Green Chemistry as a tool to prevent Pharmaceutical












- Slides: 34

Green Chemistry as a tool to prevent Pharmaceutical Hazards and Pollution Dr. Gannu Praveen Kumar M. Pharm. , Ph. D Professor and Principal Department of Pharmaceutics Sahasra Institute of Pharmaceutical Sciences CDSCO

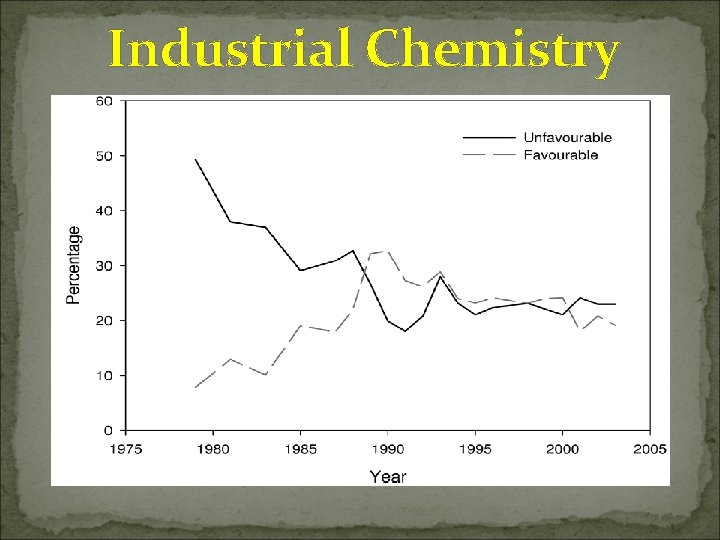
Industrial Chemistry

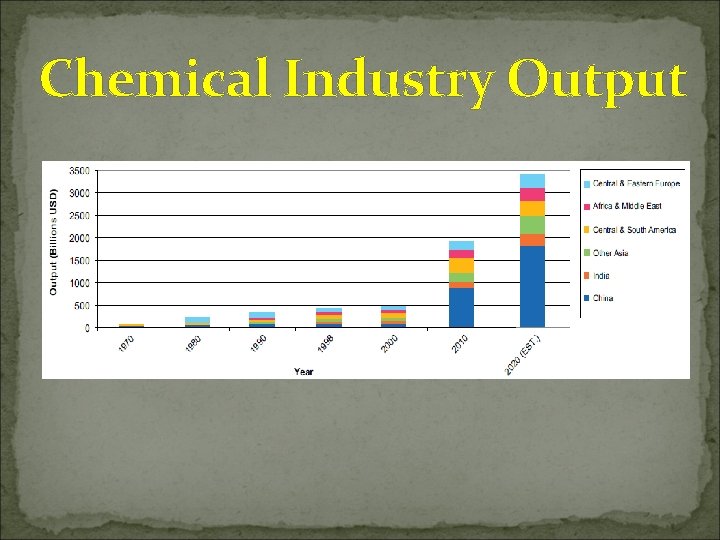
Chemical Industry Output

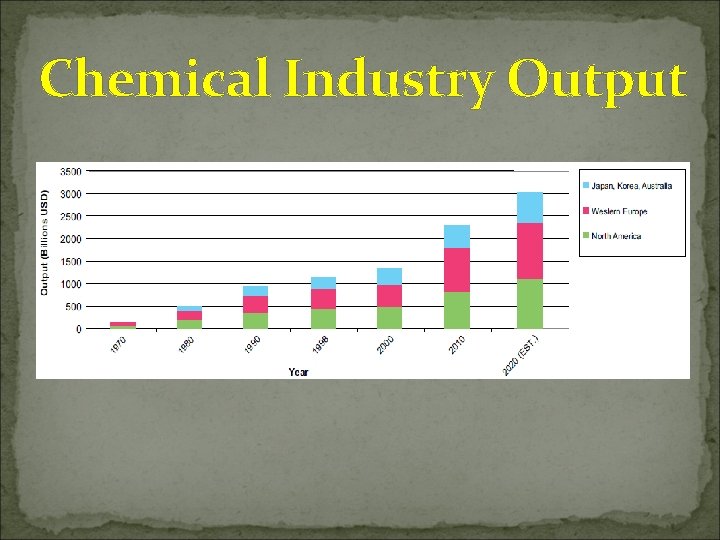
Chemical Industry Output

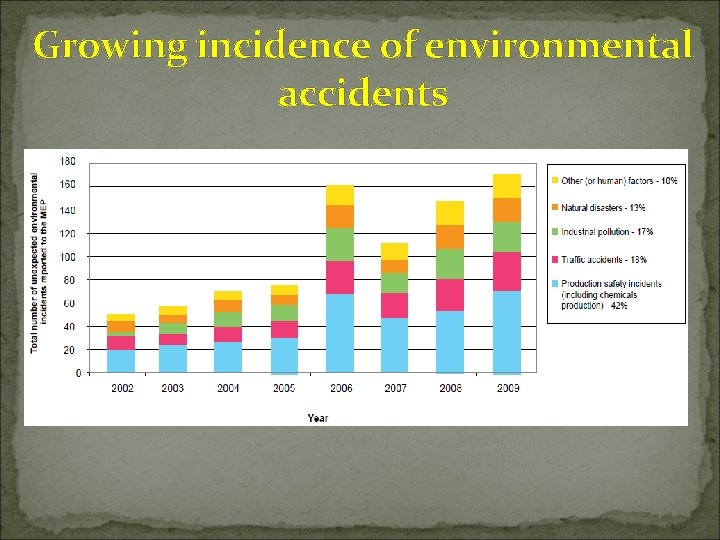
Growing incidence of environmental accidents

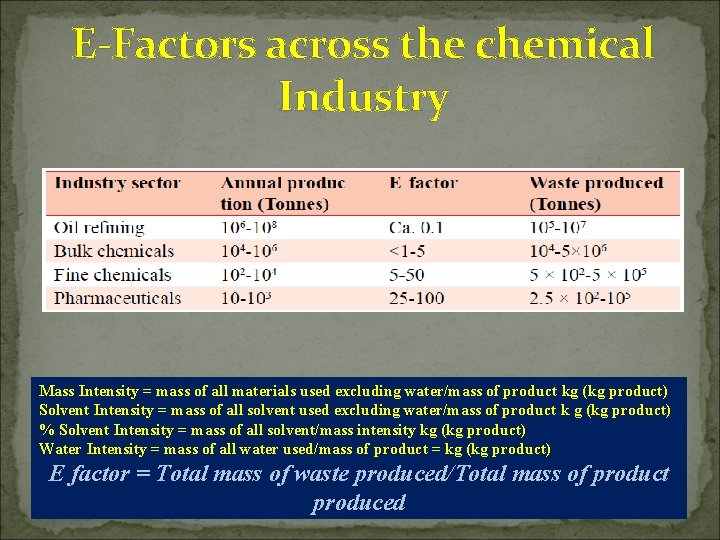
E-Factors across the chemical Industry Mass Intensity = mass of all materials used excluding water/mass of product kg (kg product) Solvent Intensity = mass of all solvent used excluding water/mass of product k g (kg product) % Solvent Intensity = mass of all solvent/mass intensity kg (kg product) Water Intensity = mass of all water used/mass of product = kg (kg product) E factor = Total mass of waste produced/Total mass of product produced

Solvent usage for APIs Synthesis

Green Chemistry Green chemistry is the design of chemical products and processes that reduce or eliminate the use and generation of hazardous substances. Application: to advance the implementation of green chemistry and engineering principles into all aspects of the chemical enterprise Education and Research Education Industrial Implementation

Green Chemistry = Pharmaceutical Hazard & Pollution Free “Green chemistry is the science that introduces new substances into the world and we have a responsibility for their impact in the world. ”

Fundamentals of Green Chemistry Increase awareness and understanding of green chemistry principles, alternatives, practices and benefits. Integrate the principles of Green Chemistry & Green Engineering into the curricula. Equip chemists challenges. to meet Risk = f(Hazard*Exposure) tomorrow’s scientific

Principles of Green Chemistry CATEGORY METHODS Prevention Waste prevention is better than treatment or clean-up Atom Economy Chemical synthesis should maximize the incorporation of all starting materials Chemical synthesis ideally should use and generate nonhazardous substances Chemical products should be designed to be nontoxic Energy demands in chemical syntheses should be minimized Less Hazardous Syntheses Design Safer Chemicals Design for Energy Efficiency Safer Solvents and Auxillaries Inherently Safer Chemistry The use of auxiliaries should be minimized Renewable Feedstocks Reduce Derivatives Raw materials increasingly should be renewable Derivations should be minimized Catalysis Catalysts are superior to reagents Design for Degradation Chemical products should break down into innocuous products Chemical processes require better control Real-time Analysis Substances should have minimum potential for accidents EXAMPLES Use of solvent less sample preparation techniques Hydrogenation of carboxlic acid to aldehydes using solid catalysts Adipic acid synthesis by oxidation of cyclohexene using hydrogen peroxide New less hazardous pesticides Polyolefins-polimer alternative to PWC (polymerization may be carried with lower energy consumption) Supercritical fluid extraction, synthesis in ionic liquids Di-Me carbonate (DMC) is an environmentally friendly substitute for di-Me sulfate and Me halides in methylation reactions Production of surfactants On-fiber derivatization vs derivatization in solution in sample preparation Efficient Au(III)-cata; yzed synthesis of benaminones from 1, 3 -dicarbonyl compds. And amines Synthesis of biodegradable polymers Use of in-line analyzers for wastewater monitoring

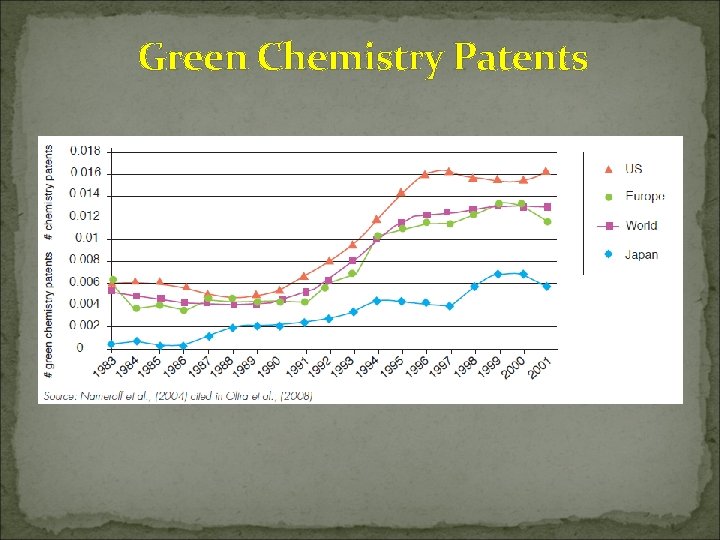
Green Chemistry Patents

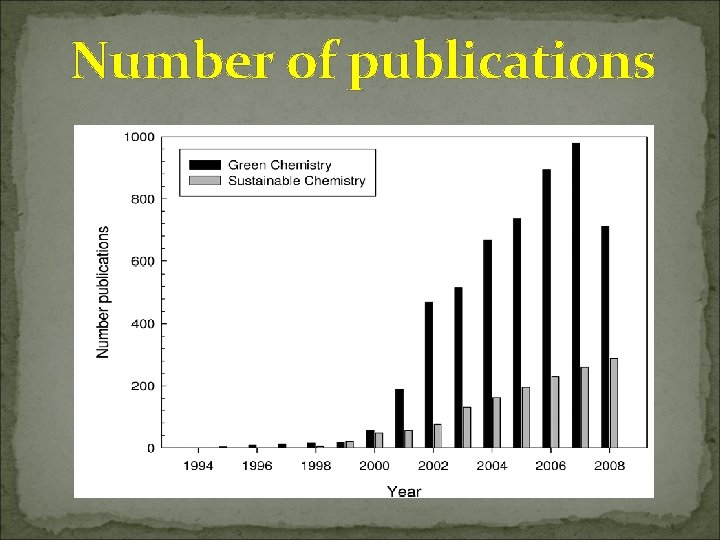
Number of publications

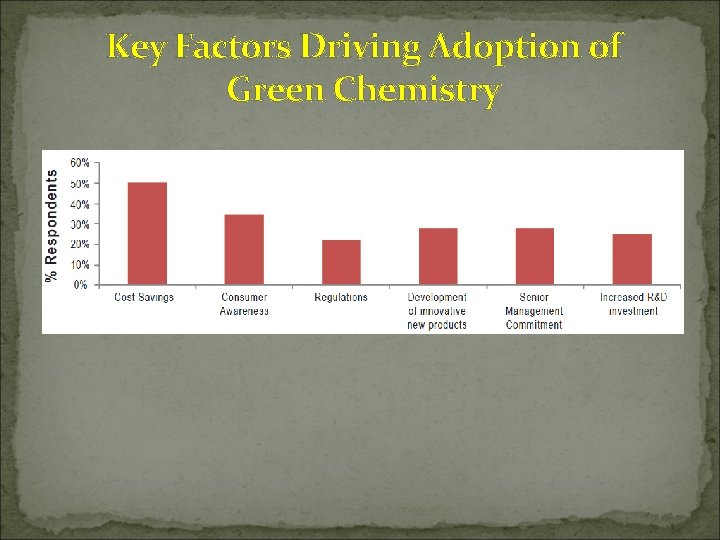
Key Factors Driving Adoption of Green Chemistry

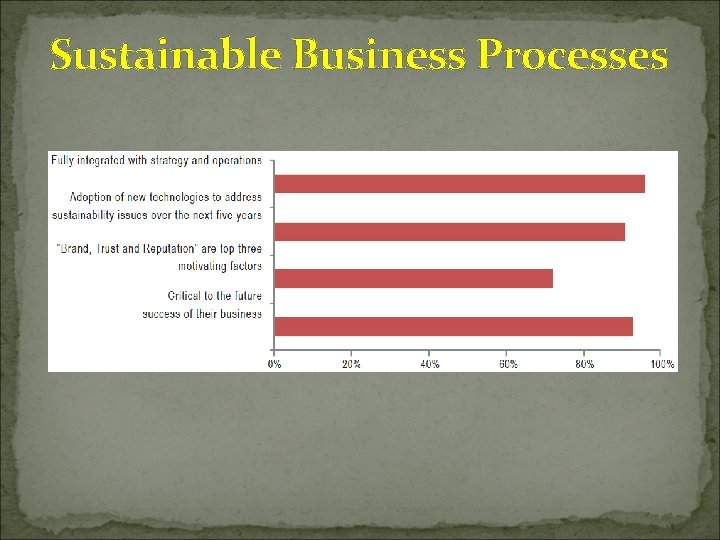
Sustainable Business Processes

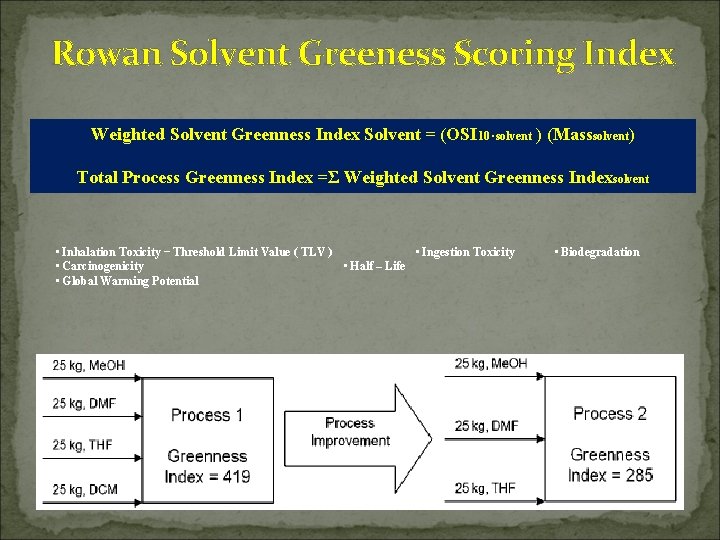
Rowan Solvent Greeness Scoring Index Weighted Solvent Greenness Index Solvent = (OSI 10⋅solvent ) (Masssolvent) Total Process Greenness Index =Σ Weighted Solvent Greenness Indexsolvent • Inhalation Toxicity − Threshold Limit Value ( TLV ) • Ingestion Toxicity • Carcinogenicity • Half – Life • Global Warming Potential • Biodegradation

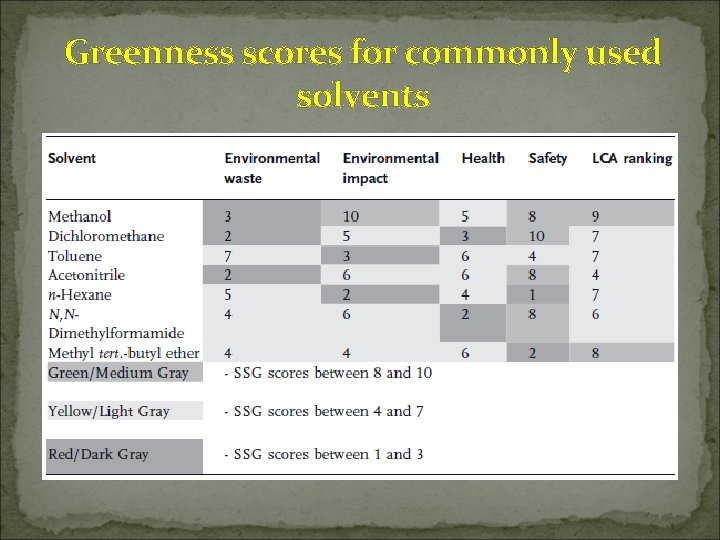
Greenness scores for commonly used solvents

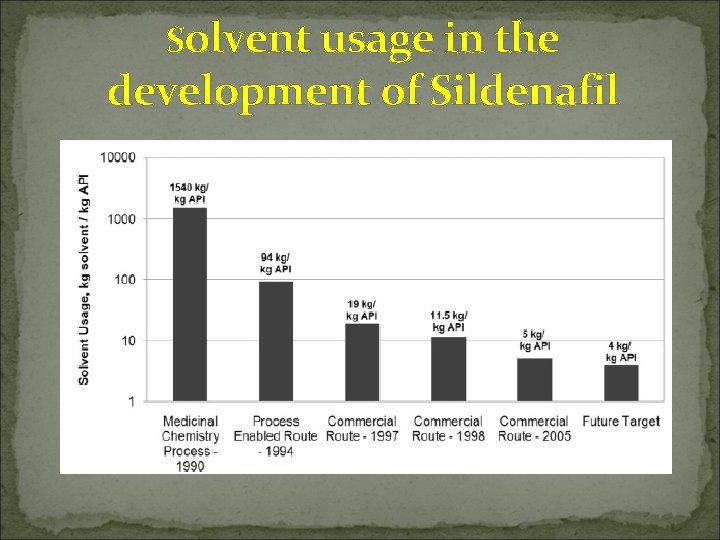
Solvent usage in the development of Sildenafil

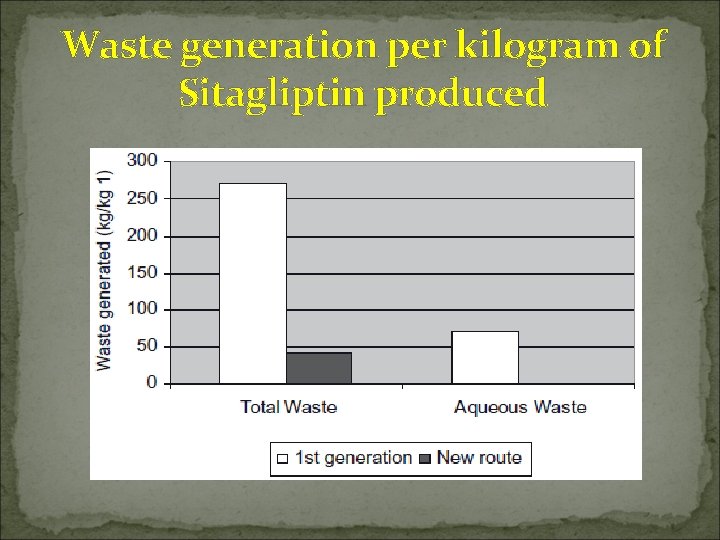
Waste generation per kilogram of Sitagliptin produced

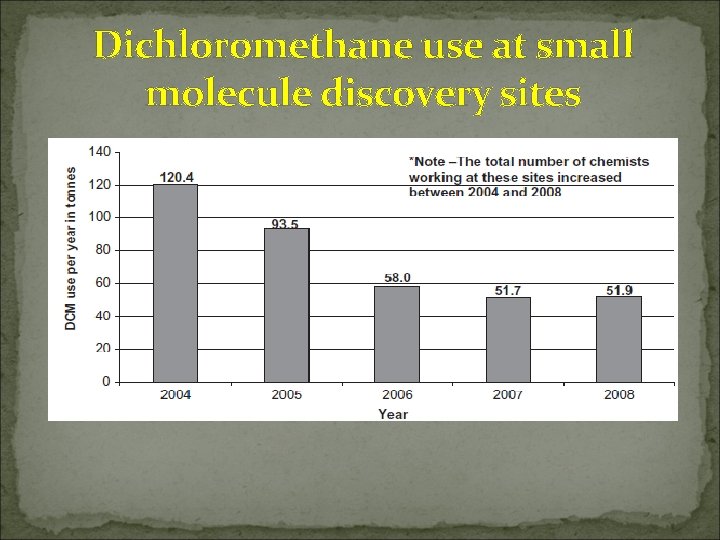
Dichloromethane use at small molecule discovery sites

Importance of Green Chemistry in Nanotechnology In recent years, the development of efficient green chemistry methods for synthesis of nanoparticles has become a major focus of researchers. An eco-friendly technique for production of well-characterized nanoparticles. Production of metal nanoparticles using organisms ( living or dead) Plants seem to be the best candidates and they are suitable for largescale biosynthesis of nanoparticles. Nanoparticles produced by plants are more stable and the rate of synthesis is faster than in the case of microorganisms.

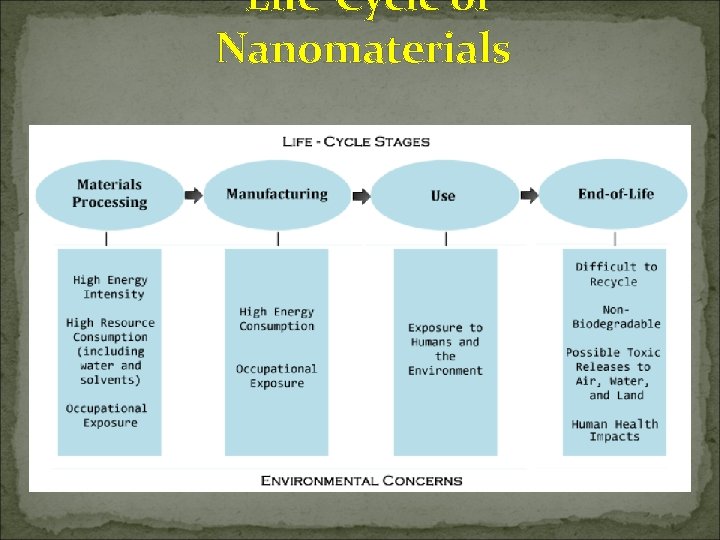
Life-Cycle of Nanomaterials

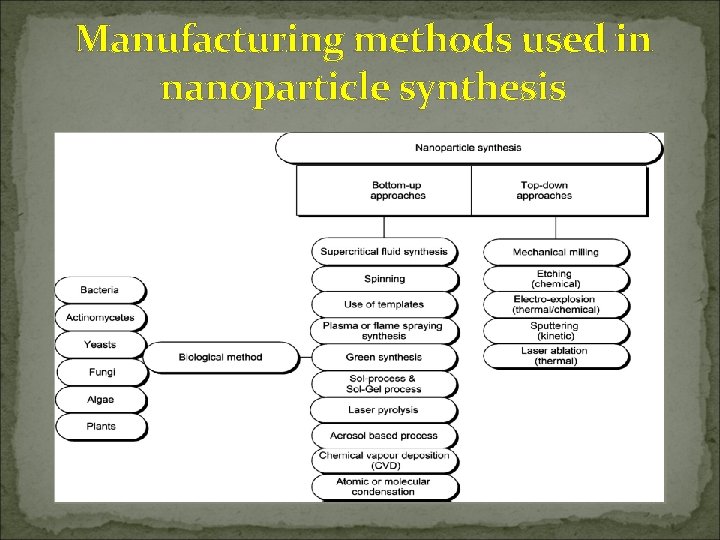
Manufacturing methods used in nanoparticle synthesis

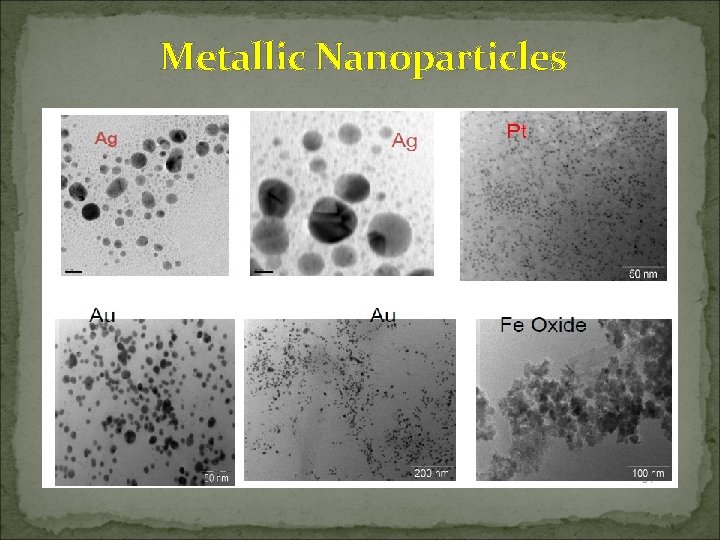
Metallic Nanoparticles Gold Sliver Palladium Some Important Synthetic Methods Chemical reduction of salts; Biological synthesis Microemulsion; Biological synthesis Chemical reduction; Biological synthesis Zinc Oxide Microemulsion; Biological synthesis Magnetitie Copper Indium Oxide Microemulsion; Biological synthesis Microemulsion; Biological Synthesis

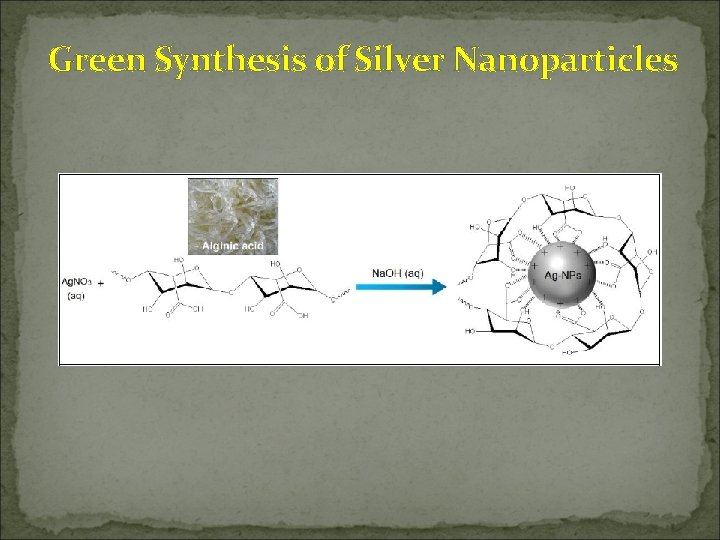
Green Synthesis of Silver Nanoparticles

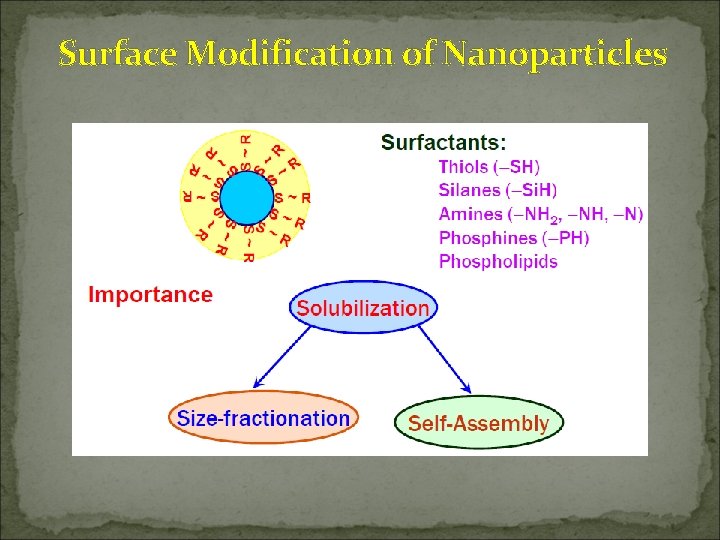
Surface Modification of Nanoparticles

Metallic Nanoparticles

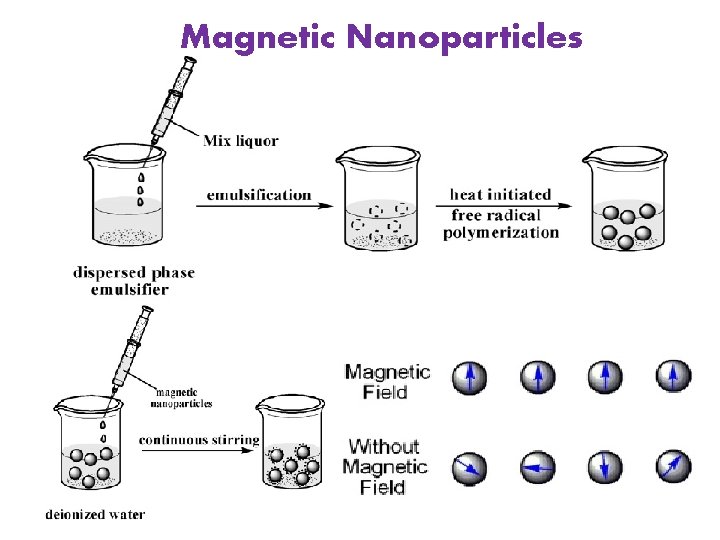
Magnetic Nanoparticles

Separation of magnetic colloidal carriers

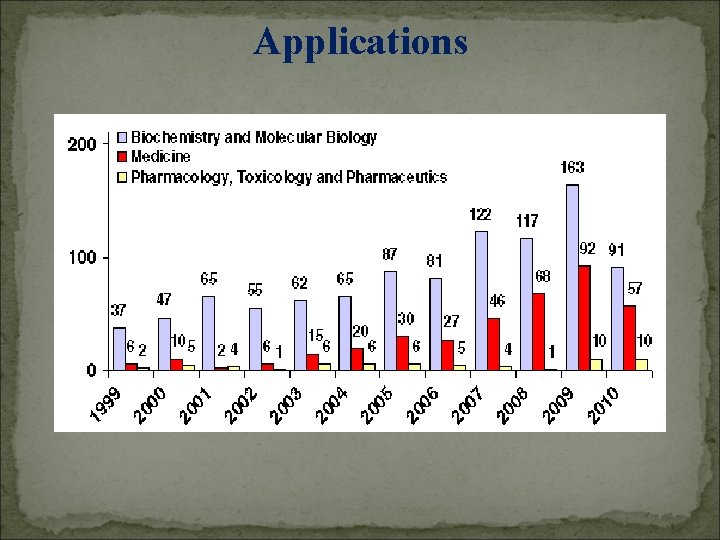
Applications

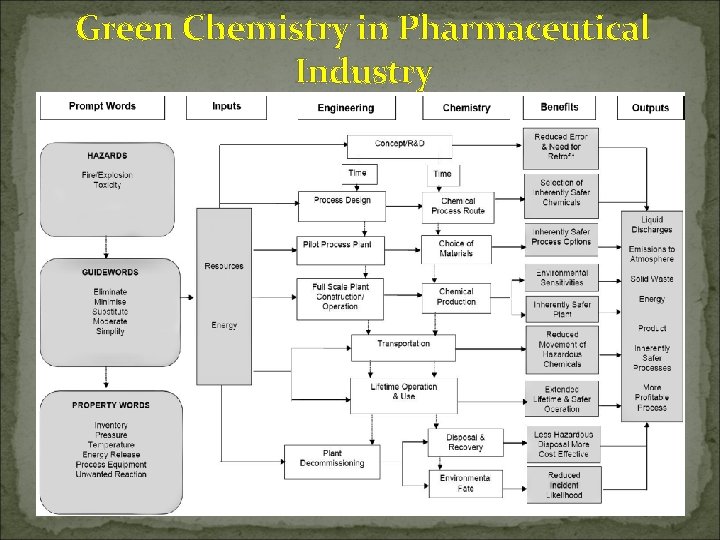
Green Chemistry in Pharmaceutical Industry

Green Pharmaceutical Industry Design

Conclusion The Unique Green Chemistry Applications: • Non-toxic manufacture of metallic nanoparticles • Solvent Consumption Reduction • Safer Environment • Cost Reduction

Thank You