Group 17 Trends Group 17 Trends Describe and

Group 17 Trends…

Group 17 Trends… Describe and explain the trend in boiling points down group 17 Swap with someone behind/in front of you and mark their work (in a different colour!)

Group 17 Trends… Describe and explain the trend in boiling points down group 17 • Boiling Points increase • Down the group the number of electron shells increases • Leads to increased instantaneous dipole-induced dipole forces • More energy is required to overcome these forces
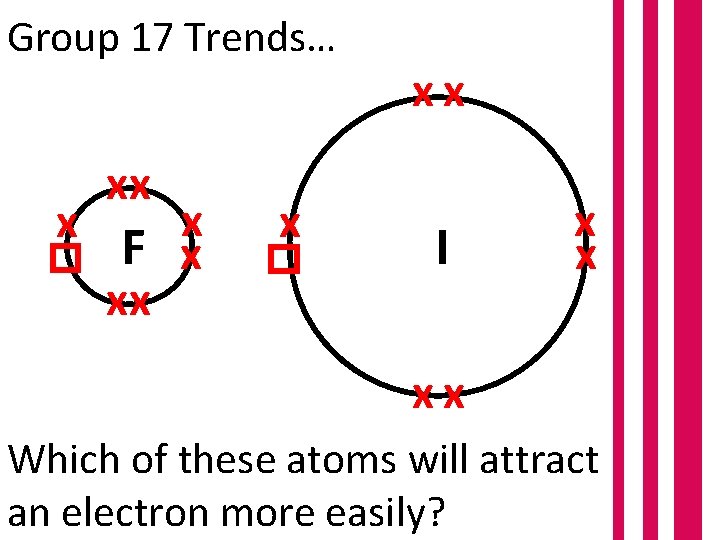
Group 17 Trends… x xx x F x xx F I x x xx Which of these atoms will attract an electron more easily?

Group 17 Trends… Iodine has a larger atomic radius due to increased shielding and a decreased EFFECTIVE nuclear charge. Less attractive to electrons and therefore less reactive.

Group 17 Trends… Which of the following reactions will work? Cl 2 + 2 HBr Cl 2 + 2 H+ + 2 Br- Br 2 + 2 HCl Br 2 + 2 H+ + 2 Cl-

Group 17 Trends… Which of the following reactions will work? Cl 2 + 2 HBr Br 2 + 2 HCl • Chlorine is more electronegative • It removes the extra electron from the Bromine ion • Chlorine is REDUCED • Bromine is OXIDISED

Group 17 Trends… Define Disproportionation: A reaction where the same element is both oxidised and reduced.
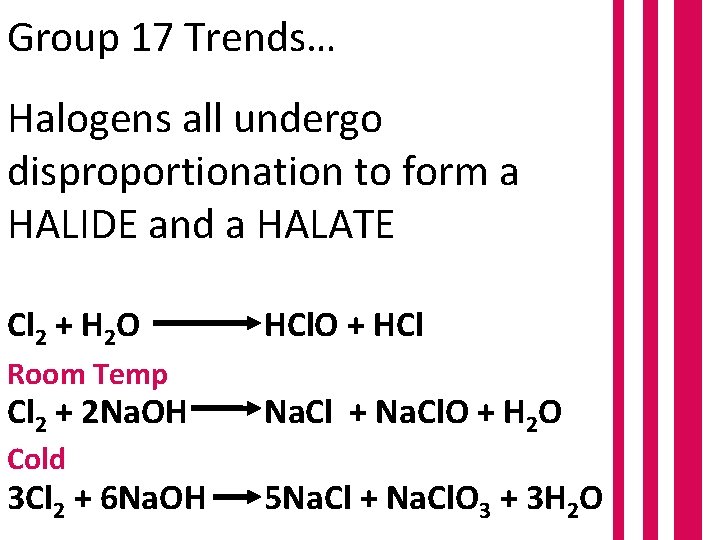
Group 17 Trends… Halogens all undergo disproportionation to form a HALIDE and a HALATE Cl 2 + H 2 O Room Temp Cl 2 + 2 Na. OH Cold 3 Cl 2 + 6 Na. OH HCl. O + HCl Na. Cl + Na. Cl. O + H 2 O 5 Na. Cl + Na. Cl. O 3 + 3 H 2 O

Group 17 Trends… Suggest an equation for the reaction of Iodine with cold sodium hydroxide 3 Cl 2 + 6 Na. OH 5 Na. Cl + Na. Cl. O 3 + 3 H 2 O 3 I 2 + 6 Na. OH 5 Na. I + Na. IO 3 + 3 H 2 O

Group 17 Trends… When undertaking experiments with halogen compounds we need to be able to identify them quantitatively: Colour of Gas Colour in Water Colour in Cyclohexane Chlorine Yellow/ Green Pale Green Bromine Orange Iodine Violet Brown Violet Halogen

Group 2 Trends… On mini whiteboards Predict the reactivity of Group 2 metals with water and JUSTIFY your reasons.

Group 2 Trends… Reaction with acids M(s) + 2 HX(aq) MX 2(aq) + H 2(g) M(s) + H 2 X(aq) MX(aq) + H 2(g)

Group 2 Trends… Write balanced equations and identify which elements have been oxidised/reduced for these reactions: • Strontium and Sulphuric Acid • Magnesium and Nitric Acid

Group 2 Trends… Reaction with water M(s) + 2 H 2 O(l) M(OH)2 (aq) + H 2(g) The hydroxides are white compounds once the solvent (water) has been evaporated and form ALKALINE solutions
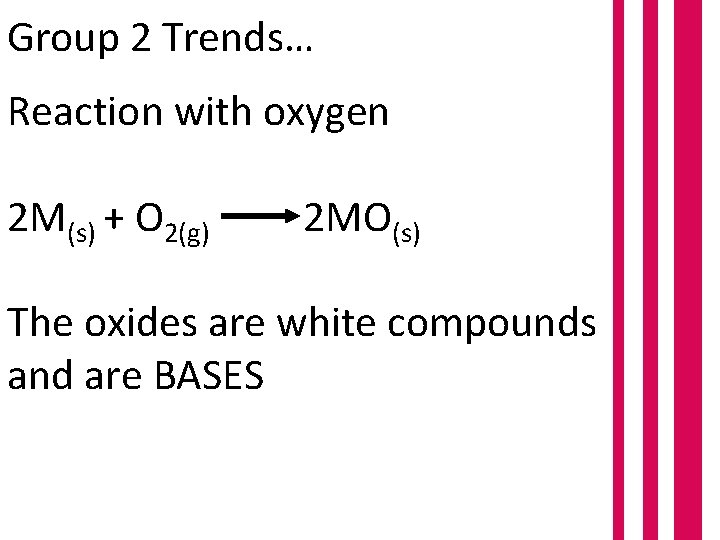
Group 2 Trends… Reaction with oxygen 2 M(s) + O 2(g) 2 MO(s) The oxides are white compounds and are BASES

Group 2 Trends… Compare the solubilities of the group 2 hydroxides and oxides. • Compounds dissolve if the energy released on dissolving is greater than the energy required to break down the ionic lattice • Group 2 hydroxides release more energy than group 2 oxides when they dissolve
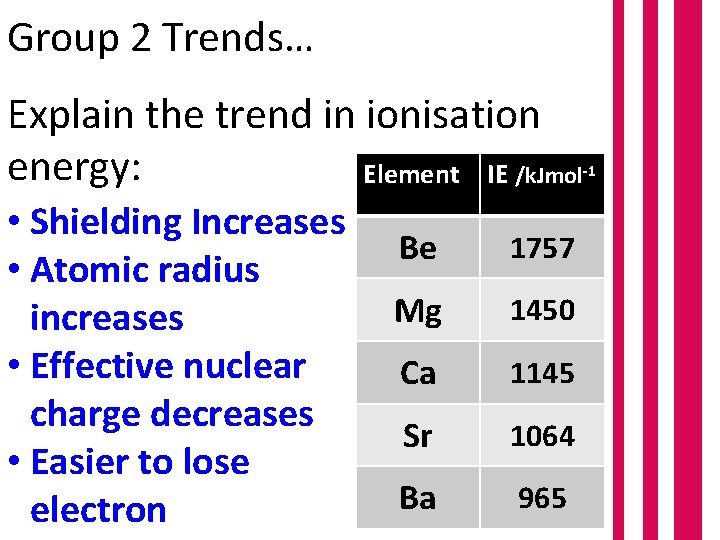
Group 2 Trends… Explain the trend in ionisation energy: Element IE /k. Jmol • Shielding Increases • Atomic radius increases • Effective nuclear charge decreases • Easier to lose electron Be 1757 Mg 1450 Ca 1145 Sr 1064 Ba 965 -1

Group 2 Trends… Explain the trend in ionisation energy: Element IE /k. Jmol Therefore, which is more reactive? Was your prediction correct? Be 1757 Mg 1450 Ca 1145 Sr 1064 Ba 965 -1

Group 2 Trends… Write balanced equations for: • Barium Oxide and Water • Strontium Oxide and Hydrochloric acid • Beryllium Hydroxide and Sulphuric Acid

Group 2 Trends… The products of reacting Group 2 metals (and metal oxides) to water are: • Be(OH)2 (s) • Mg(OH)2 (aq) – partially (~25%) dissolves • Ca(OH)2 (aq) What is the difference in the resulting solutions of Mg(OH)2 and Ca(OH)2 if 3 mol of each are reacted with water?

Group 2 Trends… 3 mol of Ca(OH)2 = 6 mol OH 3 mol of Mg(OH)2 =0. 25 * 6 mol OH=1. 5 mol OHOH- is the species responsible for alkalinity. Therefore Ca(OH)2 is more alkaline than Mg(OH)2

Group 2 Trends… There are three uses of Group 2 compounds that you need to be aware of: • Soil neutralisation • Indigestion relief • Construction - Ca(OH)2 - Mg(OH)2 - Ca. CO 3
- Slides: 23