Great Ice Cube Race With a partner discuss
Great Ice Cube Race • With a partner discuss how you can melt an ice cube in a zip lock snack bag at your desk/table the fastest?

Activator • Explain how you differentiate between solids, liquids, and gases?
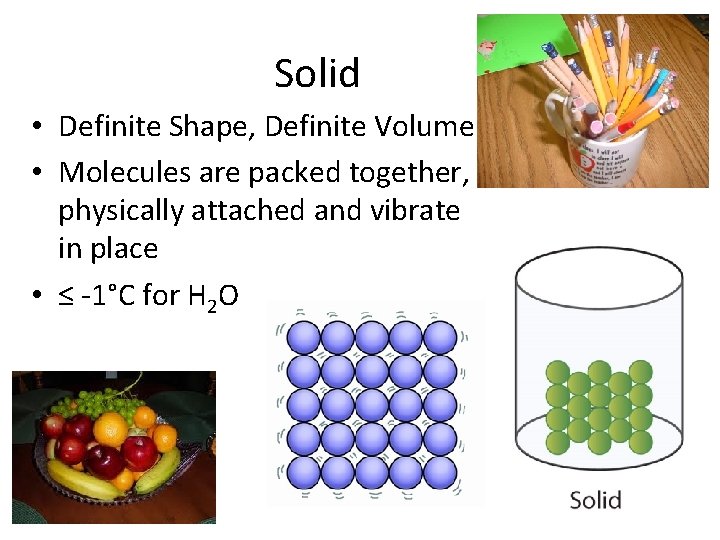
Solid • Definite Shape, Definite Volume • Molecules are packed together, physically attached and vibrate in place • ≤ -1°C for H 2 O

Solid to Liquid • Molecules are moving as fast as they can as a solid • Physical bonds break one by one • Molecules break loose and fall to the bottom as liquid molecules • 0°C for H 2 O
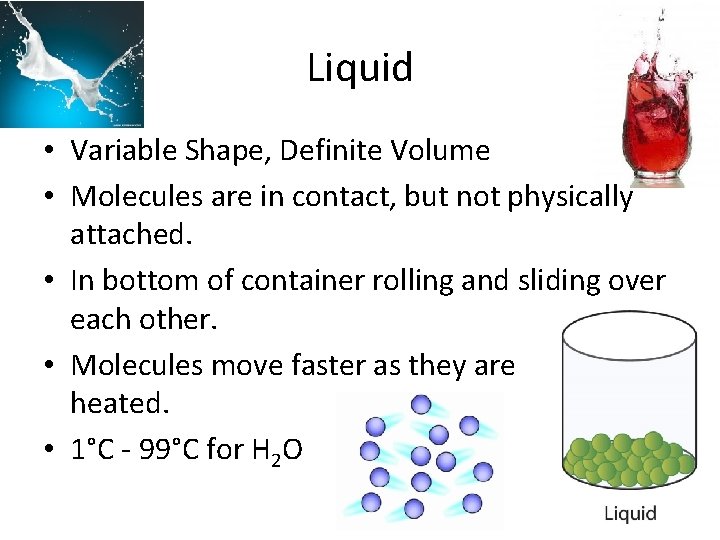
Liquid • Variable Shape, Definite Volume • Molecules are in contact, but not physically attached. • In bottom of container rolling and sliding over each other. • Molecules move faster as they are heated. • 1°C - 99°C for H 2 O

Liquid to Gas • Liquid molecules are moving as fast as they can • Molecules separate from each other and bounce around wildly • 100°C for H 2 O

Gas • Variable Shape, Variable Volume • Molecules are flying around the container and bouncing off of each other • ≥ 101°C for H 2 O

Plasmas • 99% of the matter in space is a plasma • Ex. Stars, Lightning • Superheated gas molecules that are moving so fast that their electrons have stripped off.

Summary • What is the difference between solids, liquids and gases in terms of molecular motion? • Write a thorough, thoughtful paragraph with complete sentences.
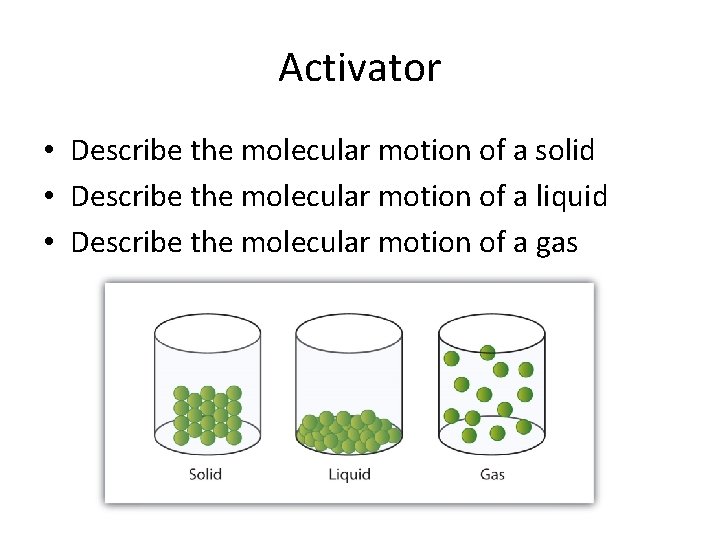
Activator • Describe the molecular motion of a solid • Describe the molecular motion of a liquid • Describe the molecular motion of a gas

Writing Assignment • Write a story in first person where you are a water molecule at -20°C, 50°C, 100°C, 150°C, and 25, 000°C. Write an introduction and a paragraph for each temperature. Be sure to describe your molecular motion at each temperature and end with a summary paragraph.
- Slides: 11