Gram positive rods Gram Positive Bacilli Gram positive

Gram positive rods

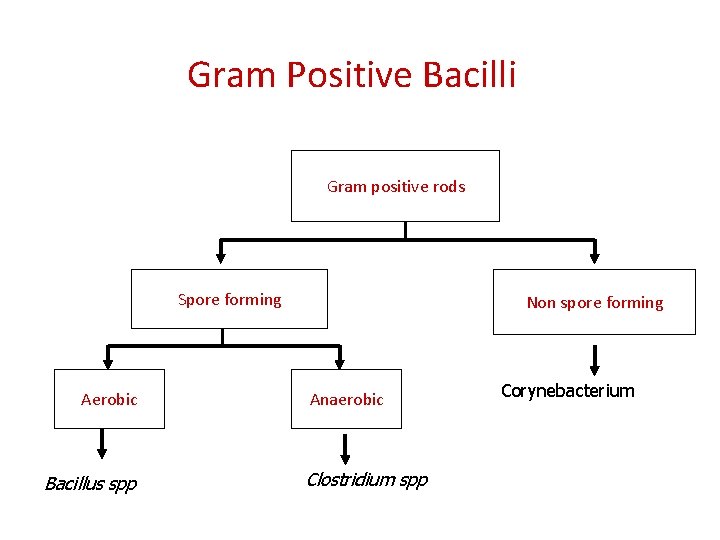
Gram Positive Bacilli Gram positive rods Spore forming Aerobic Bacillus spp Non spore forming Anaerobic Clostridium spp Corynebacterium

Clinically important Gram positive bacilli Spore forming 1. Bacillus 2. Clostridium Non spore forming 1. Corynebacterium 2. Listeria 3. Lactobacillus Bacilli w/ branching filaments 1. Actinomyces 2. Nocardia

Gram positive rods • Medically, Bacillus, Corynebacterium and Listeria are highly pathogenic bacteria which causes of a variety of currently uncommon human diseases. • Their existence poses a continued threat to human survival – deaths caused by diptheria (old killer diseases) or potential bioterrorism of anthrax.

Bacillus With the exception of one species, the genus bacillus are low virulence saprophytes that are widespread in the environment. Saprophytic *B. cereus B. subtilis B. megaterium B. Circulans Pathogenic B. anthracis

General Characters of Bacillus spp • • • Very large Gram positive bacilli 1 -1. 2 µm in width x 3 -5µm in length Arranged in long chains Motile except B. anthracis Spore forming (outside the host) Capsulated (inside the host) Non Fastidious Facultative anaerobic Breakdown glucose by oxidative and fermentative i. e. O+/F+ Catalase positive (diff from clostridium) It is found in soil habitats

1. BACILLUS • Bacillus anthracis – Human pathogen – Isolation also considered to be clinically significant – Zoonosis • Bacillus cereus – Environmental organism – Contaminates food – Common cause of food poisoning • Bacillus (geobacilus) stearothermophilus – Tolerates very high temperatures – Used for quality control of autoclaves

B. anthracis It is aerobic and G+ve, square-ended (rectangular)bacilli with oval central spores non motile, capsulated, arranged singly or in short chains, the organism grows well on blood agar. Hemolysis is uncommon (common with saprophytic bacilli). Colonies characterized by rough uneven surface with multiple curled extensions. The colony is described as "Medusa- head“. The colony is composed of continuous chain of bacilli. • By G. stain, some spores will be seen in between the vegetative cells and these spores appear as empty spaces without stain. • The spores usually resist boiling, some chemicals & dry heat of 140°C but they are destroyed by autoclaving (moist heat) for 15 min. • •

B. anthracis Colony characteristics • • the organism grows well on blood agar. Large (4 -5 mm) Opaque/white/gray Forms capsule at 37º C, 5 -20% CO 2

B. anthracis Biochemical reaction • Catalase +ve • Reduce nitrates to nitrites.

Survival factors for B. anthracis • Environmental Survival – Spores are hardy • Resistant to drying, boiling <10 minutes • Survive for years in soil • Still viable for decades in perma-frost(soil at or below freezing point of water) – Favorable soil factors for spore viability • • High moisture Organic content Alkaline p. H High calcium concentration

Virulence • Virulence Factors – All necessary for full virulence • Polysaccharide somatic antigen – Composed of N-acetylglucoseamine and D-galactose • Capsule (plasmid p. XO 2) – Capsular polypeptide consisting of D-glutamic acid has antiphagocytic properties => B. anthracis that does not produce capsule is not virulent • Toxin(plasmid p. XO 1) – Protective Antigen – Edema Factor – Lethal Factor

Components of anthrax toxin • Protective Antigen – Binds Edema Factor to form Edema Toxin – Facilitates entry of Edema Toxin into cells • Edema Factor – Massive edema by increasing intracellular c. AMP – Also inhibits neutrophil function • Lethal Factor – Stimulates macrophage release of TNF-α, IL-1β – Initiates cascade of events leading to sepsis – Cause of death

• Anthrax produces 3 potentially deadly toxins that can take effect even after the bacteria has been annihilated by antibiotics • The first toxin, protective antigen, paves way for the second toxin, lethal factor to enter cells. • As its name implies, lethal factor then kills the cells. As the cells die, they release inflammatory agents that bring on septic shock then death • Third toxin, edema toxin, cause fluid to accumulate in the lungs or other parts of the body. Ef is doubly dangerous and can be lethal itself

Types of anthrax There are three clinical types of anthrax based on the route of infection • 1 -Skin or cutaneous anthrax (Malignant pustule) • 2 - Respiratory or pulmonary anthrax (wool sorters' disease) • 3 - Intestinal anthrax (rare) in primitive or poor society.

Epidemiology • • All ages and genders affected Occurs worldwide Endemic areas - Africa, Asia True incidence not known – World 20, 000 -100, 000 in 1958 – U. S. 235 total reported cases 1955 -1994 • 18 cases inhalational since 1900, last one 1976 • Until 2001, last previous case cutaneous 1992

Epidemiology • Mortality – Inhalational 86 -100% (despite treatment) • Era of crude intensive supportive care – Cutaneous <5% (treated) – 20% (untreated) – GI approaches 100%

Epidemiology • Incubation Period – Time from exposure to symptoms – Very variable for inhalational • 2 -43 days reported • Theoretically may be up to 100 days • Delayed germination of spores

Epidemiology • Human cases – historical risk factors – Agricultural • Exposure to livestock – Occupational • Exposure to wool and hides • Woolsorter’s disease = inhalational anthrax • Rarely laboratory-acquired

Epidemiology • Transmission – No human-to-human – Naturally occurring cases • Skin exposure • Ingestion • Airborne – Bioterrorism • Aerosol (likely) • Small volume powder (possible) • Foodborne (unlikely)

Epidemiology • Transmission – Inhalational • • Handling hides/skins of infected animals Microbiology laboratory Intentional aerosol release Small volume powdered form – In letters, packages, etc – Questionable risk, probably small

Epidemiology • Transmission – Cutaneous • • Handling hides/skins of infected animals Bites from arthropods (very rare) Handling powdered form in letters, etc. Intentional aerosol release – May see some cutaneous if large-scale

Epidemiology • Transmission – Gastrointestinal • Ingestion of meat from infected animal • Ingestion of intentionally contaminated food – Not likely in large scale – Spores not as viable in large volumes of water • Ingestion from powder-contaminated hands • Inhalational of spores on particles >5 m – Land in oropharynx

Pathogenesis • Anthrax is a zoonosis. The portal of entry is the respiratory tract. Through a cut in skin or the mouth, spores enter the human body. They lodge in one place and start to germinate. They will produce toxins & enzymes till hemorrhage and oedema are produced. • Once the organism looses its capsule, it will die easily because it looses its virulence.

Anthrax Pathogenesis and clinical presentations Cutaneous anthrax About 20% mortality Virulence factors Capsule (antiphagocytic) Toxin (oedema & death) Inhalation anthrax Gastrointestinal anthrax High mortality

Pathogenesis • Disease requires entry of spores into body • Exposure does not always cause disease – Inoculation dose – Route of entry – Host immune status – May depend on pathogen strain characteristics

Pathogenesis • Inhalational – Spores on particles 1 -5 m – Inhaled and deposited into alveoli – Estimated LD 50 = 2500 – 55, 000 spores • Dose required for lethal infection in 50% exposed • Contained in imperceptibly small volume

Pathogenesis • Inhalational – Phagocytosed by alveolar macrophages – Migration to mediastinal/hilar lymph nodes – Germination into vegetative bacilli • Triggered by nutrient-rich environment • May be delayed up to 60 days – Factors not completely understood – Dose, host factors likely play a role – Antibiotic exposure may contribute » Delayed germination after antibiotic suppression

Pathogenesis • Inhalational – Vegetative bacillus is the virulent phase • Active toxin production • Hemorrhagic necrotizing mediastinitis – Hallmark of inhalational anthrax – Manifests as widened mediastinum on CXR • Does NOT cause pneumonia • Followed by high-grade bacteremia – Seeding of multiple organs, including meninges

Pathogenesis • Inhalational – Toxin production • Has usually begun by time of early symptoms • Stimulates cascade of inflammatory mediators – Sepsis – Multiorgan failure – DIC(disseminated intravascular coagulation) • Eventual cause of death – Symptoms mark critical mass of bacterial burden – Usually irreversible by this time » Clearance of bacteria unhelpful as toxinmediated » Early research on antitoxin promising

Pathogenesis • Cutaneous – Spores in contact with skin • Entry through visible cuts or microtrauma – Germination in skin – Disease begins following germination • Toxin production – Local edema, erythema, necrosis, lymphocytic infiltrate – No abscess or suppurative lesions • Eventual eschar(a dry, dark scab or falling away of dead skin typically caused by infection with anthrax) formation
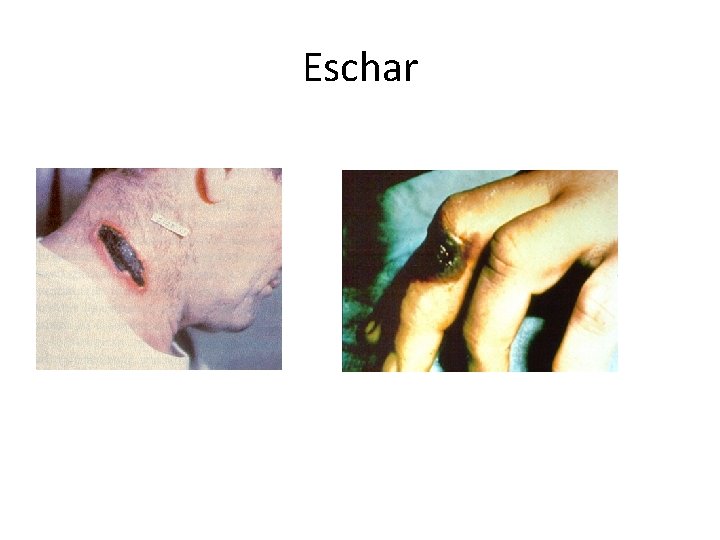
Eschar

Clinical findings • Skin anthrax: a papule first develops within 12 -36 hrs after entry of the organisms or spores through a scratch. This papule rapidly changes into a vesicle, then a pustule, and finally a necrotic ulcer from which the infection may disseminate. Giving rise to septicemia. • Respiratory anthrax: In inhalational anthrax , early manifestations may be mediastanitis, sepsis, meningitis, or hemorrhagic pulmonary edema. Hemorrhagic pneumonia with shock is a terminal event. • Intestinal anthrax: Ingestion of spores cause severe enteritis accompanied by bloody diarrhea with high fatality. Hence, all types of anthrax, if not treated in time, progress to septicemia and death occurs from overwhelming infection.

Clinical Features • Laboratory Findings – Gram positive bacilli in direct blood smear – Electrolyte imbalances common • Radiographic Findings – Widened mediastinum • Minimal or no infiltrates – Can appear during prodrome phase

Diagnosis • Early diagnosis is difficult – Non specific symptoms – Initially mild – No readily available rapid specific tests

Diagnosis • Presumptive diagnosis – History of possible exposure – Typical signs & symptoms – Rapidly progressing nonspecific illness – Widened mediastinum on CXR – Large Gram+ bacilli from specimens • Can be seen on Gram stain if high-grade bacteremia – Appropriate colonial morphology – Necrotizing mediastinitis, meningitis at autopsy

Diagnosis • Definitive diagnosis – Direct culture on standard blood agar • • Gold standard, widely available Alert lab to work up Gram + bacilli if found 6 -24 hours to grow Sensitivity depends on severity, prior antibiotic – Blood, fluid from skin lesions, pleural fluid, CSF, ascites – Sputum unlikely to be helpful (not a pneumonia) • Very high specificity if non-motile, non-hemolytic • Requires biochemical tests for >99% confirmation – Available at Reference laboratories

Diagnosis • Definitive diagnosis – Rapid confirmatory tests • Role is to confirm if cultures are negative • In the US, available currently only at CDC – Polymerase Chain Reaction (PCR) » Hi sensitivity and specificity » Detects DNA » Viable bacteria/spores not required – Immunohistochemical stains – Most clinical specimens can be used

Diagnosis • Other diagnostic tests – Anthraxin skin test • Chemical extract of nonpathogenic B. anthracis • Subdermal injection • 82% sensitivity for cases within 3 days symptoms • 99% sensitivity 4 weeks after symptom onset • Not much experience with use in U. S. – not used

Diagnosis • Testing for exposure – Nasal swabs • Can detect spores prior to illness • Currently used only as epidemiologic tool – Decision for PEP based on exposure risk – May be useful for antibiotic sensitivity in exposed • Culture on standard media • Swabs of nares and facial skin – Serologies • May be useful from epidemiologic standpoint • Investigational – only available at CDC

Diagnosis • Environmental samples – Suspicious powders • Must be sent to reference laboratories as part of epidemiologic/criminal investigation • Assessed using cultures, stains, PCR – Air sampling – First responders • Handheld immunoassays – Not validated – Useful for detecting massive contamination

Mcfadyeans Reaction • A special staining reaction, demonstrating a pink capsule around a blue cell after staining with methylene blue which is used as presumptive diagnosis for anthrax in a blood smear

Treatment • Immediately treat presumptive cases – Prior to confirmation – Rapid antibiotics may improve survival • Differentiate between cases and exposed – Cases • Potentially exposed with any signs/symptoms – Exposed • Potentially exposed but asymptomatic • Provide Post-Exposure Prophylaxis

Treatment • Hospitalization • IV antibiotics – Empiric until sensitivities are known • Intensive supportive care – Electrolyte and acid-base imbalances – Mechanical ventilation – Hemodynamic support

Treatment • Antibiotic selection – Naturally occurring strains • Rare penicillin resistance, but inducible β-lactamase • Penicillins, aminoglycosides, tetracyclines, erythromycin, chloramphenicol have been effective • Ciprofloxacin very effective in vitro, animal studies • Other fluoroquinolones probably effective – Engineered strains • Known penicillin, tetracycline resistance • Highly resistant strains = mortality of untreated

Treatment • Alternative antibiotics – If susceptible, or cipro/doxy not possible • Penicillin*, amoxicillin *FDA Approved • Gentamicin, streptomycin • Erythromycin, chloramphenicol • Ineffective antibiotics – Trimethoprim/Sulfamethoxazole – Third generation cephalosporins

Treatment • Susceptibility testing should be done – Narrow antibiotic if possible – Must be cautious • Multiple strains with engineered resistance to different antibiotics may be coinfecting • Watch for clinical response after switching antibiotic

Treatment • Otherapies – Passive immunization • Anthrax immunoglobulin from horse serum • Risk of serum sickness – Antitoxin • Mutated Protective Antigen – Blocks cell entry of toxin – Still immunogenic, could be an alternative vaccine – Animal models promising

Postexposure Prophylaxis • Who should receive PEP? – Anyone exposed to anthrax – Not for contacts of cases, unless also exposed • Empiric antibiotic therapy • Vaccination

Prevention • Vaccine – Anthrax Vaccine Adsorbed (AVA) – Supply • Limited, controlled by CDC • Production problems – Single producer – Bioport, Michigan – Failed FDA standards – None produced since 1998

Prevention • Vaccine – Inactivated, cell-free – Adsorbed onto Al(OH)3 – Protective Antigen • Immunogenic component • Necessary but not sufficient

Prevention • Vaccine – Effective and Safe – Efficacy • >95% protection vs. aerosol in animal models • >90% vs. cutaneous in humans – Older vaccine that was less immunogenic – Protection vs inhalational but too few cases to confirm

Anthrax –prevention • Prevention – Vaccination of animal herds – Proper disposal of carcasses • Active immunisation with live attenuated bacilli

Bacillus cereus

Bacillus cereus • Large, motile, saprophytic bacillus • Heat resistant spores • Pre formed heat and acid stable toxin (Emetic syndrome) • Heat labile enterotoxin (Diarrhoeal disease) • Lab diagnosis – Demonstation of large number of bacilli in food

B. cereus • B. cereus is a normal inhabitant of soil • Also isolated from food such as grains and spices • B. cereus causes two types of food poisoning – Emetic form or short incubation: • • It is caused by heat stable enterotoxin Nausea, vomiting and abdominal cramps Incubation period of 1 -6 hrs It resembles S. aureus food poisoning – Diarrheal form or long incubation: • • • It is caused by heat labile enterotoxin Abdominal cramps and diarrhea Incubation period of 8 -16 hrs Diarrhea may be a small volume or profuse and watery It resembles food poisoning caused by Cl. perfringens In either type, the illness usually lasts < 24 hrs after onset

Differential characteristics of B. anthracis & B. cereus B. anthracis B. cereus Hemolysis No hemolysis -hemolysis Motility Non-Motile

Nonspore forming G +ve bacilli Corynebacterium Listeria Erisipelothrix Actinomycytes

Corynebacterium spp • Gram positive bacilli, with characteristic morphology (club shaped and beaded) • Non motile • Non spore forming • Non capsulated • Facultative anaerobic (A facultative anaerobe is a microorganism that can exist regardless of whethere is molecular oxygen. When there is oxygen, the anaerobe creates ATP using anaerobic respiration. If oxygen is not present, then the organism ferments) • Breakdown glucose by oxidative and fermentative i. e. O+/F+ • C. diphtheriae is fastidious while diphtheriods are nonfastidious • Catalase positive • Oxidase negative

Corynebacterium diphtheriae Toxigenicity • Infection of non toxigenic species with bacteriophage can lead to toxigenicity (offsprings become lysogenic and toxigenic) • Invasiveness is under the control of the bacterial genes unlike toxigenicity which is controlled by the phage gene.
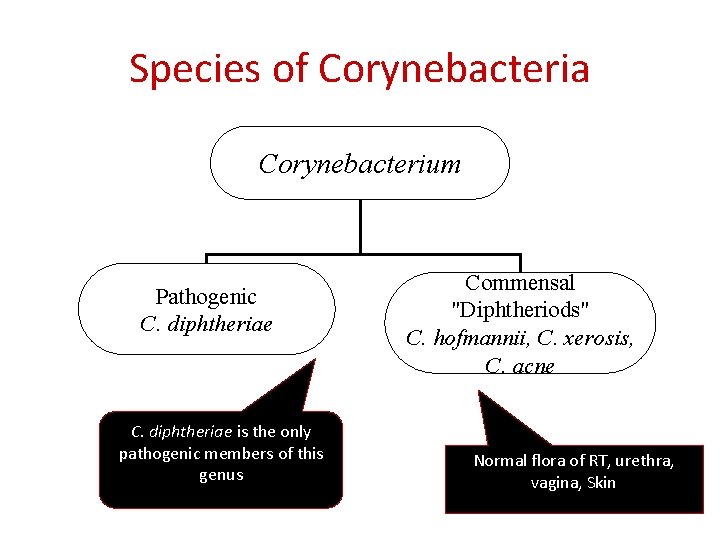
Species of Corynebacteria Corynebacterium Pathogenic C. diphtheriae is the only pathogenic members of this genus Commensal "Diphtheriods" C. hofmannii, C. xerosis, C. acne Normal flora of RT, urethra, vagina, Skin
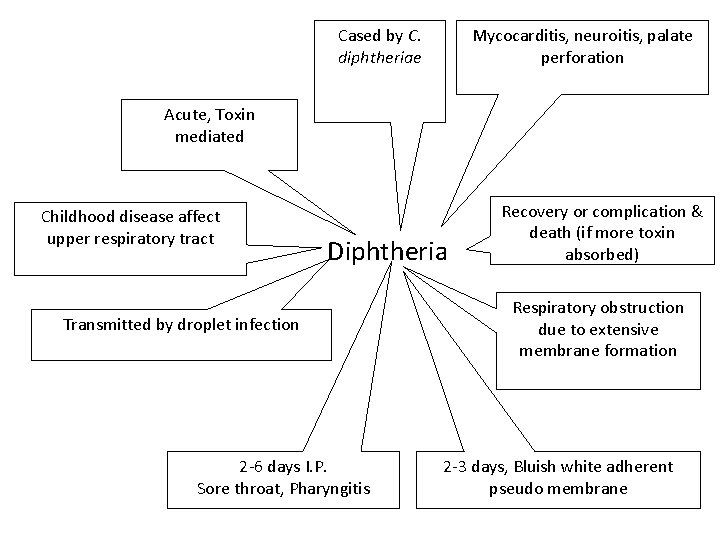
Cased by C. diphtheriae Mycocarditis, neuroitis, palate perforation Acute, Toxin mediated Childhood disease affect upper respiratory tract Diphtheria Transmitted by droplet infection 2 -6 days I. P. Sore throat, Pharyngitis Recovery or complication & death (if more toxin absorbed) Respiratory obstruction due to extensive membrane formation 2 -3 days, Bluish white adherent pseudo membrane

Diagnosis of diphtheria Clinical Diagnosis Specific treatment must be never delayed for laboratory results Laboratory Diagnosis To confirm the clinical manifestation
Diagnosis of diphtheria Diagnosis of case Symptomatic patient Diagnosis of carrier Asymptomatic

Laboratory diagnosis of case – Specimen: A throat swap – Culture: The swap is inoculated on Loeffler's serum medium and/or on blood tellurite agar aerobically at 37 C for 24. • On Loeffler's serum medium: • Corynebacteria grow much more readily than other respiratory pathogens – Used to enhance the characteristic microscopical appearance of corynebacteria – The colonies of C. diphtheriae are small, granular, grey, smooth, and creamy with irregular edges Loefflers serum

Loefflers serum medium – Developed by Friedrich Loeffler in 1887 – Loefflers medium is used to grow corynebacteria – Most recent modification by Buck in 1940 to enhance the growth of C. diphtheria – Consists of Beef serum, heart muscle and animal tissue peptone, sodium chloride, dextrose and egg. – The primary value of Loeffler Medium is in the growth and morphological characterization of members of the genus Corynebacterium; enhances the formation of metachromatic granules within the cells of the organism.

Cultural characteristics • On blood tellurite agar (Mcloed’s blood agar) – It is selective medium for isolation of C. diphtheriae (Potassium tellurite) – 3 biotypes of C. diphtheriae are characterized on BTA – i. e. Gravis, mitis and intermedius biotypes – The most severe disease is associated with the gravis biotype – Colony of gravis biotype is large, non-hemolytic & grey. – Colonies of mitis biotype are small, hemolytic and black – Colonies of intemedius biotype are intermediate in size, nonhemolytic with black center & grey margin.

• Morphology – Gram +ve, nonspore forming nonmotile bacilli – Club-shaped (Coryne= club) arranged at acute angles or parallel to each other (Chinese letters appearance) – Beaded (metachromatic granules) • Stain – Gram stain: • C. diphteriae are gram positive bacilli arranged in Chinese letters form often club shaped – Polychrome methylene blue stain: • C. diphteriae appears beaded due to the presence of intercellular “Metachromatic or volutin" granules • By stain, the granules appear red while the rest of organism appears blue.

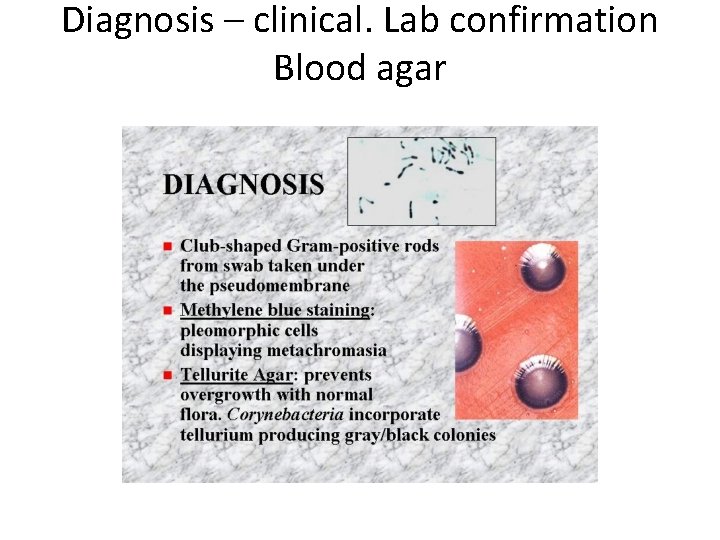
Diagnosis – clinical. Lab confirmation Blood agar

Gram stain of C. diphtheriae on BTA

Biochemical Reaction • All Corynebacterium species are catalase positive (Also, Staphylococcus and Bacillus species are catalase positive)

2 - Carbohydrate Fermentation Test: Principle: § § Each species of corynebacteria has its specific carbohydrate fermentation pattern C. diphtheriae can be differentiated from other Corynebacterium species by fermentation of glucose and maltose but not sucrose with production of acid only Procedure: Ø Inoculate three tubes of carbohydrate fermentation medium (broth containing one type of sugar and phenol red as the p. H indicator) with the test organism Ø Incubate the tubes at 37 o. C for 24 hrs. Glucose Maltose Sucrose

Results: Sugar fermentation can be indicated by change of color of the medium from red to yellow due to formation of acid which decrease the p. H Glucose +ve Maltose +ve C. xerosis Sucrose +ve Glucose +ve Maltose +ve C. diphtheriae Sucrose - ve
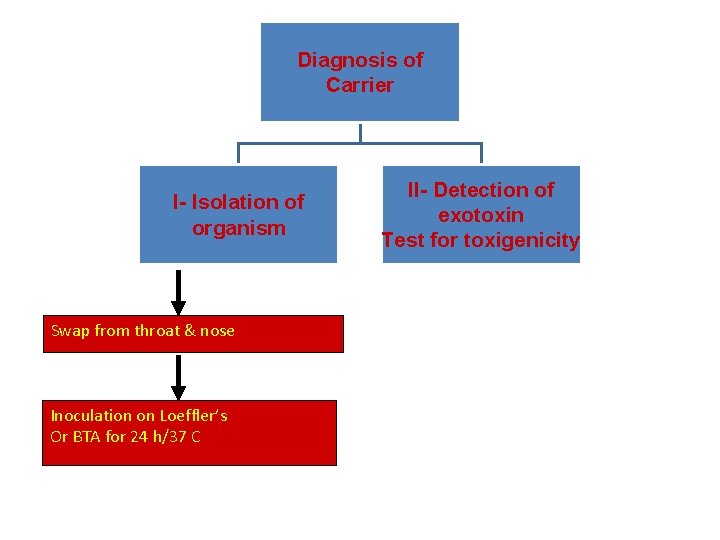
Diagnosis of Carrier I- Isolation of organism Swap from throat & nose Inoculation on Loeffler’s Or BTA for 24 h/37 C II- Detection of exotoxin Test for toxigenicity

In Vitro: Elek’s Test • Principle: – It is toxin/antitoxin reaction – Toxin production by C. diphtheriae can be demonstrated by a precipitation between exotoxin and diphtheria antitoxin • Procedure: • A strip of filter paper impregnated with diphtheria antitoxin is placed on the surface of serum agar • The organism is streaked at right angels to the filter paper • Incubate the plate at 37 C for 24 hrs
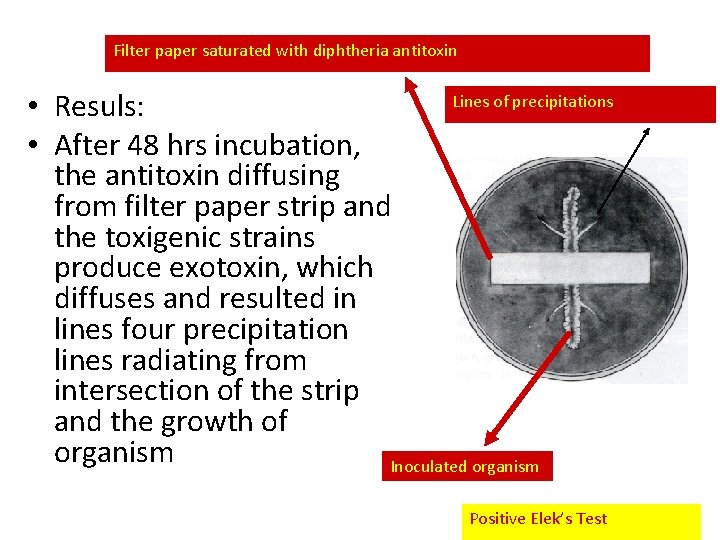
Filter paper saturated with diphtheria antitoxin Lines of precipitations • Resuls: • After 48 hrs incubation, the antitoxin diffusing from filter paper strip and the toxigenic strains produce exotoxin, which diffuses and resulted in lines four precipitation lines radiating from intersection of the strip and the growth of organism Inoculated organism Positive Elek’s Test

LISTERIA MONOCYTOGENES

Statistics • About 2, 500 people in the U. S develop Listeriosis each year. • 5 out of every 100 people carry Listeria Monocytogenes in their intestines. • Listeria Monocytogenes reached the blood and cerebrospinal fluid in 89% of cases. • About 20% of people die from the infection. • Listeriosis results is a higher number of hospitalizations than any other food-born illness. • Pregnant women account for 27% of cases, people with immunodeficiency disorders account for 70% of cases. • AIDS patients are 280 times more likely to contract Listeriosis than others.

Listeria monocytogenes • L. monocytogenes can survive the following – refrigerator temperature (4 degrees). – Low p. H – High salt • Short, G +ve non spore forming rod • catalase positive • Motility at 22 -28 but not at 37 degrees

Listeria monocytogenes • Grows well on 5% sheep blood agar and exhibits the characteristic small zone of hemolysis around and under colonies • Facultative anaerobe • Catalase positive • Produces acid but not gas from utilization of carbohydrates

Listeria monocytogenes • Has several adhesin proteins (Ami, Fbp A and flagellin) that facilitate bacterial binding to the host cells to contribute to virulence • Its cell wall surface protein (Internalin A) interacts with a receptor (E-cadherin) on epithelial cells, promoting phagocytosis into the cell. • After phagocytosis, the bacterium is enclosed in a phagolysosome, where the low p. H activates the bacterium to produce listerolysin O, an enzyme that lyses the membrane of the phagolysome and allows the listeriae to escape into the cytoplasm of the epithelial cell. • Iron is another important virulence factor and Listeriae produce siderophores and are able to obtain iron from transferrin.

What is Listeriosis • Listeriosis is a serious infection caused by eating foods contaminated with the bacterium Listeria Monocytogenes. • This disease affects primarily pregnant women, newborn, and adults with weakened immune systems.

Symptoms Fever Muscle ache GI Sx: Nausea, diarrhea Pregnant women: mild flulike Sx, miscarriage, still birth, premature delivery, or infected newborn. • Lethargy • irritability • • • If infection spreads to the nervous system: headache, stiff neck, confusion, loss of balance, or convulsions. • Listeria can cause Pneumonia, Meningitis, and Sepsis.

Contamination • Listeria Monocytogenes is found in soil and water. • Vegetables can become contaminated from the soil or from manure used as fertilizer. • Animals can carry the bacterium without appearing ill and can contaminate foods of animal origin such as meats and dairy products. • The bacterium has been found in uncooked meats and vegetables, soft cheeses, deli cold-cuts, and unpasteurized milk or foods. • If acquired at birth, the incubation period is 7 to 28 days. • The average incubation period is 31 days; with a range from 11 to 47 days.
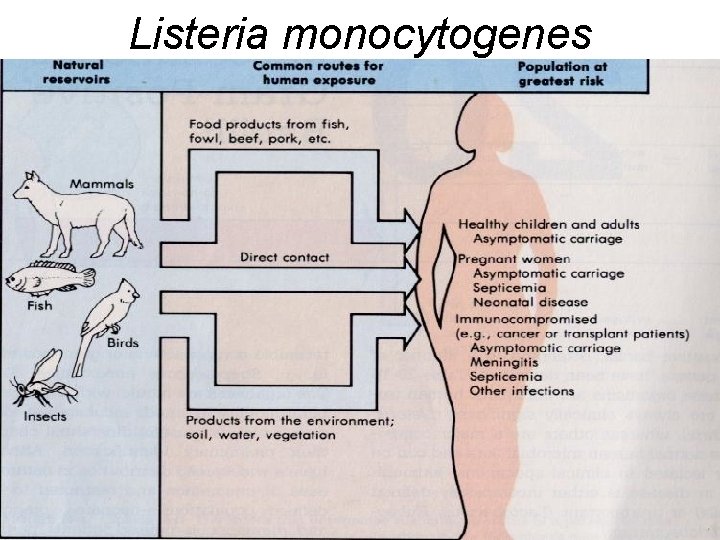
Listeria monocytogenes

Diagnosis • There is no routine screening test for susceptibility. • If symptoms of fever, or stiff neck occurs, consult your doctor. • Blood or cerebrospinal fluid culture will show Listeria. • During pregnancy, a blood test is the most reliable way to find out if symptoms are due to Listeriosis.

Prevention • Thoroughly cook raw food from • Consume perishable and animal sources. ready-to-eat foods as • Wash raw vegetables soon as possible. thoroughly before eating. • Listeria monocytogenes is • Separate uncooked meats from killed by cooking or by vegetables, cooked foods and heating methods, ready-to-eat foods. including pasteurization. • Avoid unpasteurized milk or food. • Wash hands, knives, and cutting boards with hot soapy water for at least 20 sec after handling uncooked foods. • Observe all expiration dates on food items.


Treatment • Listeriosis is a serious disease requiring hospitilization. • A combination of antibiotics is given intravenously • When infection occurs during pregnancy, antibiotics must be given promptly to the mother to prevent infection of the fetus or newborn. • Babies with Listeriosis receive the same antibiotics as adults. • The duration of antibiotic treatment is at least 2 weeks. • Even with prompt treatment, some infections result in death. • Ampicillin, eryhtromycin and trimethoprim-sulfamethoxazole are the susceptible antibiotics; cephalosporins and flourquinolones are not active against L. monocytogenes
5. LACTOBACILLUS (ANEROBES) Normal flora of female genital tract Obligaete anaerobes Low virulence
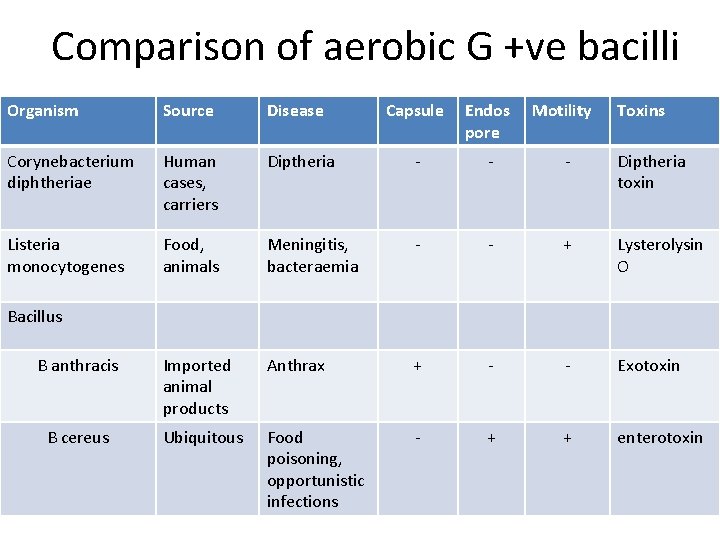
Comparison of aerobic G +ve bacilli Organism Source Disease Capsule Endos pore Corynebacterium diphtheriae Human cases, carriers Listeria monocytogenes Motility Toxins Diptheria - - - Diptheria toxin Food, animals Meningitis, bacteraemia - - + Lysterolysin O Imported animal products Anthrax + - - Exotoxin Ubiquitous Food poisoning, opportunistic infections - + + enterotoxin Bacillus B anthracis B cereus
- Slides: 93