Good Web Sites about Minerals http galaxy einet

Good Web Sites about Minerals: • http: //galaxy. einet. net/images/ge ms/gems-icons. html • Smithsonian Gem and Mineral Collection -- lots of spectacular mineral pictures and good information

The Rock Cycle A model that describes all the crustal processes by which rock is formed, modified, transported, decomposed, and reformed. N. Lindsley-Griffin, 1999

Rocks are made up of minerals N. Lindsley-Griffin, 1999

What is a Mineral? • Occurs naturally • Not previously living (inorganic) • Solid • Definite chemical composition • Orderly internal atomic arrangement N. Lindsley-Griffin, 1998

Ivory, bone, seashells, coral, petrified wood, and other previously living things… are NOT minerals N. Lindsley-Griffin, 1998
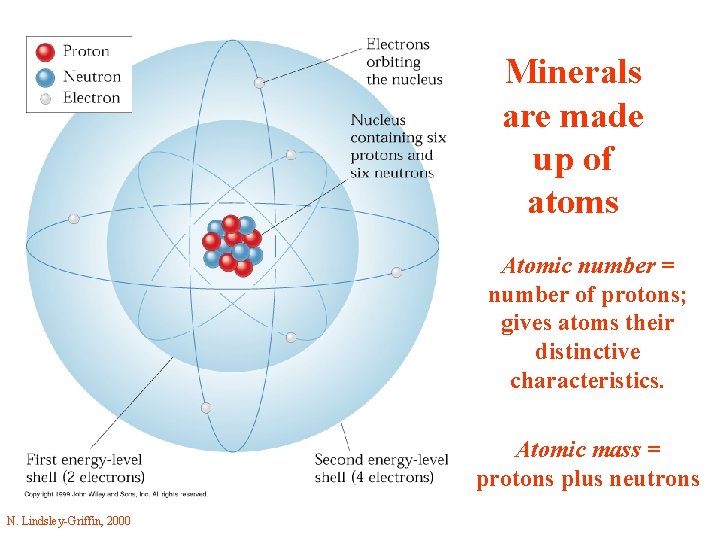
Minerals are made up of atoms Atomic number = number of protons; gives atoms their distinctive characteristics. Atomic mass = protons plus neutrons N. Lindsley-Griffin, 2000

Ions combine to form compounds Here, the Li atom loses an electron to become a + positively charged cation. The F atom gains an electron to become a negatively charged anion. Positive and negative charges attract each other to form an IONIC BOND N. Lindsley-Griffin, 1999
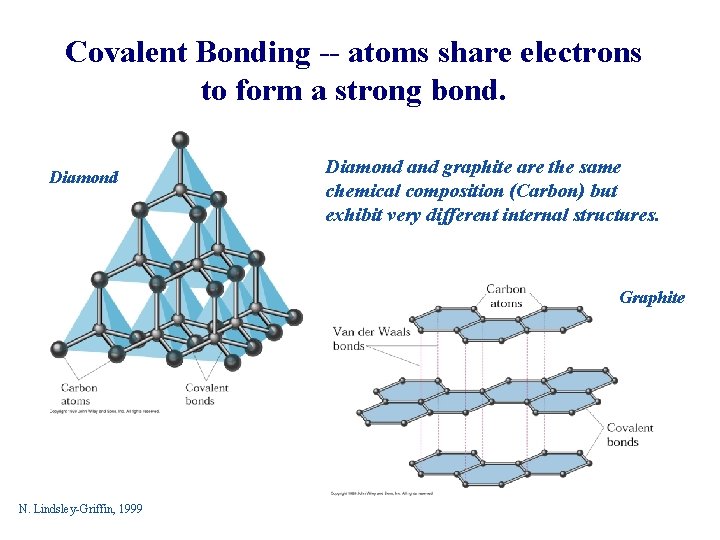
Covalent Bonding -- atoms share electrons to form a strong bond. Diamond and graphite are the same chemical composition (Carbon) but exhibit very different internal structures. Graphite N. Lindsley-Griffin, 1999
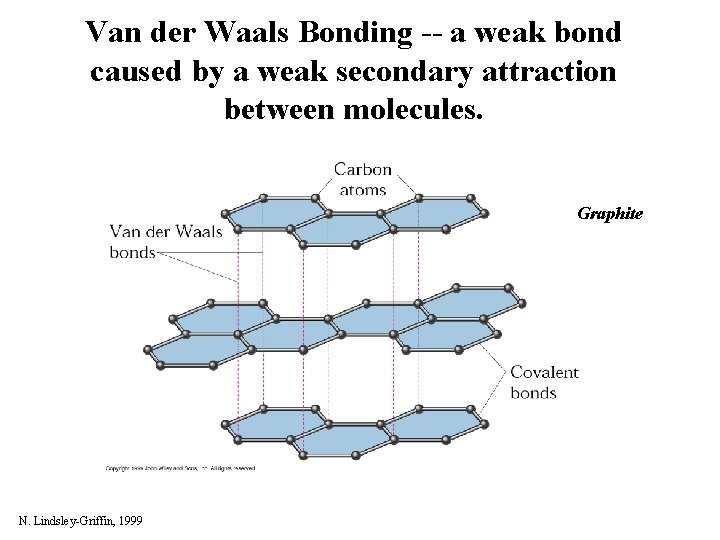
Van der Waals Bonding -- a weak bond caused by a weak secondary attraction between molecules. Graphite N. Lindsley-Griffin, 1999

Orderly array of atoms in the mineral Galena: large atoms - sulfur, small - lead As seen with a scanning-tunneling microscope N. Lindsley-Griffin, 1999

The orderly arrangement of atoms in Galena controls its physical properties. Error in textbook on Figure 2. 7: Sulfur atoms, not “oxygen” Sulfur atom N. Lindsley-Griffin, 1999

Crystal Interfacial Angles Interfacial angles of a given mineral are constant for all crystals The faces of quartz always form 120 -degree angles © Houghton Mifflin 1998; Lindsley 2000

Crystal Interfacial Angles • Even when the crystals are different sizes, angles between equivalent faces are the same • For different minerals, angles are different. 3 2 4 4 3 1 2 Quartz N. Lindsley-Griffin, 1999 1

Habit - distinctive shape of the way the mineral commonly (habitually) appears N. Lindsley-Griffin, 1999 Concentric shells -- malachite

Habit N. Lindsley-Griffin, 1998

Crystal shape and Habit result from internal atomic structure N. Lindsley-Griffin, 1998

Cleavage - tendency to break along planes of weak bonds N. Lindsley-Griffin, 1998

Mica cleaves along planes of the weakest bonds N. Lindsley-Griffin, 1998

Fracture describes how a mineral breaks on an irregular surface rather than along cleavage planes Types of fracture: Smooth Rough Splintery Conchoidal N. Lindsley-Griffin, 1998

Hardness -- the resistance to being scratched Mohs scale measures relative hardness, the ability of one mineral to scratch another Diamond Corundum Topaz Quartz Feldspar Apatite Fluorite Calcite Gypsum Talc 10 9 8 7 6 5 4 3 2 1 Steel file Glass (6. 5) (5. 5 - 6) Copper penny Fingernail (3. 0) (2. 5) Mohs hardness scale: 1 = softest mineral; 10 = hardest © Houghton Mifflin 1998; Lindsley 2000

The 10 minerals of Mohs relative hardness scale 3 Calcite 1 Talc 4 Fluorite 2 Gypsum 9 Corundum 6 Feldspar N. Lindsley-Griffin, 1999 5 Apatite 7 Quartz 8 Topaz 10 Diamond

Luster - the appearance of a mineral in reflected light Types of luster: Metallic Nonmetallic Pearly Vitreous Resinous Silky Dull Earthy Metallic luster of pyrite © Houghton Mifflin; N. Lindsley-Griffin, 1998. Earthy luster of limonite

Metallic Luster is characteristic of ore minerals and others that contain metal cations. N. Lindsley-Griffin, 1998

Resinous luster in sphalerite Pearly luster in talc N. Lindsley-Griffin, 1999

Color is useful in recognizing some minerals, but not all N. Lindsley-Griffin, 1998

Color adds value to gems N. Lindsley-Griffin, 1999

Streak, the color of the mineral’s powder, may be more useful than color in identifying a mineral N. Lindsley-Griffin, 1998
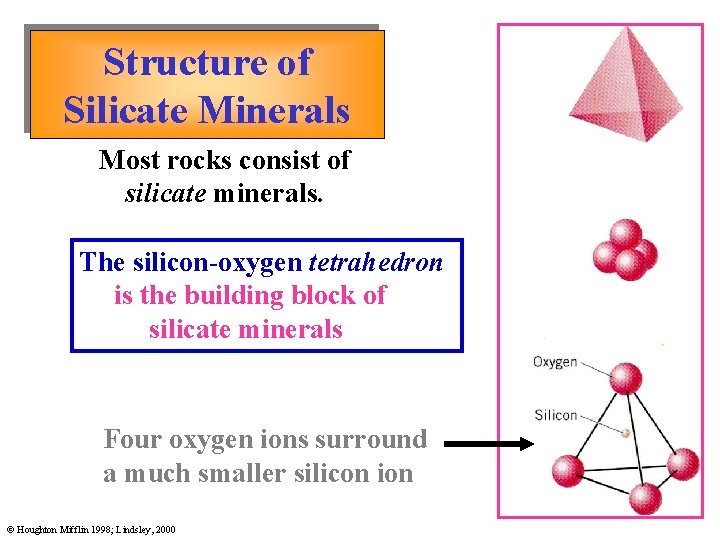
Structure of Silicate Minerals Most rocks consist of silicate minerals. The silicon-oxygen tetrahedron is the building block of silicate minerals Four oxygen ions surround a much smaller silicon ion © Houghton Mifflin 1998; Lindsley, 2000
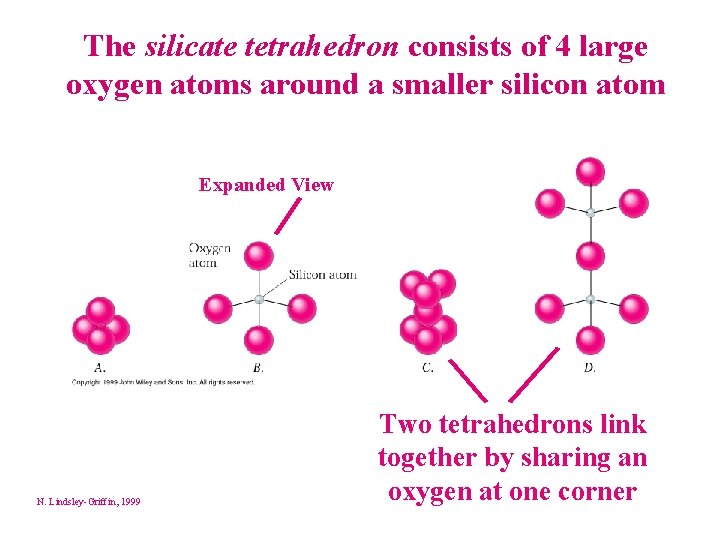
The silicate tetrahedron consists of 4 large oxygen atoms around a smaller silicon atom Expanded View N. Lindsley-Griffin, 1999 Two tetrahedrons link together by sharing an oxygen at one corner

Silicate tetrahedrons can form chains, sheets, and three-dimensional nets by sharing their oxygen atoms. Each influences mineral properties. N. Lindsley-Grifin, 1999
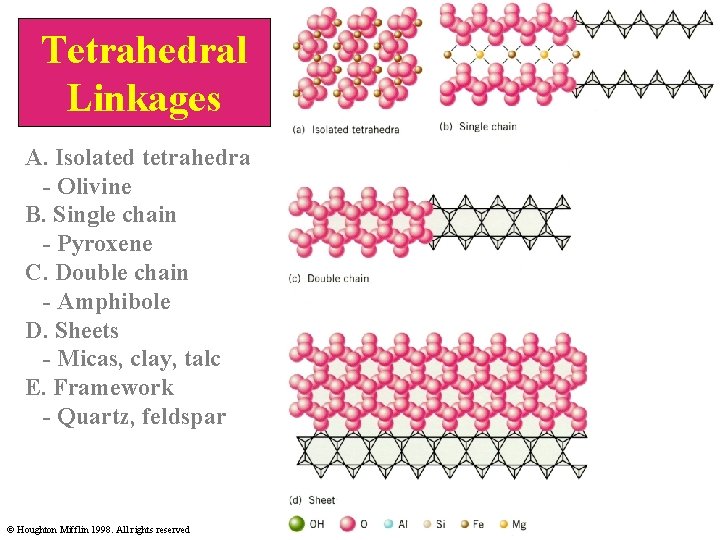
Tetrahedral Linkages A. Isolated tetrahedra - Olivine B. Single chain - Pyroxene C. Double chain - Amphibole D. Sheets - Micas, clay, talc E. Framework - Quartz, feldspar © Houghton Mifflin 1998. All rights reserved

Polymorphs are minerals with the same chemical composition, but different internal atomic structures N. Lindsley-Griffin, 1998

SUMMARY: PHYSICAL PROPERTIES of MINERALS Crystal Structure Crystal Form Interfacial Angles Habit Cleavage Fracture © Houghton Mifflin 1998; N. Lindsley-Griffin, 1999. All rights reserved Hardness Luster Color Streak Specific Gravity (Density)
- Slides: 33