Govt Bilasa Girls P G Auto College Bilaspur

Govt. Bilasa Girls P. G. (Auto. ) College Bilaspur (C. G. ) Department Of Chemistry Seminar On Environmental Chemistry Topic : - Residual Chlorine & Chlorine Demand Presented by Dr. Pushpa Bhandari

Contents : 1. Residual Chlorine 2. Introduction 3. Reagent & Preparation of Reagent 4. 5. 6. 7. 8. 9. 10. 11. 12. 13. Estimation of free Chlorine in water: - Principal Reagent Procedure Calculation Result Chlorine Demand Introduction Graph Process Conclusion

• RESIDUAL CHLORINE • Introduction: - • Purpose of water disinfectant we use chlorine or hypochlorite. Hence clorination of water takes place • Chlorine determination is necessary in those water samle that have been treated with chlorine or hypochlorites. • Reagent: - • Orthotoluidine Reagent • Preparation of the Reagent: - 1 gram of orthotoluidine paste and in 5 ml of 25% HCL Paste is then dissolved in 200 ml of water Transfer to a litre measuring flask Further diluted 505 ml of water+ makeup 25% HCL in the mark of flask • Shake well & stored in colour bottle at room temperature • •

• Estimation of free chlorine in water: • Principal: • It is based on the oxidation of KI by free chlorine. When water samples is treated with an excess KI solution, the free chlorine, present in water sample oxidises KI & liberated I 2 in equivalent amounts. • The iodine so formed is dissolved in excess of KI giving a deep violet complex KI

• The amount of liberated iodine is then titrated against a standard solution of sodium thiosulphate using starch as an indicator. Reagent: 1. Standard Sodium thiosulphate solution (0. 025 N) 2. Glacial acetic acid 3. KI & starch

• Procedure: • Take 500 ml of the sample in a conical flask. Add 1 gram of KI & 5 ml acetic acid. Titrate with 0. 0025 N hypo solution until the colour of iodine is discharged. Add 1 ml starch indicator and titrate until the blue colour disappears. • Calculation: • Result: • Excess of chlorine in water can accelerate corrosion, deteriorate health, may cause tastes in canned foods or frozen products. Therefore residual chlorine content in pure water should not exceed 2 mg/L.

CHLORINE DEMAND • Introduction: • Chlorine demand may be defined as the quantity of chlorine required to produce a residual chlorine content between a trace and 0. 1 ppm after 10 minute contact.
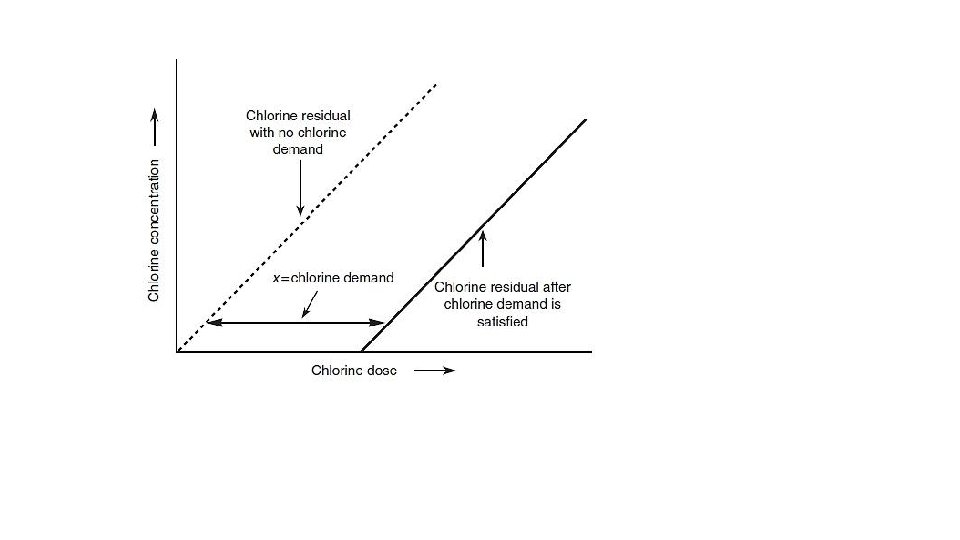

• Process: - • Take a regent bottle containing 250 ml of the sample and add to it standard chlorine water containing 0. 5 mg of chlorine from a burette. • Stir the mixture well and withdraw 0. 25 ml of the solution with a pipette on a spot place and immediately test it for chlorine. • Chlorine Test: - 0. 25 ml samle containing chlorine water on spot plate + a drop of orthotoluidine reagent • Note : - condition for orthotoluidine react with water 1. p. H is less than four 2. At low concentration of chlorine react with this reagent gives a colour pale yellow & high concentration give orange. • Continue adding the standard chlorine water in 0. 5 ml portion, mixing and testing as before until there is a light excess of chlorine which is indicated by light yellow colour. • The quantity of chlorine required is regarded as the immediate chlorine demand. • Now again take a similar sample and add to it standard chlorine water in an amount equivalent to the immediate chlorine demand plus 0. 5 ml more • Stir the mixture well and allow it to stand for exactly 10 minute. • After completion 10 minute, immediately test a portion for residual chlorine as described above.


- Slides: 11