GORE VIABAHN Endoprosthesis Speakers Presentation Resource GORE VIABAHN
GORE VIABAHN® Endoprosthesis Speaker’s Presentation Resource GORE, VIABAHN ®, and designs are trademarks of W. L. Gore & Associates. © 2009 W. L. Gore & Associates, Inc. 1

Agenda • Product Description • Clinical Performance • Propaten Bioactive Surface • Latest Device Revisions 2 GORE, VIABAHN ®, and designs are trademarks of W. L. Gore & Associates. © 2009 W. L. Gore & Associates, Inc.
Product Description GORE, VIABAHN ®, and designs are trademarks of W. L. Gore & Associates. © 2009 W. L. Gore & Associates, Inc. 3
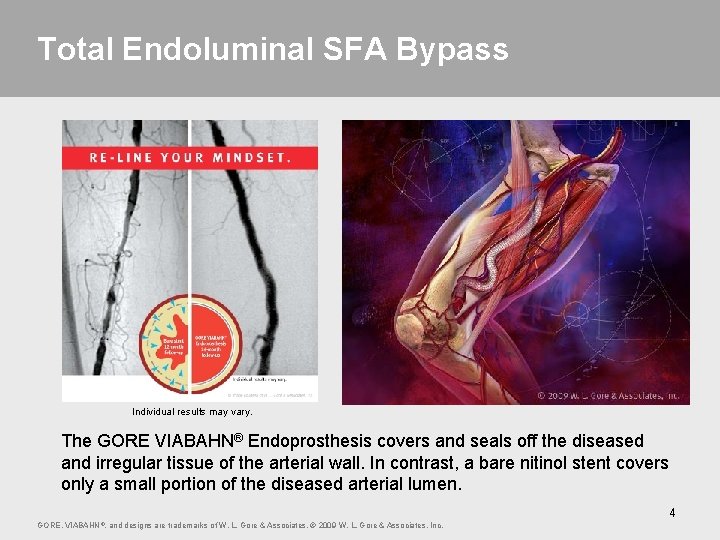
Total Endoluminal SFA Bypass Individual results may vary. The GORE VIABAHN® Endoprosthesis covers and seals off the diseased and irregular tissue of the arterial wall. In contrast, a bare nitinol stent covers only a small portion of the diseased arterial lumen. 4 GORE, VIABAHN ®, and designs are trademarks of W. L. Gore & Associates. © 2009 W. L. Gore & Associates, Inc.
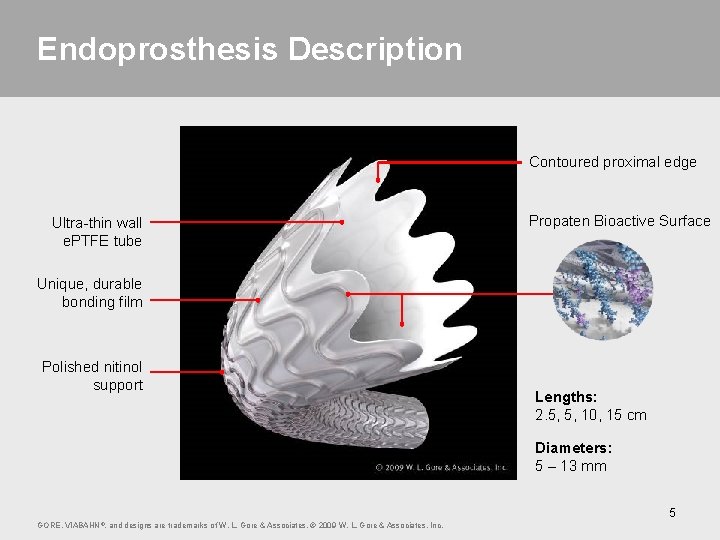
Endoprosthesis Description Contoured proximal edge Ultra-thin wall e. PTFE tube Propaten Bioactive Surface Unique, durable bonding film Polished nitinol support Lengths: 2. 5, 5, 10, 15 cm Diameters: 5 – 13 mm 5 GORE, VIABAHN ®, and designs are trademarks of W. L. Gore & Associates. © 2009 W. L. Gore & Associates, Inc.

Features and Benefits • Nitinol Stent • e. PTFE Lining • Lower Profile • Heparin-bonded Surface • Contoured proximal edge – Conformable and durable – Covers diseased tissue – Streamlined delivery system facilitates treating challenging SFA lesions – Intended to provide sustained thromboresistance – May improve flow dynamics as blood enters endoprosthesis 6 GORE, VIABAHN ®, and designs are trademarks of W. L. Gore & Associates. © 2009 W. L. Gore & Associates, Inc.

TIP to HUB Deployment 0. 035" guidewire Guidewire Access 1. Gain access to lesion with the guidewire. Device Deployment 4. Slowly pull deployment knob in a smooth motion. Balloon Dilatation 2. Pre-dilate with appropriately sized balloon. Balloon Touch-up 5. Seat balloon well inside device during touch-up. Device Location 3. Confirm initial landing zone before deployment. Device Location 6. Land proximal edge at least 1 cm into healthy vessel. 7 GORE, VIABAHN ®, and designs are trademarks of W. L. Gore & Associates. © 2009 W. L. Gore & Associates, Inc.

Clinical Lessons Learned • • Avoid non-compliant lesions Ensure adequate inflow and outflow (e. g. , at least one vessel run-off) Correct sizing is key Land device at least 1 cm into healthy vessel proximally and distally to the lesion Every region pre-treated with Percutaneous Transluminal Angioplasty (PTA) needs to be covered by the device During post dilatation, only balloon inside the region covered by the device Consider an antiplatelet regimen post-procedure 8 GORE, VIABAHN ®, and designs are trademarks of W. L. Gore & Associates. © 2009 W. L. Gore & Associates, Inc.
Clinical Performance GORE, VIABAHN ®, and designs are trademarks of W. L. Gore & Associates. © 2009 W. L. Gore & Associates, Inc. 9
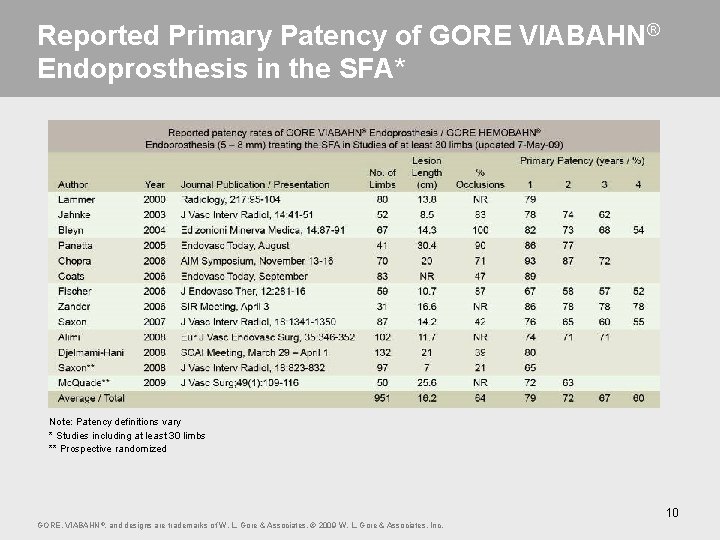
Reported Primary Patency of GORE VIABAHN® Endoprosthesis in the SFA* Note: Patency definitions vary * Studies including at least 30 limbs ** Prospective randomized 10 GORE, VIABAHN ®, and designs are trademarks of W. L. Gore & Associates. © 2009 W. L. Gore & Associates, Inc.
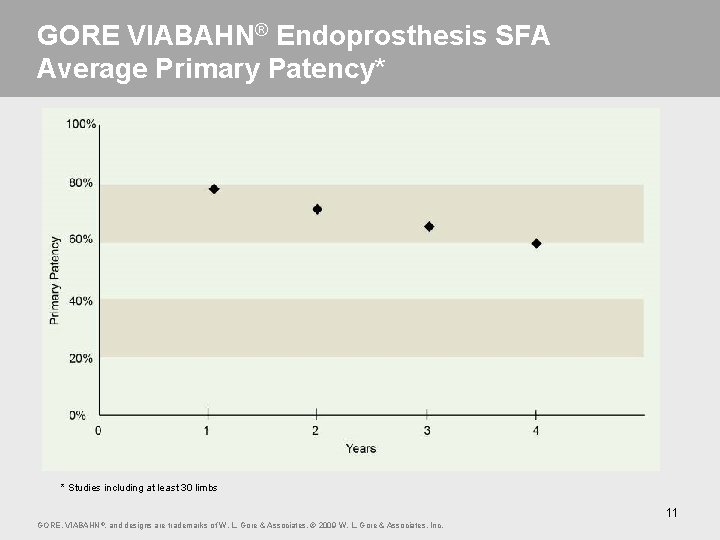
GORE VIABAHN® Endoprosthesis SFA Average Primary Patency* * Studies including at least 30 limbs 11 GORE, VIABAHN ®, and designs are trademarks of W. L. Gore & Associates. © 2009 W. L. Gore & Associates, Inc.
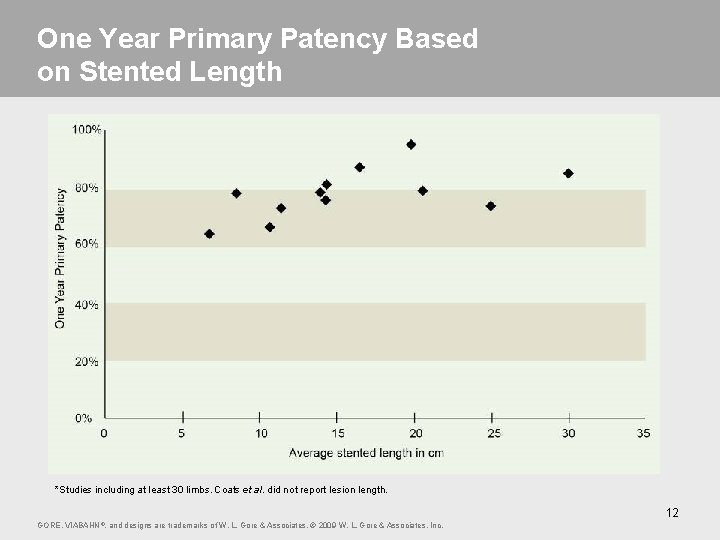
One Year Primary Patency Based on Stented Length *Studies including at least 30 limbs. Coats et al. did not report lesion length. 12 GORE, VIABAHN ®, and designs are trademarks of W. L. Gore & Associates. © 2009 W. L. Gore & Associates, Inc.
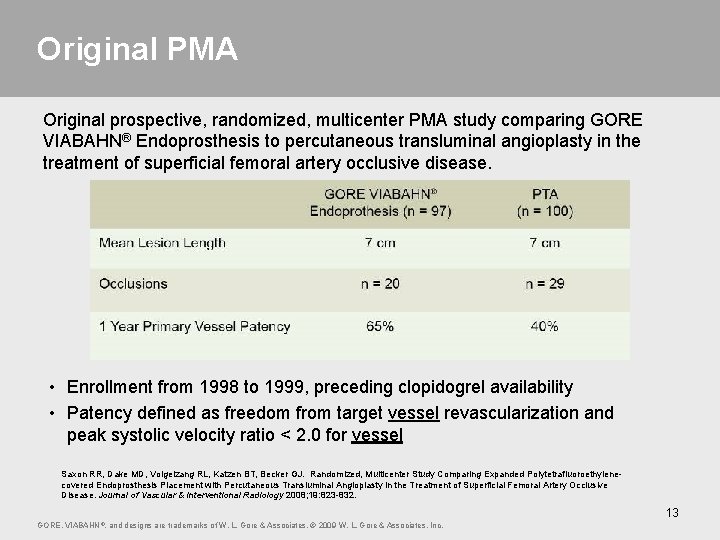
Original PMA Original prospective, randomized, multicenter PMA study comparing GORE VIABAHN® Endoprosthesis to percutaneous transluminal angioplasty in the treatment of superficial femoral artery occlusive disease. • Enrollment from 1998 to 1999, preceding clopidogrel availability • Patency defined as freedom from target vessel revascularization and peak systolic velocity ratio < 2. 0 for vessel Saxon RR, Dake MD, Volgelzang RL, Katzen BT, Becker GJ. Randomized, Multicenter Study Comparing Expanded Polytetrafluoroethylenecovered Endoprosthesis Placement with Percutaneous Transluminal Angioplasty in the Treatment of Superficial Femoral Artery Occlusive Disease. Journal of Vascular & Interventional Radiology 2008; 19: 823 -832. 13 GORE, VIABAHN ®, and designs are trademarks of W. L. Gore & Associates. © 2009 W. L. Gore & Associates, Inc.
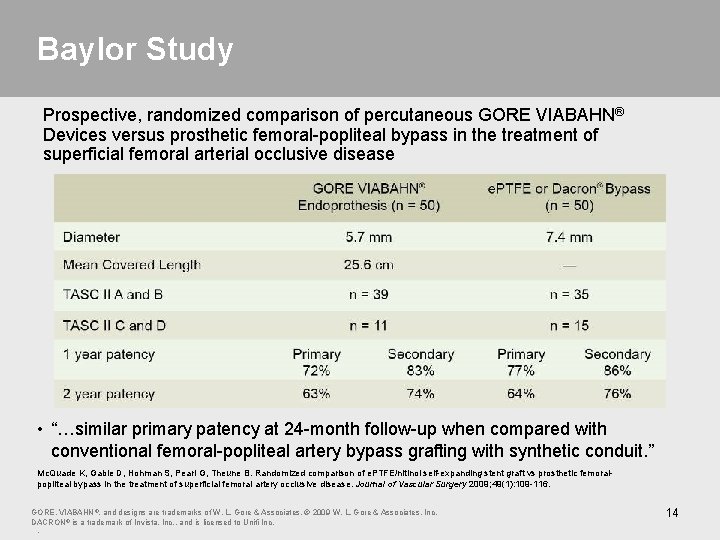
Baylor Study Prospective, randomized comparison of percutaneous GORE VIABAHN® Devices versus prosthetic femoral-popliteal bypass in the treatment of superficial femoral arterial occlusive disease • “…similar primary patency at 24 -month follow-up when compared with conventional femoral-popliteal artery bypass grafting with synthetic conduit. ” Mc. Quade K, Gable D, Hohman S, Pearl G, Theune B. Randomized comparison of e. PTFE/nitinol self-expanding stent graft vs prosthetic femoralpopliteal bypass in the treatment of superficial femoral artery occlusive disease. Journal of Vascular Surgery 2009; 49(1): 109 -116. GORE, VIABAHN ®, and designs are trademarks of W. L. Gore & Associates. © 2009 W. L. Gore & Associates, Inc. DACRON® is a trademark of Invista, Inc. , and is licensed to Unifi Inc. . 14
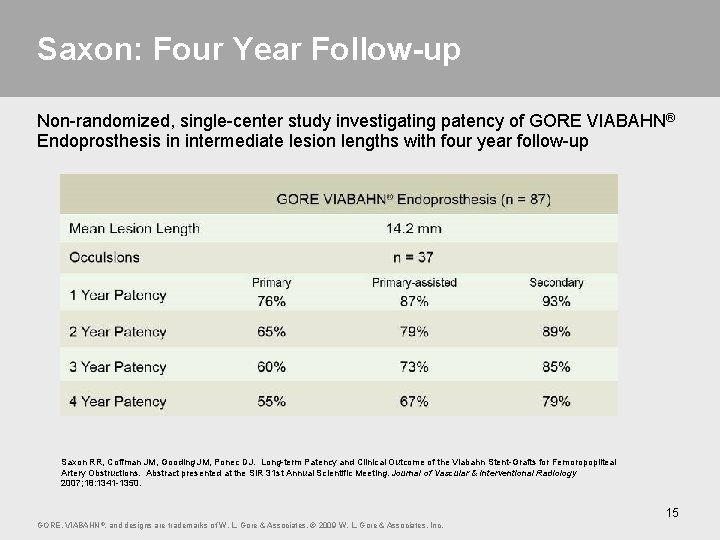
Saxon: Four Year Follow-up Non-randomized, single-center study investigating patency of GORE VIABAHN® Endoprosthesis in intermediate lesion lengths with four year follow-up Saxon RR, Coffman JM, Gooding JM, Ponec DJ. Long-term Patency and Clinical Outcome of the Viabahn Stent-Grafts for Femoropopliteal Artery Obstructions. Abstract presented at the SIR 31 st Annual Scientific Meeting. Journal of Vascular & Interventional Radiology 2007; 18: 1341 -1350. 15 GORE, VIABAHN ®, and designs are trademarks of W. L. Gore & Associates. © 2009 W. L. Gore & Associates, Inc.
GORE VIABAHN® Endoprosthesis with Propaten Bioactive Surface GORE, VIABAHN ®, and designs are trademarks of W. L. Gore & Associates. © 2009 W. L. Gore & Associates, Inc. 16

Unique Bioactive Heparin Bonding Technology • Heparin molecules are bonded directly to the surface of the endoprosthesis – Heparin is a polysaccharide anticoagulant with a long history of clinical use. 1 – Heparin has a potent antiproliferative effect on vascular smooth muscle cells. 2 • A Gore proprietary end-point attachment mechanism (CARMEDA® Bio. Active Surface Technology (CBAS®) allows for retention of bioactivity • The result is an endoprosthesis intended to provide sustained thromboresistance 1 Hirsh J, Anand SS, Halperin JL, Fuster V. AHA Scientific statement. Guide to anticoagulant therapy: heparin. Circulation 2001; 103: 2994 -3018. AW, Karnowsky MJ. Suppression by heparin of smooth muscle cell proliferation in injured arteries. Nature 1977; 265: 625 -6. 2 Clowes GORE, VIABAHN ®, and designs are trademarks of W. L. Gore & Associates. © 2009 W. L. Gore & Associates, Inc. CARMEDA® and CBAS ® are trademarks of Carmeda AB, a wholly owned subsidiary of W. L. Gore & Associates. 17
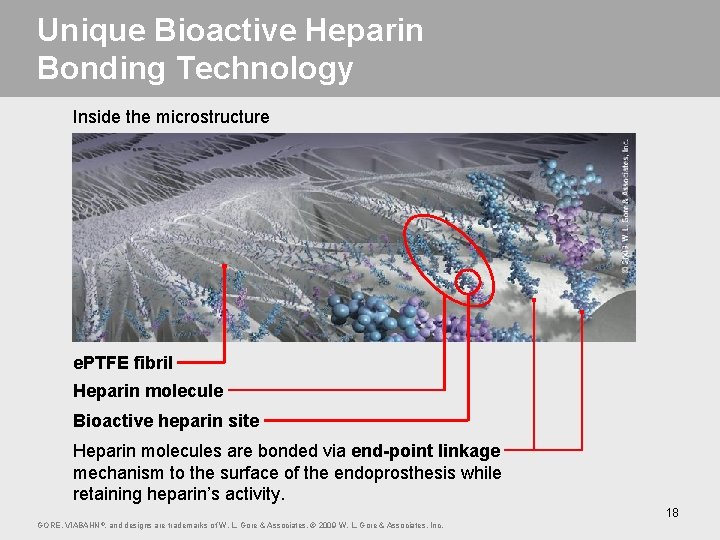
Unique Bioactive Heparin Bonding Technology Inside the microstructure e. PTFE fibril Heparin molecule Bioactive heparin site Heparin molecules are bonded via end-point linkage mechanism to the surface of the endoprosthesis while retaining heparin’s activity. 18 GORE, VIABAHN ®, and designs are trademarks of W. L. Gore & Associates. © 2009 W. L. Gore & Associates, Inc.
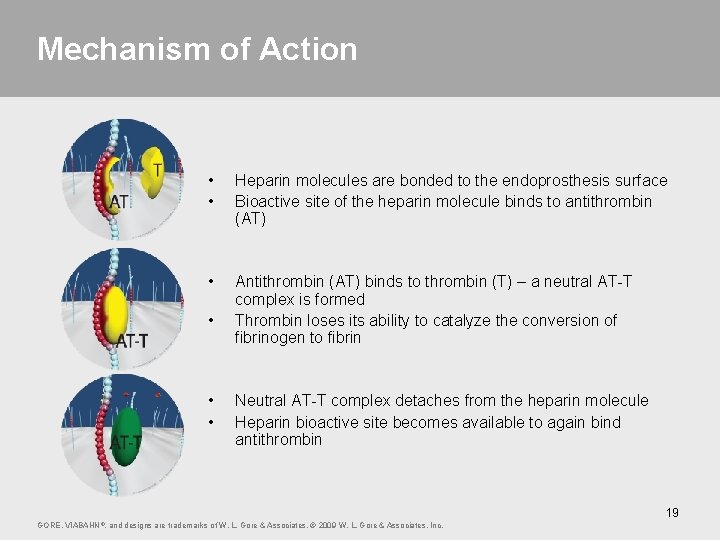
Mechanism of Action • • Heparin molecules are bonded to the endoprosthesis surface Bioactive site of the heparin molecule binds to antithrombin (AT) • Antithrombin (AT) binds to thrombin (T) – a neutral AT-T complex is formed Thrombin loses its ability to catalyze the conversion of fibrinogen to fibrin • • • Neutral AT-T complex detaches from the heparin molecule Heparin bioactive site becomes available to again bind antithrombin 19 GORE, VIABAHN ®, and designs are trademarks of W. L. Gore & Associates. © 2009 W. L. Gore & Associates, Inc.
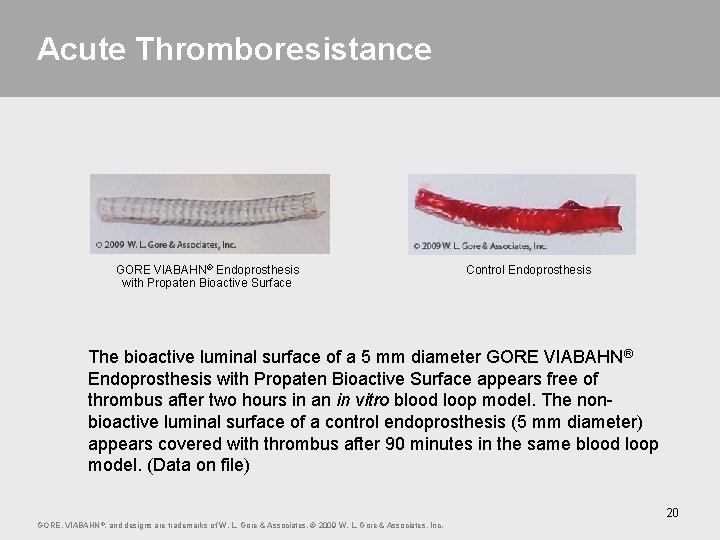
Acute Thromboresistance GORE VIABAHN® Endoprosthesis with Propaten Bioactive Surface Control Endoprosthesis The bioactive luminal surface of a 5 mm diameter GORE VIABAHN® Endoprosthesis with Propaten Bioactive Surface appears free of thrombus after two hours in an in vitro blood loop model. The nonbioactive luminal surface of a control endoprosthesis (5 mm diameter) appears covered with thrombus after 90 minutes in the same blood loop model. (Data on file) 20 GORE, VIABAHN ®, and designs are trademarks of W. L. Gore & Associates. © 2009 W. L. Gore & Associates, Inc.
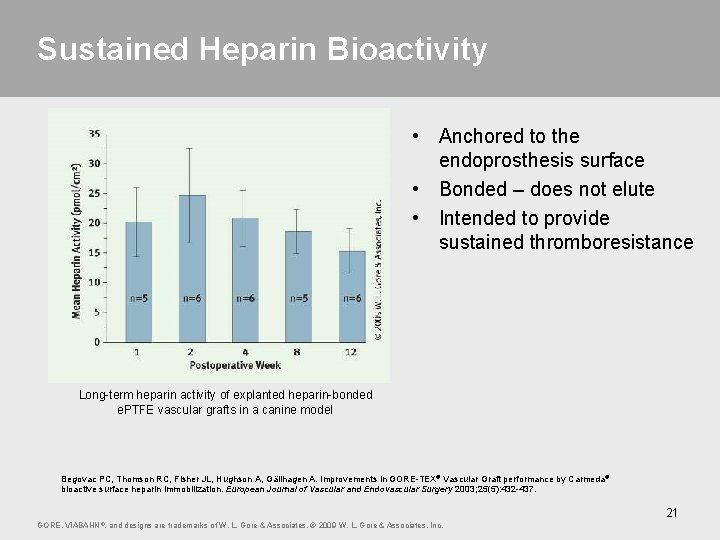
Sustained Heparin Bioactivity • Anchored to the endoprosthesis surface • Bonded – does not elute • Intended to provide sustained thromboresistance Long-term heparin activity of explanted heparin-bonded e. PTFE vascular grafts in a canine model Begovac PC, Thomson RC, Fisher JL, Hughson A, Gällhagen A. Improvements in GORE-TEX® Vascular Graft performance by Carmeda® bioactive surface heparin immobilization. European Journal of Vascular and Endovascular Surgery 2003; 25(5): 432 -437. 21 GORE, VIABAHN ®, and designs are trademarks of W. L. Gore & Associates. © 2009 W. L. Gore & Associates, Inc.
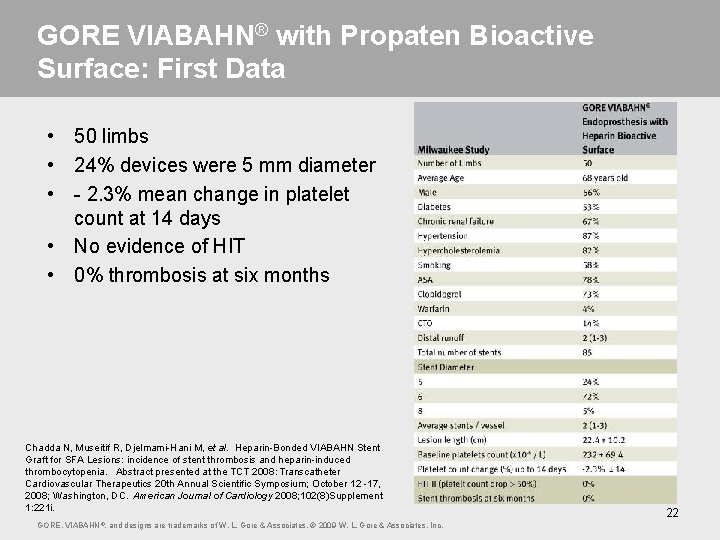
GORE VIABAHN® with Propaten Bioactive Surface: First Data • 50 limbs • 24% devices were 5 mm diameter • - 2. 3% mean change in platelet count at 14 days • No evidence of HIT • 0% thrombosis at six months Chadda N, Museitif R, Djelmami-Hani M, et al. Heparin-Bonded VIABAHN Stent Graft for SFA Lesions: incidence of stent thrombosis and heparin-induced thrombocytopenia. Abstract presented at the TCT 2008: Transcatheter Cardiovascular Therapeutics 20 th Annual Scientific Symposium; October 12 -17, 2008; Washington, DC. American Journal of Cardiology 2008; 102(8)Supplement 1: 221 i. GORE, VIABAHN ®, and designs are trademarks of W. L. Gore & Associates. © 2009 W. L. Gore & Associates, Inc. 22
Latest Device Revisions GORE, VIABAHN ®, and designs are trademarks of W. L. Gore & Associates. © 2009 W. L. Gore & Associates, Inc. 23

A New Beginning for the Proximal End New Contoured Edge GORE, VIABAHN ®, and designs are trademarks of W. L. Gore & Associates. © 2009 W. L. Gore & Associates, Inc. 24
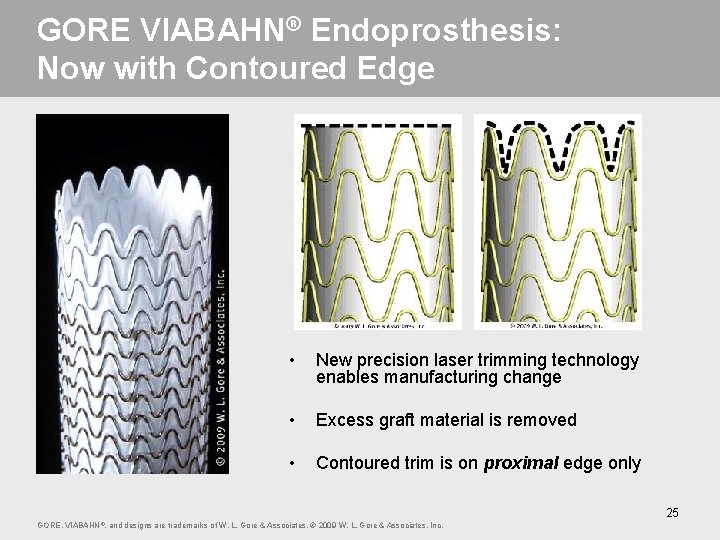
GORE VIABAHN® Endoprosthesis: Now with Contoured Edge • New precision laser trimming technology enables manufacturing change • Excess graft material is removed • Contoured trim is on proximal edge only 25 GORE, VIABAHN ®, and designs are trademarks of W. L. Gore & Associates. © 2009 W. L. Gore & Associates, Inc.

Reason for Modification • Improves device apposition to the vessel wall when oversizing prevents device expansion to its nominal diameter – Contoured edge may improve flow dynamics at proximal end 26 GORE, VIABAHN ®, and designs are trademarks of W. L. Gore & Associates. © 2009 W. L. Gore & Associates, Inc.
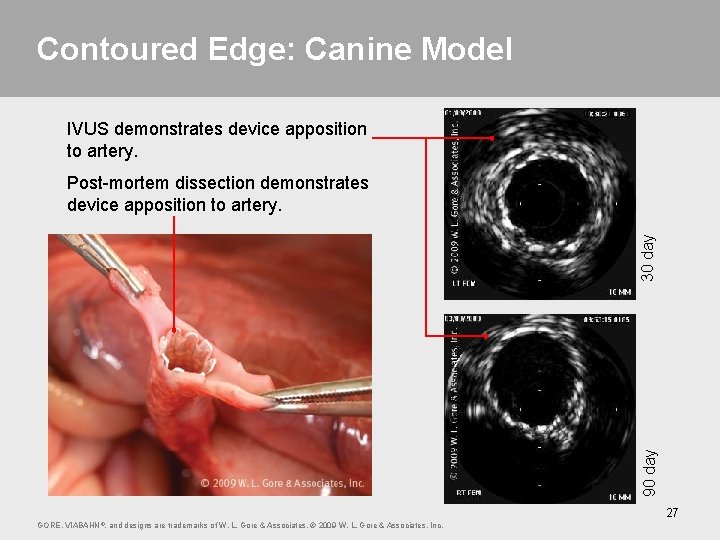
Contoured Edge: Canine Model IVUS demonstrates device apposition to artery. 90 day 30 day Post-mortem dissection demonstrates device apposition to artery. 27 GORE, VIABAHN ®, and designs are trademarks of W. L. Gore & Associates. © 2009 W. L. Gore & Associates, Inc.

Summary of Modification • Result of a manufacturing change implementing laser edge trimming technology • Excess material at the proximal edge removed • The Instructions for Use, including sizing and placement recommendations remain unchanged • May improve the flow dynamics of blood entering the endoprosthesis 28 GORE, VIABAHN ®, and designs are trademarks of W. L. Gore & Associates. © 2009 W. L. Gore & Associates, Inc.

The Evolution of Performance 29

Streamlined Deployment for Larger Sizes • 9 – 13 mm diameter endoprosthesis now incorporate the same delivery system as the 5 – 8 mm diameter devices 30 GORE, VIABAHN ®, and designs are trademarks of W. L. Gore & Associates. © 2009 W. L. Gore & Associates, Inc.

Product Comparison You knew it as GORE HEMOBAHN® Endoprosthesis; now it is the next generation 9 – 13 mm diameter GORE VIABAHN® Endoprosthesis with PROPATEN Bioactive Surface GORE, VIABAHN ®, and designs are trademarks of W. L. Gore & Associates. © 2009 W. L. Gore & Associates, Inc. 31
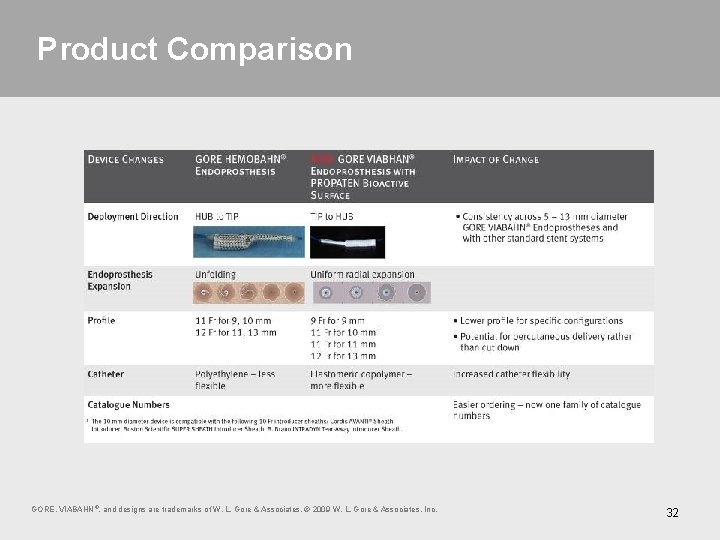
Product Comparison GORE, VIABAHN ®, and designs are trademarks of W. L. Gore & Associates. © 2009 W. L. Gore & Associates, Inc. 32

Large Diameter Deployment Changes TIP to HUB deployment Uniform radial expansion – 0. 035" guidewire compatibility – Lower profile on some configurations HUB to TIP deployment Unrolling expansion – 0. 025" guidewire compatibility 33 GORE, VIABAHN ®, and designs are trademarks of W. L. Gore & Associates. © 2009 W. L. Gore & Associates, Inc.
Currently Enrolling Clinical Studies GORE, VIABAHN ®, and designs are trademarks of W. L. Gore & Associates. © 2009 W. L. Gore & Associates, Inc. 34

Gore VIPER Clinical Study 35
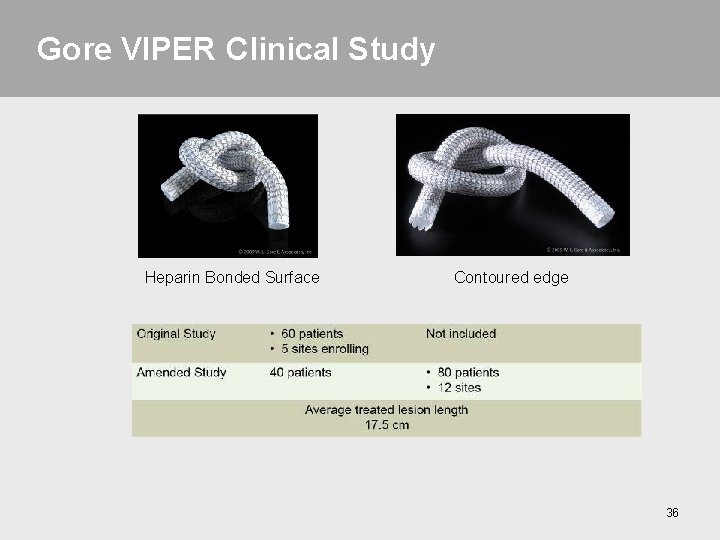
Gore VIPER Clinical Study Heparin Bonded Surface Contoured edge 36

VIASTAR Clinical Study 37

38 Products listed may not be available in all markets. GORE, VIABAHN ®, and designs are trademarks of W. L. Gore & Associates. © 2009 W. L. Gore & Associates, Inc. AL 0752 -EU 3 SEPTEMBER 2009
- Slides: 38