Gravitational Force You already know about gravitational force

Gravitational Force

You already know about gravitational force • “The Earth’s gravitational pull on you” • Your weight! • w = mg

Newton’s law of Universal Gravitation • Relates mass, distance and gravitational force between any two bodies • G is a scaling constant: • m , m = masses • r = distance between the two centers of mass 1 2

Practice Problems • If an apple weighs 1 N at the surface of the earth, how much does it weigh a distance of 2 earth radii from the center of the earth? 3 earth radii? • Find F on a 3 kg rock at the Earth’s surface. How does the force exerted by the Earth on the rock compare to the force exerted by the rock on the Earth? g – M =6 x 10^24 kg – R =6 x 10^6 m e e

• G is the gravitational constant everywhere in the universe – It scales F appropriately (gives it the correct units) – It does not vary from place to place, object to object in the universe g • g is freefall acceleration on Earth, due to Earth’s gravitational pull on other objects

Electrostatics Charge, conservation of charge, acquisition of charge

Where does charge come from? • Atoms are made of positive, negative, and neutral sub particles • Neutrons (neutral charge) and protons (positive charge) are located in a tight cluster at the center of the atom – the nucleus • Electrons (negative charge) move in clouds (spherical shells) around the nucleus These charged particles exert forces on each other, and on the charged particles in surrounding atoms
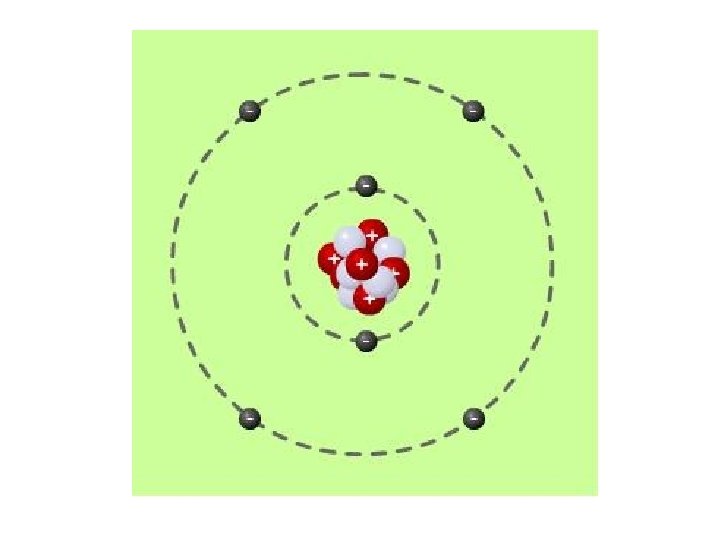

Charge • Protons have greater mass than electrons, but both have the same magnitude of charge (1. 6 x 10^-19 C) – To create an object with positive charge, you must remove electrons – To create an object with negative charge, you must add extra electrons • Most atoms and objects are electrically neutral – the # of electrons = # of protons • Like charges repel, opposite charges attract

Some atoms/molecules hold on to their electrons more tightly than others • Eek! Chemistry. • Example: Atoms in hair and fur give up electrons more easily than atoms in plastic, so if you rub a piece of plastic on fur or hair, electrons are pulled off of the fur and stick to the plastic – The plastic has more electrons than protons and is now negatively charged – The fur has more protons than electrons and is now positively charged

Conservation of charge • When we charge something, no electrons are created or destroyed • electrons are merely transferred from one object or substance to another • Any object that has electrical charge has an excess or deficiency of a whole number of electrons • electrons are an elementary particle and can not be divided into smaller particles (charge is quantized)

Electric Force • Coulomb’s Law: • Relationship discovered in 1700 s by Charles Coulomb – F = force between two charged particles – q 1, q 2 = quantity of charge of each particle – r = distance between two particles (or distance between the CM of two charged objects) – k = 9 x 10^9 N*m²/C²

What is q? • Quantity of charge is measured by amount of charge provided by number of extra electrons or number of missing electrons (extra protons) • q = +/- Ne

Comparison: F vs. F elec grav • Force of gravity: • Electric force: • Similarities – mathematical form, decrease as d increases, both forces act on objects that are not in physical contact with each other • Differences – size (G vs. k), attraction vs. repulsion

Charge and conductivity • Conductor – good conductors share outer valance electrons: outer layer electrons move in a single cloud around all of the atoms/molecules – When conductors carry net charge, charge spreads out (as far from each other as possible) • Insulator – good insulators have tightly bound electrons: electrons don’t move around from atom to atom – When insulators carry net charge, extra charge stays put

2 Ways to acquire an electric charge 1. Charging by contact • transfer of charge from one object to another by physical contact • Electrons “hop” from the substance (like fur) that gives up the electron to the substance (like plastic) that takes an extra electron • Now the fur has less electrons than protons and is positively charged • The plastic has more electrons than protons and is negatively charged

2. Charging by induction • An object is charged by the attraction or repulsion of a nearby charged object, without physical contact. • Conductor: Bring a charged object near a neutral conductor Electrons in the conductor move to one side under the influence of the electric force provided by the charged object. Ground the conductor or separate/break it to create a permanent charge

Electroscopes • Concepts: conductor induction, like charges repel - - --- + +++ + - Electroscope conductor - - Charged object

Charging by induction/polarization • Insulator: charge polarization – Atoms and molecules within the insulator are distorted so that centers of positive and negative charge in a neutral atom or molecule no longer align. – Sticking a balloon to a wall
- Slides: 19