Grafting of a polyol polyether chain onto TEMPO









- Slides: 16

Grafting of a polyol polyether chain onto TEMPO nanocellulose Jaka Levanič, mag. inž. Les. Ida Poljanšek, doc. dr. Primož Oven, prof. dr. SP Technical Research Institute of Sweden, Borås, Sweden 13 th – 14 th April 2016 Conference and Joint Working Group Meeting: Cellulosic material properties and industrial potential

Introduction & aim § Cellulose is an abundant natural polymer § Widely available from numerous lignocellulosic as well as some microbial and animal sources § Cellulose nanofibers have exceptional mechanical properties § Desire to produce a cast able resin where cellulose nanofibers would act as part of the resin matrix (chemical bonds instead of physical interactions) § Grafting of a polyol polyether chain with an unsaturated C – C bond which would allow crosslinking Moon in sod. , 2011. Chem. Soc. Rev. 40: 391 -3994

Goals § Using the TEMPO reagent to oxidize the hydroxyl group on the surface of cellulose fibers to carboxyl groups § Using a combination of mechanical procedures to prepare TEMPO oxidized cellulose nanofibers § Graft a polyol polyether chain via the „grafting to“ approach onto the surface of TEMPO oxidized cellulose nanofibers Roy in sod. , 2009. Chem. Soc. Rev. 38, 7: 1825 - 2148

Materials and methods § Grinding and pre – swelling of the dried cellulose fibers § TEMPO oxidation of the cellulose fibers § Mechanical disintegration of the oxidized cellulose fibers using a high shear mixer and ultrasound § Grafting of the polyol polyether chain onto the TEMPO oxidized cellulose nanofibers using a modified Steiglich esterification process (Moore and Stupp, 1990) § Characterization of the products (SEM, FTIR, NMR, conductometric titration, TGA and DSC)

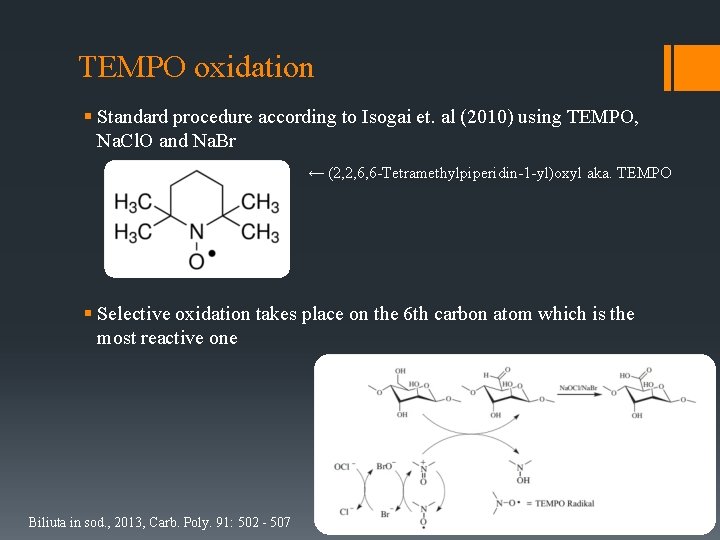
TEMPO oxidation § Standard procedure according to Isogai et. al (2010) using TEMPO, Na. Cl. O and Na. Br ← (2, 2, 6, 6 -Tetramethylpiperidin-1 -yl)oxyl aka. TEMPO § Selective oxidation takes place on the 6 th carbon atom which is the most reactive one Biliuta in sod. , 2013, Carb. Poly. 91: 502 - 507

TEMPO oxidation § p. H was maintained at 10, 0 +/- 0, 1 using 0, 5 M Na. OH § Reaction was carried out until the p. H remain unchanged for a set amount of time § The oxidation process was stopped by lowering the p. H to 7, 0 using 0, 5 M HCl § Fibers were filtered and washed with distilled water

Mechanical treatment of the oxidized fibers § TEMPO oxidized cellulose fiber suspension in water (2%) § Mechanical processing was done with a combination of high shear mixing (Ultra. Turrax) and sonication § Cycles were kept short to prevent overheating and discoloration of the suspension § The goal was to break the TEMPO oxidized fibers into individual nanofibers

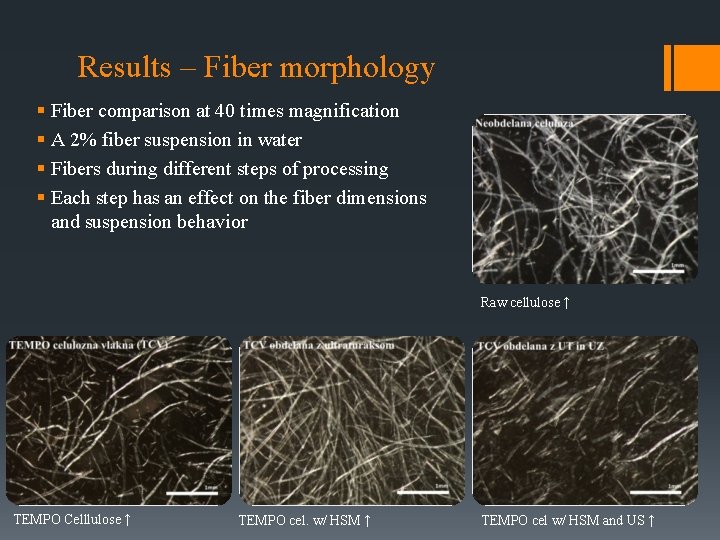
Results – Fiber morphology § Fiber comparison at 40 times magnification § A 2% fiber suspension in water § Fibers during different steps of processing § Each step has an effect on the fiber dimensions and suspension behavior Raw cellulose ↑ TEMPO Celllulose ↑ TEMPO cel. w/ HSM ↑ TEMPO cel w/ HSM and US ↑

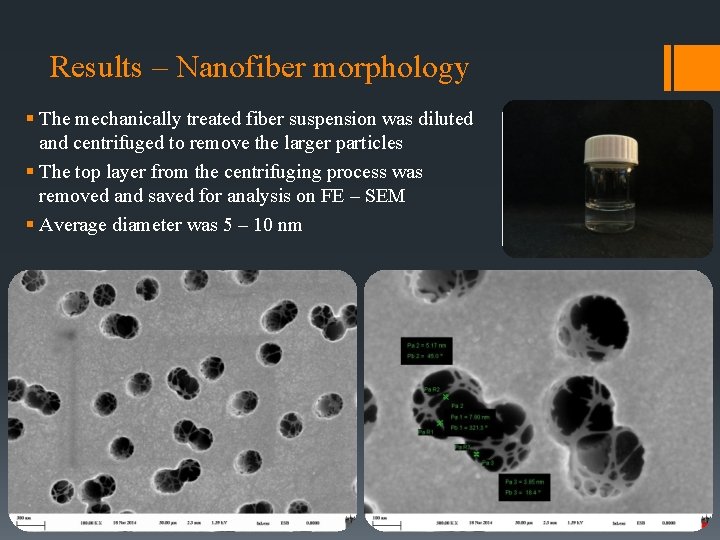
Results – Nanofiber morphology § The mechanically treated fiber suspension was diluted and centrifuged to remove the larger particles § The top layer from the centrifuging process was removed and saved for analysis on FE – SEM § Average diameter was 5 – 10 nm

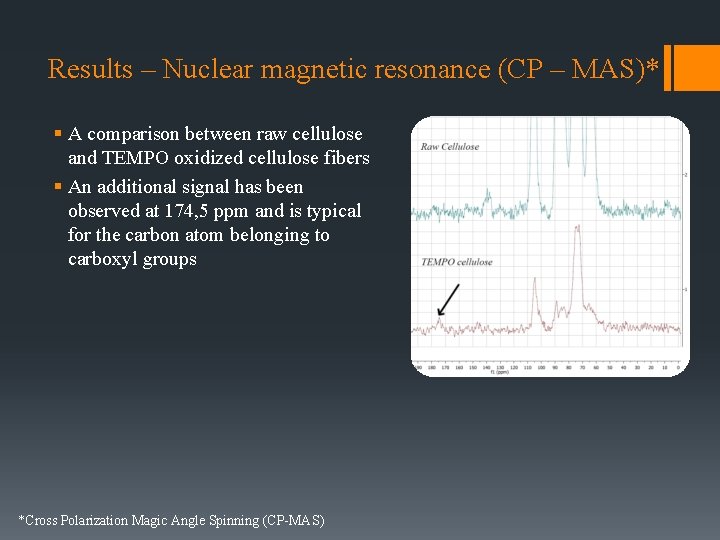
Results – Nuclear magnetic resonance (CP – MAS)* § A comparison between raw cellulose and TEMPO oxidized cellulose fibers § An additional signal has been observed at 174, 5 ppm and is typical for the carbon atom belonging to carboxyl groups *Cross Polarization Magic Angle Spinning (CP-MAS)

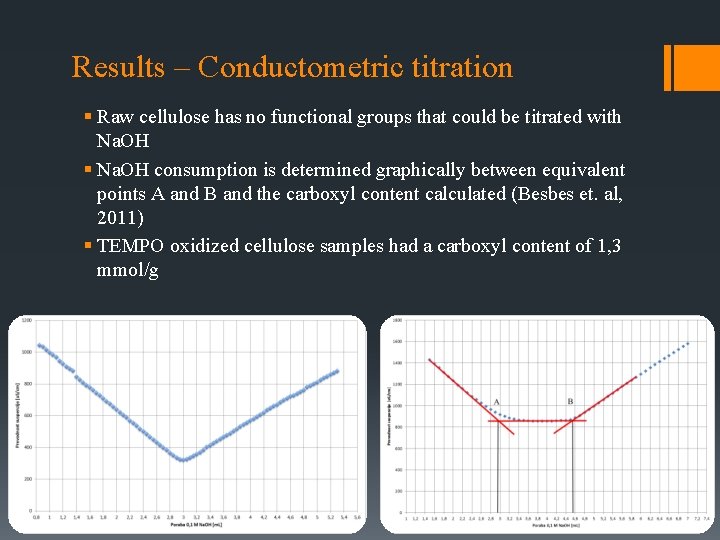
Results – Conductometric titration § Raw cellulose has no functional groups that could be titrated with Na. OH § Na. OH consumption is determined graphically between equivalent points A and B and the carboxyl content calculated (Besbes et. al, 2011) § TEMPO oxidized cellulose samples had a carboxyl content of 1, 3 mmol/g

Results – Grafting § Grafting was performed with a modified Steiglich esterification process in dichloromethane (DCM) § To promote grafting, DPTS* and DCC* were used § The reaction was carried under normal pressure and temperature § Only mild agitation was used § All samples were reacted for 24 hours § Final product was filtered and washed with cold DCM to remove unreacted reagents § Samples were then dried under vacuum and room temperature *DPTS = 4 -(dimethylamino) pyridinium 4 -toluene sulfonate *DCC = N, N'-Dicyclohexylcarbodiimide

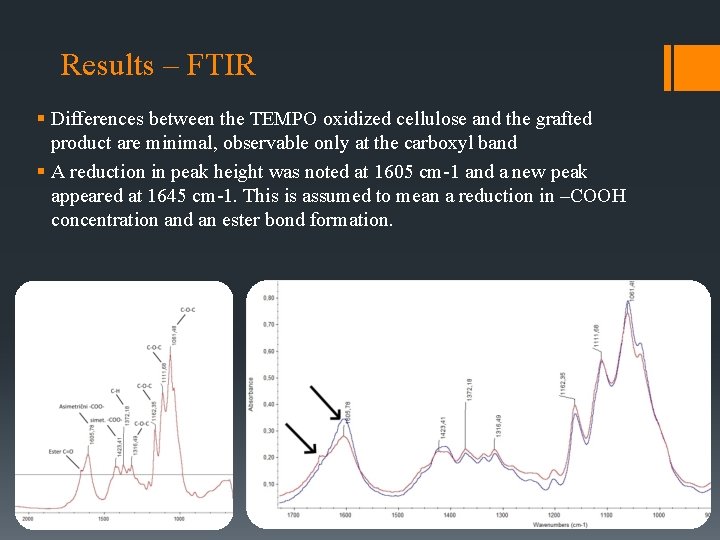
Results – FTIR § Differences between the TEMPO oxidized cellulose and the grafted product are minimal, observable only at the carboxyl band § A reduction in peak height was noted at 1605 cm-1 and a new peak appeared at 1645 cm-1. This is assumed to mean a reduction in –COOH concentration and an ester bond formation.

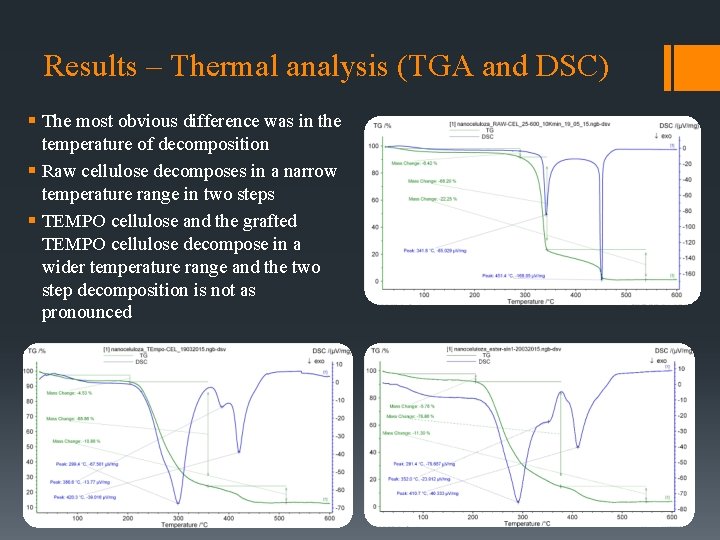
Results – Thermal analysis (TGA and DSC) § The most obvious difference was in the temperature of decomposition § Raw cellulose decomposes in a narrow temperature range in two steps § TEMPO cellulose and the grafted TEMPO cellulose decompose in a wider temperature range and the two step decomposition is not as pronounced

Conclusions § Hydroxyl groups were successfully oxidized to carboxyl groups with the use of TEMPO § The concentration of carboxyl groups was in accordance with the published articles, around 1, 3 mmol/g § Average nanofiber diameter for TEMPO oxidized nanocellulose was between 5 and 10 nm § The polyol polyether chain was successfully grafted to the TEMPO nanocellulose backbone § The decomposition temperature of TEMPO nanocellulose and the grafted TEMPO nanocellulose are significantly lower than the raw cellulose

Acknowledgement § The authors wish to gratefully acknowledge the Ministry of Higher Education, Science and Technology of the Republic of Slovenia, within the Program P 4 -0015 § COST Action FP 1205: Innovative applications of regenerated wood cellulose fibers for making it possible for me to attend this meeting