Gram Positive EndosporeForming Bacilli THE GENUS BACILLUS Dr

Gram Positive, Endospore-Forming Bacilli THE GENUS BACILLUS Dr. Wasan Abdul-ilah Bakir
BACILLUS Spp. There are two medically important Bacillus species: Bacillus anthracis and Bacillus cereus

Bacillus anthracis Basic Facts • Zoonotic Disease: usually carried by animals, can be transmitted to human hosts under natural conditions • Rod-shaped • Gram-positive • Aerobic • Exotoxin producing • Endospore-forming, remain viable in soil and animal products for decades. Spores are oval and centrally located.
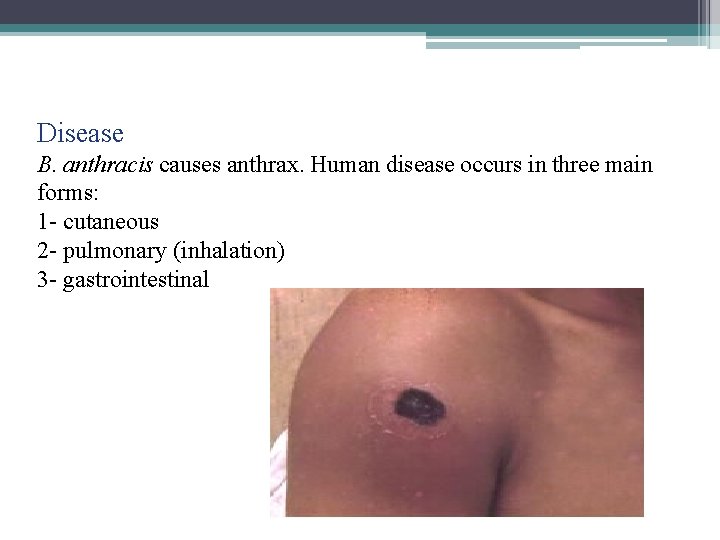
Disease B. anthracis causes anthrax. Human disease occurs in three main forms: 1 - cutaneous 2 - pulmonary (inhalation) 3 - gastrointestinal

• B. anthracis is a large gram-positive rod with square ends, frequently found in chains. • Its antiphagocytic capsule is composed of D-glutamate. (This is unique— capsules of other bacteria are polysaccharides). • It is nonmotile, whereas other members of the genus are motile. • Anthrax toxin is encoded on one plasmid, and the polyglutamate capsule is encoded on a different plasmid.

Transmission Spores of the organism persist in soil for years. Humans are most often infected cutaneously at the time of trauma to the skin, which allows the spores on animal products, such as hides, bristles, and wool, to enter. Spores can also be inhaled into the respiratory tract. Pulmonary (inhalation) anthrax occurs when spores are inhaled into the lungs. Gastrointestinal anthrax occurs when contaminated meat is ingested.

Inhalation anthrax is not communicable from person to person, despite the severity of the infection. The events in the fall of 2001 resulted in 22 cases of anthrax— 11 inhalation and 11 cutaneous. Five of the patients with inhalation anthrax died. All of the other patients survived.

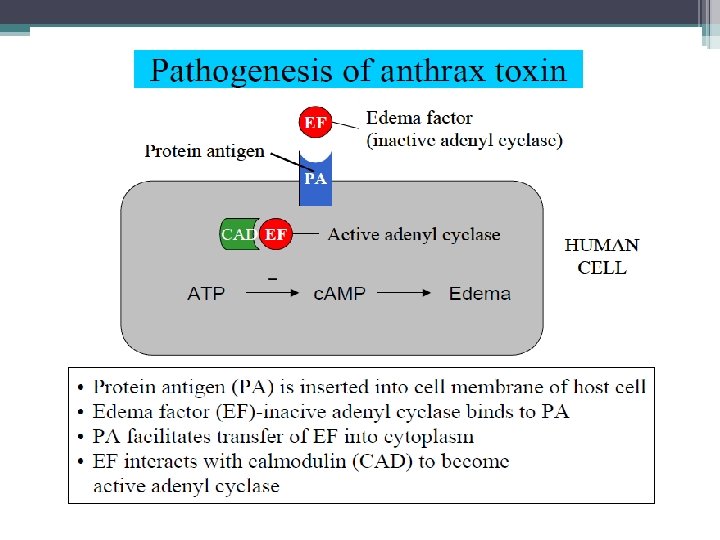
Pathogenesis Anthrax toxins are made up of three proteins, protective antigen (PA), edema factor (EF), and lethal factor (LF). PA is a protein that binds to specific cell receptors, and after proteolytic activation, it forms a membrane channel that mediates entry of EF and LF into the cell. EF is an adenylate cyclase; with PA, it forms a toxin known as edema toxin. Edema toxin is responsible for cell and tissue edema. LF plus PA form lethal toxin, which is a major virulence factor and cause of death in infected animals and humans.

Edema factor, an exotoxin, is an adenylate cyclase that causes an increase in the intracellular concentration of cyclic adenosine monophosphate (AMP). This causes an outpouring of fluid from the cell into the extracellular space, which manifests as edema. (Note the similarity of action to that of cholera toxin. ) Lethal factor is a protease that cleaves the phosphokinase that activates the mitogen-activated protein kinase (MAPK) signal transduction pathway. This pathway controls the growth of human cells, and cleavage of the phosphokinase inhibits cell growth.


Clinical Findings Cutaneous Anthrax In humans, approximately 95% of cases are cutaneous anthrax, and 5% are inhalation. Gastrointestinal anthrax is very rare; it has been reported from Africa, Asia, and the United States when people have eaten meat from infected animals. Cutaneous anthrax generally occurs on exposed surfaces of the arms or hands followed in frequency by the face and neck. A pruritic papule develops 1– 7 days after entry of the organisms or spores through a scratch. Initially, it resembles an insect bite. The papule rapidly changes into a vesicle or small ring of vesicles that coalesce, and a necrotic ulcerdevelops. The lesions typically are 1– 3 cm in diameter and have a characteristic central black eschar. Marked edema occurs.

Cutaneous Anthrax • B. anthracis comes into contact with a skin lesion, or cut. • The spores germinate, producing bacteria, which produce exotoxins to evade the macrophages • Infection is manifested in a painless ulcer with a necrotic (dead) center. Skin lesion of anthrax, the black eschar, a necrotic lesion covered by a crust, caused by lethal factor, an exotoxin produced by Bacillus anthracis. The area of edema surrounding the eschar, which is caused by another exotoxin called edema factor. Skin lesion of anthrax

Symptoms of cutaneous anthrax • • • Boil-like skin lesion forming a painless ulcer Swelling of the lymph glands Fever Headache malaise It may take many weeks for the lesion to heal and the edema to subside. Antibiotic therapy does not appear to change the natural progression of the disease but prevents dissemination. In as many as 20% of patients, cutaneous anthrax can lead to sepsis, the consequences of systemic infection—including meningitis—and death.
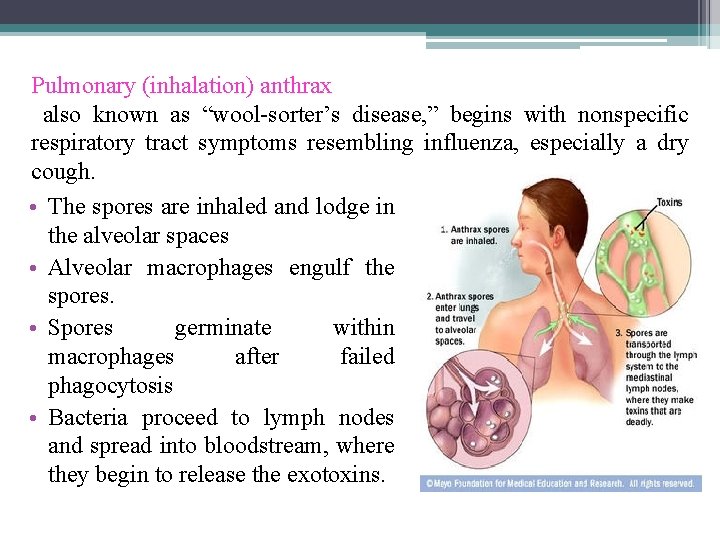
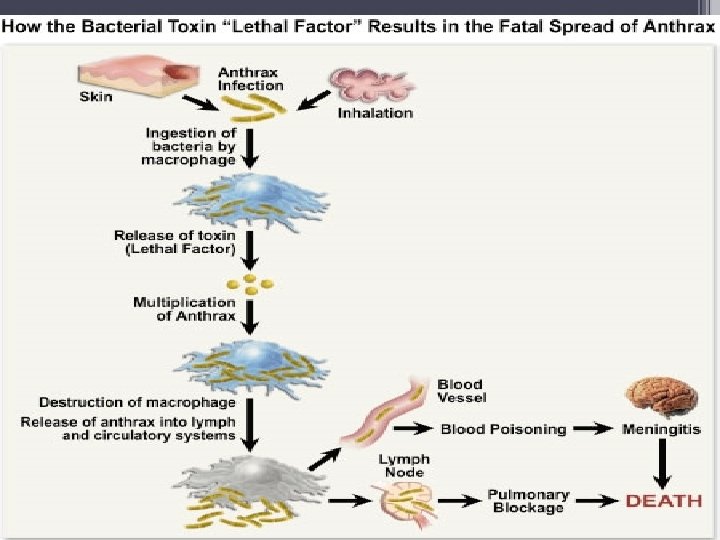
Pulmonary (inhalation) anthrax also known as “wool-sorter’s disease, ” begins with nonspecific respiratory tract symptoms resembling influenza, especially a dry cough. • The spores are inhaled and lodge in the alveolar spaces • Alveolar macrophages engulf the spores. • Spores germinate within macrophages after failed phagocytosis • Bacteria proceed to lymph nodes and spread into bloodstream, where they begin to release the exotoxins.

long as 6 weeks. The early clinical manifestations are associated with marked hemorrhagic necrosis and edema of the mediastinum. Substernal pain may be prominent, and there is pronounced mediastinal widening visible on chest radiographs. Hemorrhagic pleural effusions follow involvement of the pleura; cough is secondary to the effects on the trachea. Sepsis occurs, and there may be hematogenous spread to the gastrointestinal tract, causing bowel ulceration, or to the meninges, causing hemorrhagic meningitis. The fatality rate in inhalation anthrax is high in the setting of known exposure; it is higher when the diagnosis is not initially suspected.

Symptoms of Pulmonary Anthrax • Initial symptoms: sore throat, mild fever, muscle aches • Severe difficulty breathing • Septic shock • Development of meningitis • Respiratory failure resulting in death
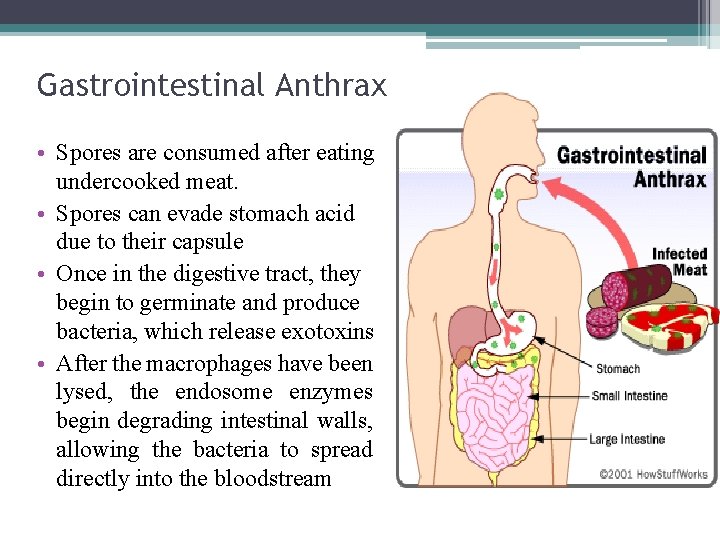
Gastrointestinal Anthrax • Spores are consumed after eating undercooked meat. • Spores can evade stomach acid due to their capsule • Once in the digestive tract, they begin to germinate and produce bacteria, which release exotoxins • After the macrophages have been lysed, the endosome enzymes begin degrading intestinal walls, allowing the bacteria to spread directly into the bloodstream

Symptoms of Gastrointestinal anthrax • • Serious gastrointestinal difficulty Nausea Fever Abdominal pain Vomiting of blood Severe bloody diarrhea Acute inflammation of intestinal tract Loss of appetite

Pathology In susceptible animals and humans, the organisms proliferate at the site of entry. The capsules remain intact, and the organisms are surrounded by a large amount of proteinaceous fluid containing few leukocytes from which they rapidly disseminate and reach the bloodstream.

Laboratory Diagnosis • A diagnosis can be made by taking a smear of a skin lesion (if cutaneous anthrax) and Gram stain shows large gram-positive rods. • Cultures: the organisms produce nonhemolytic gray to white, tenacious colonies with a rough texture and a ground-glass appearance. Comma-shaped outgrowths (Medusa head, “curled hair”) may project from the colony. Demonstration of capsule requires growth on bicarbonate- containing medium in 5– 7% carbon dioxide.

Laboratory Diagnosis Spores are usually not seen in smears of exudate because spores form when nutrients are insufficient, and nutrients are plentiful in infected tissue. rapid diagnosis can be performed in special laboratories using polymerase chain reaction (PCR)–based assays. Another rapid diagnostic procedure is the direct fluorescent antibody test that detects antigens of the organism in the lesion. Serologic tests, such as an enzyme-linked immunosorbent assay (ELISA) test for antibodies.

Treatment The treatment must be started early. Ciprofloxacin is recommended for treatment; other agents with activity include penicillin G, doxycycline, erythromycin, and vancomycin. In the setting of potential exposure to B anthracis as an agent of biologic warfare, prophylaxis with ciprofloxacin or doxycycline should be given for 60 days and three doses of vaccine (AVA Bio. Thrax) should be administered. Raxibacumab, a recombinant human monoclonal antibody, was FDA approved for treatment of and prophylaxis against inhalational anthrax. The mechanism of action is prevention of binding of PA to its receptors in host cells. The drug is used in combination with appropriate antimicrobial agents. Anthrax immune globulin intravenous (AIGIV ) is a human polyclonal antiserum that also inhibits binding of PA to its receptors. Like Raxibacumab, it is used as an adjunct to antimicrobial agents for the treatment of severe forms of anthrax.

Control • Ciprofloxacin or doxycycline was used as prophylaxis in those exposed during the outbreak in the United States in 2001. • People at high risk can be immunized with cell-free vaccine containing purified protective antigen as immunogen. • Incinerating animals that die of anthrax, rather than burying them, will prevent the soil from becoming contaminated with spores.

Bacillus cereus

Bacillus cereus Gram-positive spore forming bacilli • Produce β-hemolysis on blood agar ° Spores are found on most raw foods like rice Transmission Spores on grains such as rice survive steaming and rapid frying. The spores germinate when rice is kept warm for many hours (e. g. , reheated fried rice). The portal of entry is the gastrointestinal tract. Produce enterotoxin – ingested → food poisoning.

Pathogenesis Food poisoning caused by B cereus has two distinct forms: 1. the emetic type, which is associated with fried rice, milk, and pasta. 2. the diarrheal type, which is associated with meat dishes and sauces. The emetic form is manifested by nausea, vomiting, abdominal cramps, and occasionally diarrhea and is self-limiting, with recovery occurring within 24 hours. It begins 1– 5 hours after ingestion of a plasmid-encoded preformed cyclic peptide (emetic toxin) in the contaminated food products. B cereus is a soil organism that commonly contaminates rice. When large amounts of rice are cooked and allowed to cool slowly, the B cereus spores germinate, and the vegetative cells produce the toxin.

The diarrheal form has an incubation period of 1– 24 hours and is manifested by profuse diarrhea with abdominal pain and cramps; fever and vomiting are uncommon. In this syndrome, ingested spores that develop into vegetative cells of B cereus secrete enterotoxin which induce fluid accumulation and other physiological responses in the small intestine. B cereus is an important cause of eye infections, such as severe keratitis. Typically, the organisms are introduced into the eye by foreign bodies associated with trauma but infections can also occur after surgery.

B cereus has also been associated with localized infections, such as wound infections, and with systemic infections, including endocarditis, catheter-associated bacteremia, central nervous system infections, osteomyelitis, and pneumonia; the presence of a medical device or intravenous drug use predisposes to these infections. B cereus cause food poisoning and opportunistic infections in immunocompromised patients. B cereus can be differentiated from B anthracis on the basis of colony morphology, β-hemolysis, motility, and antimicrobial susceptibility patterns.

Treatment B cereus is resistant to a variety of antimicrobial agents, including penicillins and cephalosporins. Serious non–food borne infections should be treated with vancomycin or clindamycin with or without an aminoglycoside. Ciprofloxacin has been useful for the treatment of wound infections
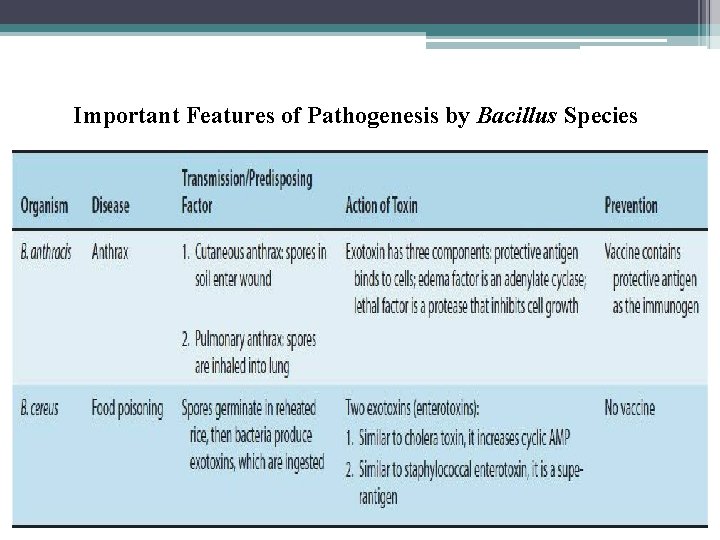
Important Features of Pathogenesis by Bacillus Species

Thank you
- Slides: 33