • Divide into two parts times. Add 20 drops of concentrated HNO 3. Heat and stir in a water bath for 5 min Add 10 drops of 0. 5 M Ag. NO 3 and stir. Add 10 drops of 2. 5 M sodium acetate. As 3+ is positive if red-brown colored Ag 3 As. O 4 precipitates are formed. Sn+4 test • • Add drop by drop Na. OH until the solution is basic. Add few drops of Sodium bismuthate solution. Sn+4 test is positive if black precipitates are formed after a while. Sb+3 test • Add 6 drops of thioacetamide and heat in a water bath. Sb+3 test is positive if orange colored Sb 2 S 3 precipitates are formed.

ATTENTION!!! *While working with KCN (For Cu+2 and Cd+2 tests), the medium must be strictly checked with litmus paper for its basicity. If the solution is acidic, then highly toxic HCN gas is evolved. KCN + H+ → HCN (g)
- Slides: 17