Good Manufacturing Practices HVAC Heating Ventilation and Air




















- Slides: 23

Good Manufacturing Practices: HVAC Heating, Ventilation and Air. Conditioning (HVAC) Part 1: Introduction and overview WHO Technical Report Series, No. 961, 2011. Annex 5 HVAC | Slide 1 of 23 2013

Good Manufacturing Practices: HVAC Objectives To understand: l The need for HVAC systems l The role of HVAC in protection: – Product – Personnel – Environment l The role of HVAC in dust control l HVAC system design and its components l Commissioning, qualification and maintenance HVAC | Slide 2 of 23 2013 1, 2

HVAC Introduction and Scope HVAC l HVAC systems can have an impact on product quality l It can provide comfortable conditions for operators l The impact on premises and prevention of contamination and cross-contamination to be considered at the design stage l Temperature, relative humidity control where appropriate l Supplement to basic GMP text | Slide 3 of 23 2013 1, 2

HVAC l This presentation focuses on HVAC systems for OSD l To fully understand the technical issues, it is important to know the definitions of the terms used HVAC l These include: l Contamination and Cross-contamination l As built, at rest, in operation l Infiltration, exfiltration etc | Slide 4 of 23 2013 1, 2

HVAC For example: What is contamination? It is "the undesired introduction of impurities (chemical/ microbial/ foreign matter into or on to starting material or intermediate – during sampling, production, packaging or repackaging". Impurities could include products or substances other than the product manufactured, foreign products, particulate matter, microorganisms, endotoxins (degraded microorganisms), etc. Glossary HVAC | Slide 5 of 23 2013

HVAC What is Cross-contamination? "Contamination of a starting material, intermediate product, or finished product with another starting material or product during production". Cross-contamination can result from, e. g. 1. Poorly designed, operated or maintained air-handling systems and dust extraction systems 2. Inadequate procedures for, and movement of personnel, materials and equipment 3. Insufficiently cleaned equipment HVAC | Slide 6 of 23 2013 Glossary, 4. 1. 11

HVAC Cross-contamination can be minimized by, e. g. 1. Personnel procedures 2. Adequate premises 3. Use of closed production systems 4. Adequate, validated cleaning procedures 5. Appropriate levels of protection of product 6. Correct air pressure cascade HVAC | Slide 7 of 23 2013

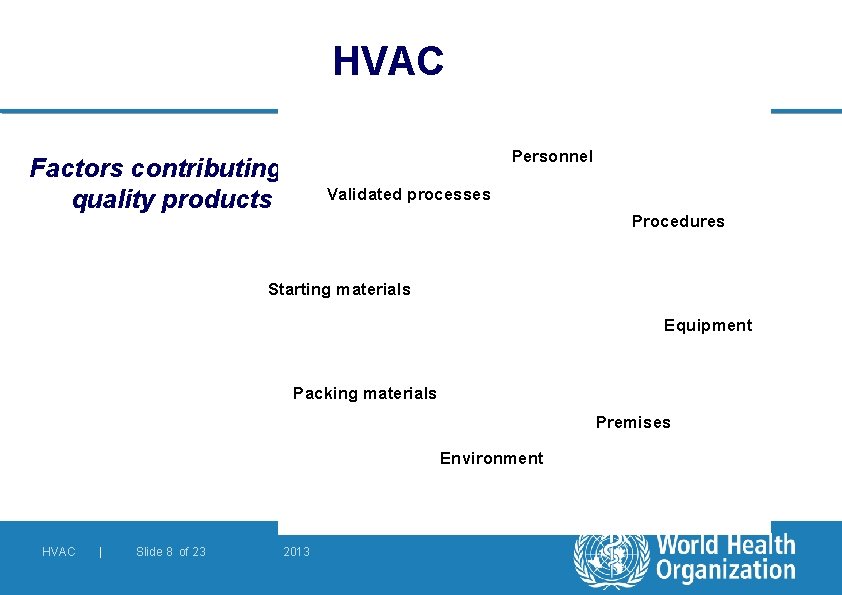
HVAC Factors contributing to quality products Personnel Validated processes Procedures Starting materials Equipment Packing materials Premises Environment HVAC | Slide 8 of 23 2013

HVAC Consider the following when designing an HVAC system: HVAC l Layout of the premises l Product range l Airlocks, Lobbies, Doors l Required pressure differentials l Air flows l Temperature and relative humidity l Prevention of contamination and cross contamination | Slide 9 of 23 2013 1, 2

HVAC The guideline further focuses on three concepts of the system: l Product protection – Contamination – Cross-contamination – Environmental conditions l Personnel protection – Prevent contact – Comfort conditions l Environment protection HVAC | Slide 10 of 23 2013 4

HVAC Protection: Product and personnel l Areas of manufacturing should be classified as "clean areas; clean zones; cleanrooms; or controlled areas” l To achieve a clean area classification – control : – Building finishes and structure – Air filtration – Air change rate – Room pressure – Temperature – Relative humidity – Material and personnel flow – Outside environment – Occupancy and type of product HVAC | Slide 11 of 23 2013 4. 1. 1 - 4. 1. 3

HVAC l Air filtration and air change rate needed to attain classification l Risk assessment. Normally need 6 – 20 air changes per hour l Air change rate is dependent on factors, e. g. – – – – HVAC | Required condition Product characteristics Quality of filtration of air Particles generated (operators, machines, process) Room configuration Supply and return / extract air locations Air required (room heat load, containment) Balance and room pressure requirements Slide 12 of 23 2013 4. 1. 4 - 4. 1. 6

HVAC The classification should be achieved in the state as specified (1): l "As built" – Bare room, without equipment or personnel 4. 1. 7 - 4. 1. 8 HVAC | Slide 13 of 23 2013

HVAC The classification should be achieved in the state as specified (2): l "At rest" – Equipment may be operating, but no operators present 4. 1. 9 HVAC | Slide 14 of 23 2013

HVAC The classification should be achieved in the state as specified (3): l "In operation" – Normal production process with equipment and personnel – Clean up time validated – normally about 20 minutes 4. 1. 10 HVAC | Slide 15 of 23 2013

HVAC Control of contaminants l Protect materials and products during manufacture l Airborne contaminants – effective ventilation and filtration l External contaminants - effective filtration l Internal contaminants - dilution and flushing, or displacement airflow l Level of protection: Airborne particulates and level of filtration 4. 1. 11 - 4. 1. 15 considered critical HVAC | Slide 16 of 23 2013

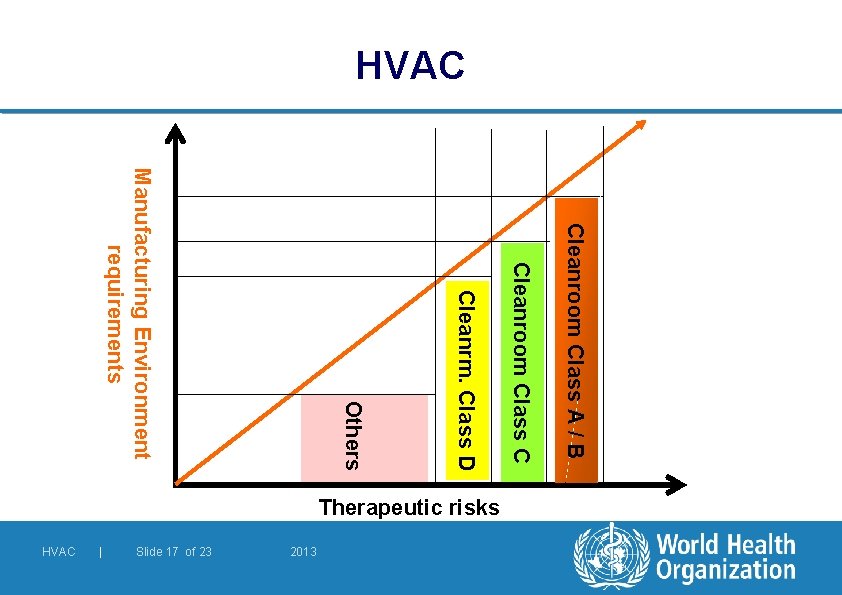
HVAC Cleanroom Class A / B Cleanroom Class C Cleanrm. Class D 2013 Slide 17 of 23 | HVAC Others Manufacturing Environment requirements Therapeutic risks

HVAC Level of protection and air cleanliness determined according to: l Product to be manufactured l Process to be used l Product susceptibility to degradation l Personnel should not be a source of contamination 4. 1. 16 - 17 HVAC | Slide 18 of 23 2013

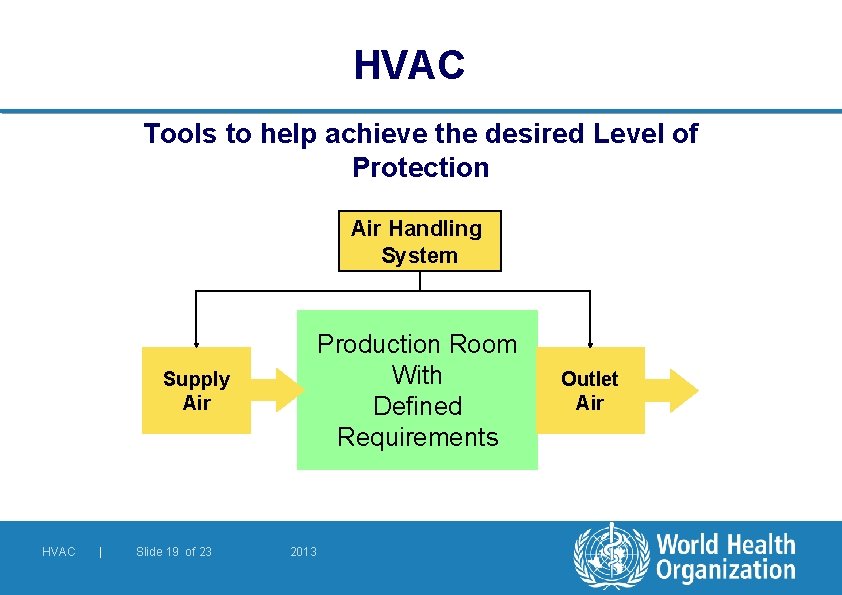
HVAC Tools to help achieve the desired Level of Protection Air Handling System Production Room With Defined Requirements Supply Air HVAC | Slide 19 of 23 2013 Outlet Air

HVAC Tools to help achieve the desired Level of Protection (2) Cleanroom Class defined by Critical Parameters Air Handling System HVAC | Slide 20 of 23 2013 Additional Measures

HVAC Examples of Levels of Protection Types of Clean room classes l A, B, C, D l Critical and controlled l Level 1, 2 or 3 l ISO 4. 8, 5, 7 or 8 HVAC | Slide 21 of 23 2013

HVAC Examples of levels of protection Level Condition Level 1 General Level 3 Controlled | Slide 22 of 23 Example of area Area with normal housekeeping, e. g. warehouse Area where steps are taken to protect exposed material/product, e. g. dispensing Level 2 Protected HVAC 4. 1. 17 Area with defined, controlled, monitored environmental conditions to prevent contamination and degradation 2013

HVAC All operations within a pharmaceutical facilility should be correlated to well-defined clean room classes, and can be included in a hygiene concept. Example: Cleanroom Class A B C Washing of containers X Preparation of solution for terminal sterilisation X Preparation of solutions for aseptic filling X Depyrogenisation of containers X Filling for terminal sterilisation Filling for aseptic process List other…. HVAC | Slide 23 of 23 2013 D X X