Greetings from CMC Vellore To Christ be all

Greetings from CMC Vellore… To Christ be all glory !

Re-use Dr. Suceena Alexander, MD, DM, FASN Professor, Dept. of Nephrology CMC Vellore 2
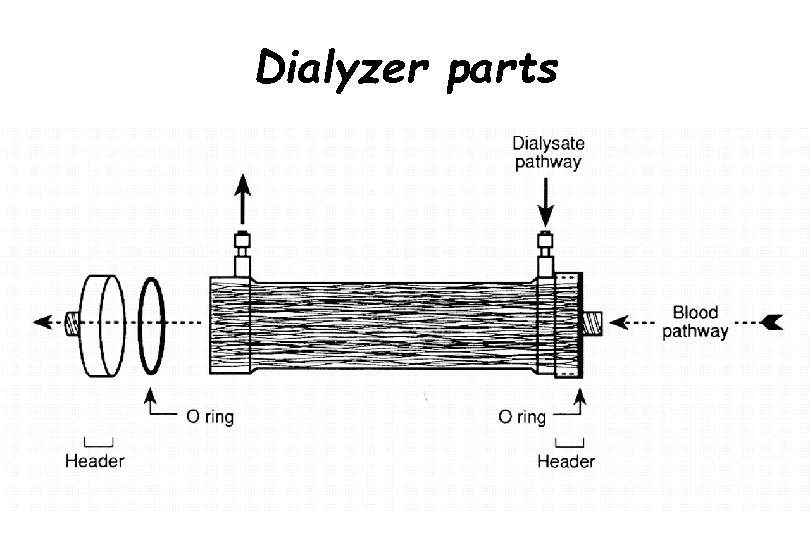
Dialyzer parts

Historical Perspective • 1914 - The first apparatus for hemodialysis in animals, made by Abel et al. at the beginning of 20 th century, was cleaned with acid-pepsin to digest blood, dis- infected with thymol, and reused for up to 30 experiments for as long as 8 months!! • WHY? ? saving time and labor!

Current perspective • Most debatable aspects of hemodialysis • Banned by law in Japan • Most European countries do not Reuse dialyzers (except Portugal, Malta and East European countries) • FDA allowed in US • Reuse practiced in rest of the World • Reuse of certain other devices, tubings, and end caps is even more controversial

India • ISN Guideline Recommendation- Hollow fibre dialysers may be reprocessed in order to reduce the cost of the hemodialysis procedure. Prior to reprocessing the dialyzer should be checked for the manufacturers labeling it for single or multiple uses. • ISN Guideline Recommendation- The process has been associated with safe and effective dialysis provided the process is strictly adhered to and monitored •

India • ISN Guideline Recommendation- Tubings, end caps, O rings and dialyser headers may be reused, however no tests of performance are available to monitor the process. – Venous and arterial transducer protectors SHOULD NOT be reused.
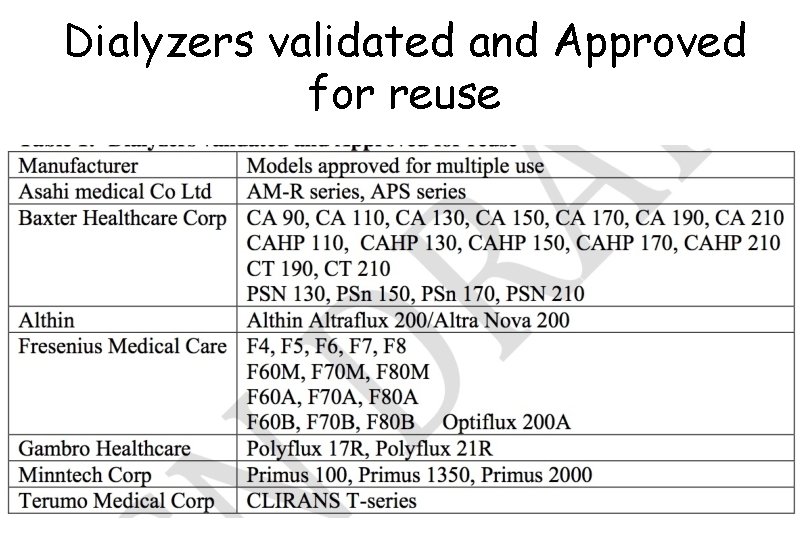
Dialyzers validated and Approved for reuse

Pre-processing of new dialyzers • NKF-K/DOQI guidelines recommend that each new dialyzer be pre-processed to obtain an accurate estimate of the FBV before first use.
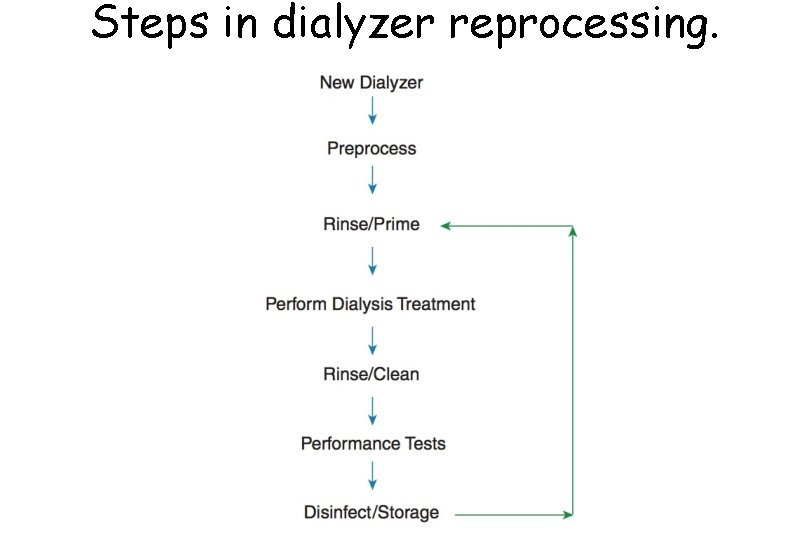
Steps in dialyzer reprocessing.
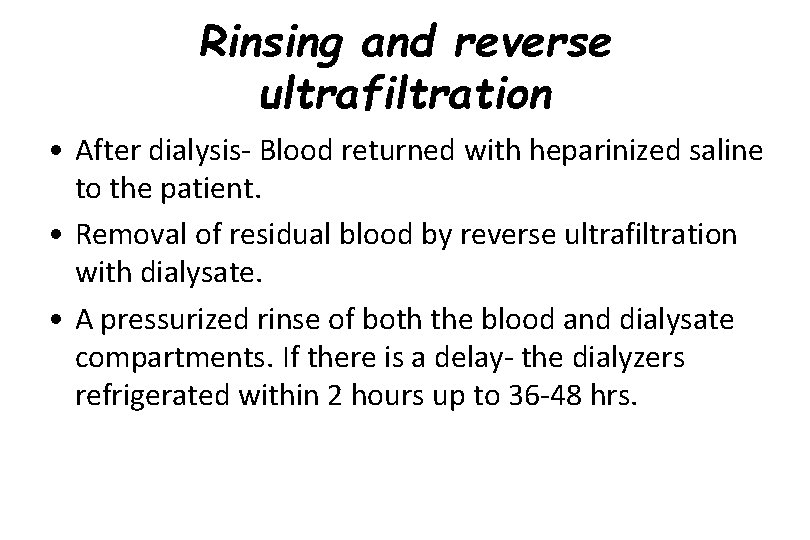
Rinsing and reverse ultrafiltration • After dialysis- Blood returned with heparinized saline to the patient. • Removal of residual blood by reverse ultrafiltration with dialysate. • A pressurized rinse of both the blood and dialysate compartments. If there is a delay- the dialyzers refrigerated within 2 hours up to 36 -48 hrs.

Cleaning • Water- Prolonged rinse with reverse-osmosis water followed by reverse ultrafiltration • Bleach- Sodium hypochlorite (bleach) < 0. 6% Increases albumin losses in high-flux dialyzers Increases ultrafiltration coefficient and causes overt membrane damage in cellulosic membranes • Other cleaning agents– Hydrogen peroxide 3% or – Renalin: peracetic acid/hydrogen peroxide/acetic acid mixtures

Tests of dialyzer performance • Pressure test for leaks – manually or automated. – instilling pressurized air or nitrogen into the blood side of the dialyzer or by producing a vacuum in the dialysate side. – also screens for defects in the dialyzer O-rings, potting compound, and end-caps.

Tests of dialyzer performance • Fiber bundle volume (FBV) or Total compartment Volume (TCV) – the total space within the blood compartment of hollow fiber hemodialyzer. – correlates closely with dialyzer surface area, a major determinant of solute clearance. – main criterion that allows a dialyzer to be reused. – reduction of 20% corresponds to 10% reduction in urea clearance, the maximum decrease acceptable for continued use.

Tests of dialyzer performance • In vitro dialyzer ultrafiltration coefficient (In vitro Kuf) – measured by determining the volume of water passing through the membrane at a given pressure and temperature. – a change in the KUf reflects changes in membrane resistance, as well as surface area – No clinical impact using machines with ultrafiltration control

Tests of dialyzer performance • Conductivity-based clearance – On-line determination of sodium or ionic clearance, is comparable to urea clearance – acceptable method of monitoring dialyzer performance ISN Guideline Recommendation- Performance testing should be performed for all reused dialyzers.

Disinfection/sterilization • High-level disinfection differs from sterilization as it may not destroy spores. • Current standards require- high-level disinfection

Disinfection/sterilization • Germicides – Chemicals: • Renalin- Peracetic acid/hydrogen peroxide/acetic acid mixtures – minimum contact time of 12 hours • Formaldehyde (vapour and liquid): – 4% at room temperature for 24 hours – 1% at 400 C for 24 hours • Glutaraldehyde (Diacide) 2%

Disinfection/sterilization • Germicides – Non-chemicals • Heated water at 1050 C (Kaufman, 1992) • Heated 1. 5% citric acid at 950 C (Levin et al. , 1995)

Disinfection/sterilization • Documenting the presence of germicide – Formaldehyde: FD&C blue dye no. 1 - light blue color indicates that formaldehyde is present. – Renalin: Potassium iodide or starch testing strips

Disinfection/sterilization • Germicide removal – Automated or manual – Flushing of the blood compartment, followed by flushing of the dialysate compartment – Air removed from the arterial line before flushing – Dialyzer rotated at intervals during flushing – Blood circuit checked for residual germicide Eg. modified Schiff reagent formaldehyde – Starch iodide paper for peracetic acid and sodium hypochlorite

Disinfection/sterilization • Heat sterilization – Reverse-osmosis water is instilled, and the dialyzers are heated in a 1050 C convection oven for 20 hours – 1. 5% citric acid solution allows for heating at 950 C for 20 hours – Polysulfone is the only membrane that is heat resistant for clinical use – Potting compounds must meet certain design specifications – Absence of color change with litmus or p. H papers for citric acid ensures removal

Disinfection/sterilization • ISN Guideline Recommendation- Chemical disinfectant reprocessing is the usual method, Polysulfone dialyzers may be processed by heat. • Final inspection – Dialyzers should not be used if they have an abnormal appearance (e. g. , brownish or blackish discoloration, clots in the header, bands of clotted fibers). •

Disinfection/sterilization • Cleaning and disinfecting agents: - Preferably be available online • Overhead tanks- 25 to 50 litres capacity • Refilled with fresh solutions every week, after cleaning. • All tanks and piping for sodium hypochlorite should be composed of medical grade PVC, and those formaldehyde glutaraldehyde and peracetic acid should be composed of 316 SS.

Automated Reuse Machines • Early Dialyzer Reuse Machines (1974– 1976) – The first machine for automatic dialyzer reuse was designed by De Palma et al. in Encino, CA, in 1974. • Second-Generation Reuse Machines (1977– 1983) – Several companies designed automated equipment. – In the late 1970 s and early 1980 s at least six reuse machines were commercially available.

Automated versus manual systems • Advantages of automated system: – Process multiple dialyzers simultaneously – Cleansing cycles- highly reproducible – Quality control tests- FBV, KUF, and pressure holding are built in. – Dialyzer labels automatically printed. – Computerized analysis of records • ISN Guideline Recommendation- Where possible automated reprocessing should be used, however for cost reasons manual reprocessing only may be feasible.

Advantages and disadvantages of dialyzer reuse • Advantages – widespread use of costlier dialyzes: Highefficiency dialyzers, high-flux synthetic dialyzers – Reduced exposure to residual industrial chemicals used in the manufacture of new dialyzers: residual amounts of ethylene oxide, perfluorocarbons. – Reduced “First use syndrome”: cellulosic (Cuprophan) dialyzers

Advantages and disadvantages of dialyzer reuse • Advantages – Enhanced dialyzer biocompatibility/reduced immune system activation (with unsubstituted cellulose membranes and when bleach is not used) – ISN Guideline Recommendation- There is no improvement in biocompatibility of reprocessed dialysers made of synthetic material. – Reduction in TREATMENT COST

Advantages and disadvantages of dialyzer reuse • Disadvantages – Exposure of patient and personnel to chemicals • Formaldehyde: – Anti-N antibodies – Acute formaldehyde reactions – Itching – Bacterial/endotoxin contamination of dialyzer • water used to rinse and clean the dialyzers and to prepare the germicides – Anaphylactoid reactions with Renalin and ACE inhibitors

Advantages and disadvantages of dialyzer reuse • Disadvantages – Loss of dialyzer mass transfer (clearance) and ultrafiltration capacity • FBV at least 80% • Ensure adequate heparin anticoagulation • Bicarbonate dialysate containing a small amount of citrate is beneficial.

Advantages and disadvantages of dialyzer reuse • Disadvantages – Transmission of an infectious agent from one dialyzer to another during the reprocessing procedure – Loss of β 2 -microglobulin clearance • High-flux dialyzer when resued with Renalin and without bleach

Noneconomic Difference Between Dialyzer Reuse and Single Use

Headers and the O rings • The headers and the O rings- placed in glutaraldehyde while the dialyzer is being reprocessed. • Improper placement of the O ring or failure to replace- results in blood leak

Reuse of Tubings • No literature supports the reuse of blood tubings • Washed free of blood by treated water and then with a 1. 6% solution of sodium hypochlorite. • Again rinsed with water and then connected to a supply of 4% formaldehyde. • No objective tests of performance are available for blood tubings.

Quality assurance/ Audits • Documentation of all aspects of present and past reuse of a patient's individual dialyzers • Each patient's dialyzer is individually labeled with a unique identifier • A log is also kept of the weekly test results of the disinfectant stock • Percentage of the program's reused dialyzers are cultured weekly for bacteria and checked for proper disinfectant concentration. • 24 -hour temperature-recording devices • Preventive maintenance program

Quality assurance/ Audits • ISN Guideline Recommendation- The process of reusing dialyzers should be monitored for efficacy and safety. – This is the duty of the attending nephrologist • ISN Guideline Recommendation- The sp. Kt/V with new and reused dialyzers should be monitored at least once a month.

Personnel and physical plant considerations • Use of protective eyewear and clothing is stressed. • Current (1990) maximum allowable time-weighted average (TWA) exposure formaldehyde is 1 ppm and for short-term exposure is 3 ppm. • Maximum exposure limits for hydrogen peroxide is 1 ppm TWA and for glutaraldehyde is 0. 2 ppm. • There are no current exposure limits for peracetic acid.

Personnel and physical plant considerations • Comprehensive training course required • ISN Guideline Recommendation- Operators concerned with the actual reprocessing procedure should wear appropriate protective gear for all reprocessing procedures. • This should include waterproof apron, goggles or spectacles.

Reuse for Viro positive patients • The risk of transmission of hepatitis B is approximately 30% after a mucosal exposure, around 3% for Hepatitis C and 0. 3% for HIV. • ISN Guideline Recommendation- Separate Reprocessing areas should be utilized for dialyzers of patients who are infected with Hepatitis C.

Reuse for Viro positive patients • Dialyzers of patients with Hepatitis B infection should not be reprocessed. • Reprocessing of dialyzers of patients infected with HIV not associated with increased risks of transmission. • The areas utilized for reprocessing should be equipped with a hood and preferably with an exhaust fan. • Sinks should have a depth of at least 45 cm with a drainage mesh at a depth of around 20 cm to prevent the dialyzer and tubing resting in the effluent. •

Reuse water • • Reuse water be treated by reverse osmosis Bacterial colony count of <200 cfu/m. L Endotoxin concentration <2 endotoxin units/m. L The reverse osmosis system should be disinfected monthly • High-flux treatment- ideally, sterile, pyrogen-free water

Reuse water • The water line pressure- 1. 3 kg/cm 2 (20 psi), • Washing area- 2 outlets or a T connection. • 2 different fittings on the water line- a standard tubing to clean the blood compartment and a Hansen connector for backwashing the dialysate compartment. • Fittings should be of 316 SS, or medical grade PVC only.

Patient monitoring • Patient's treatment and course is the primary validation of dialyzer performance: post dialysis weight, pre dialysis plasma urea, urea kinetic modeling, online clearance • ISN Guideline Recommendation- Rigors, fever and hypotension on dialysis should be investigated keeping in mind infection caused by failure of the reprocessing technique, and hemolysis caused by the chemical disinfectants.

Patient monitoring • ISN Guideline Recommendation- In case of rigors, fever and hypotension or a visible change in the color of the blood in the tubing, the dialysis should be stopped. Blood should not be returned to the patient. • Samples should be sent for culture, LDH, and smear examination.

Patient monitoring • The dialyzer should be rinsed with sterile normal saline and the effluent tested for – 1. Residual disinfectants – 2. Cultured on TSA and R 2 A at 25 o. C and 37 o. C. – 3. Endotoxin by Gel clot LAL assay.

Responsibilities of Personnel concerned with reusing dialyzers • Returning blood Reuse process – Dialysis technician or dialysis nurse. • Fixing protocol and decision to use dialyzer singly or multiple times. – Nephrologist in charge of Unit. • Adherence to reprocessing protocol - Dialysis technician or dialysis nurse. • Documentation of tests of performance – Dialysis technician /dialysis nurse.

Responsibilities of Personnel concerned with reusing dialyzers • Decision to discard dialyzer - Dialysis technician or dialysis nurse as per protocol, Nephrologist in charge in case of protocol deviation. • Verification of identity by label on dialyzer prior to starting dialysis - Dialysis technician or dialysis nurse AND concerned patient.

Responsibilities of Personnel concerned with reusing dialyzers • Monitoring of adequacy and safety of the reuse procedure - Nephrologist in charge of Unit. • Maintaining of records relevant to the reuse procedure – Senior technician or Sister in charge of the Unit. • Informed consent- for Reuse of medical devices

Bibliography • Twardowski 2006. Dialyzer Reuse—Part I: Historical Perspective. Semin in Nephrol. • Handbook of dialysis. 4 th edition. Daugirdas • Handbook of dialysis therapy. 4 th edition. Nissesson • Indian Society of Nephrology Hemodialsis Unit Guidelines

Thank you
- Slides: 50