Good Laboratory Practice 1 GLP GLP 3 3

Good Laboratory Practice (개요) 소헌영 동일시마즈㈜


1. GLP란?

GLP의 정의 3. 용어의 정의 3. 1 우수실험실 운영규정 (농촌진흥청) 1) “우수실험실 운영규정”이라 함은 시험연구기관에서 행해지는 시험이 계획․실행․점검․기록․보고되는 체계적인 과정과 그 조건들에 관하여 총체 적 사항을 규정하는 것을 말한다. Key Words • 계획, Planned • 시행, Performed • 점검, Monitored • 기록, Recorded • 보고, Reported • 문서보관, Archived Good Laboratory Practice (GLP) is a quality system concerned with the organizational process and the conditions under which non-clinical health and environmental safety studies are planned, performed, monitored, recorded, archived and reported. (OECD의 GLP 정의)




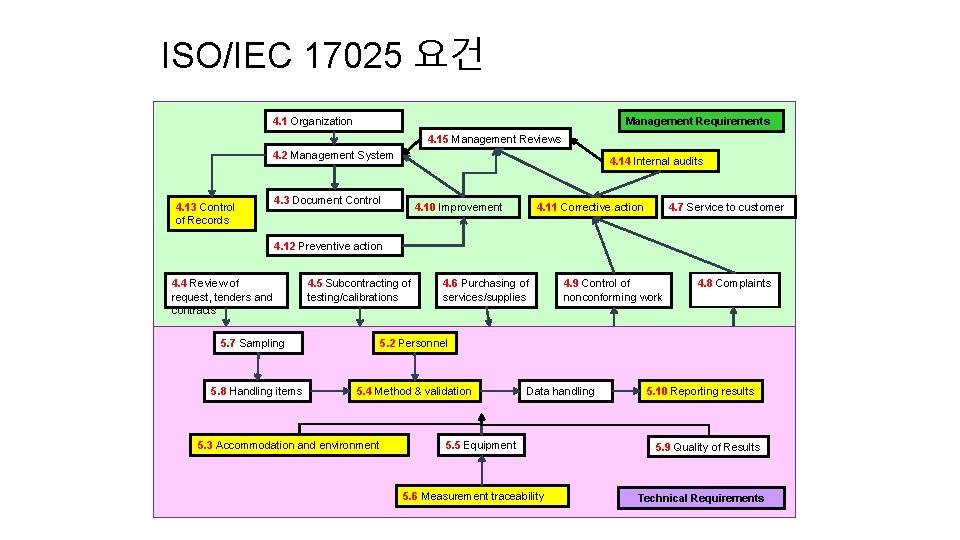
ISO/IEC 17025 요건 Management Requirements 4. 1 Organization 4. 15 Management Reviews 4. 2 Management System 4. 13 Control of Records 4. 14 Internal audits 4. 3 Document Control 4. 10 Improvement 4. 11 Corrective action 4. 7 Service to customer 4. 12 Preventive action 4. 4 Review of request, tenders and contracts 4. 5 Subcontracting of testing/calibrations 5. 7 Sampling 5. 8 Handling items 4. 6 Purchasing of services/supplies 4. 9 Control of nonconforming work 4. 8 Complaints 5. 2 Personnel 5. 4 Method & validation 5. 3 Accommodation and environment Data handling 5. 5 Equipment 5. 6 Measurement traceability 5. 10 Reporting results 5. 9 Quality of Results Technical Requirements


3. OECD GLP


OECD Environmental Health and Safety Publications Series on Principles of Good Laboratory Practice and Compliance Monitoring No. 1 , OECD Principles of Good Laboratory Practice No. 2, Revised Guides for Compliance Monitoring Procedures for Good Laboratory Practice (1995) No. 3, Revised Guidance for the Conduct of Laboratory Inspections and Study Audits (1995) No. 4, Quality Assurance and GLP (1992) No. 5, Compliance of Laboratory Suppliers with GLP Principles (1992) No. 6, The Application of the GLP Principles to Field Studies (1992) No. 7, The Application of the GLP Principles to Short-term Studies (1993) No. 8, The Role and Responsibilities of the Study Director in GLP Studies (1993) No. 9, Guidance for the Preparation of GLP Inspection Reports (1995) No. 10, The Application of the Principles of GLP to Computerised Systems (1995) No. 11, The Role and Responsibilities of the Sponsor in the Application of the Principles of GLP





Five points of all GLP texts 1. Resources: organization, personnel, facilities and equipment 2. Characterization: test items and test systems 3. Rules: study plans (or protocols) and written procedures 4. Results: raw data, final report and archives 5. Quality Assurance.


농업진흥청 GLP vs. OECD GLP 목차 GLP 기관지정기준 I. 총칙 1. 목적 2. 범위 3. 용어의 정의 II. 우수실험실 운영규정 1. 조직과 인원 2. 신뢰성보증업무 3. 시설 4. 기기, 재료, 시약 5. 시험계 6. 시험물질 및 대조물질 7. 표준작업지시서 8. 시험의 실시 9. 시험결과의 보고 10. 기록과 재료의 보관과 유지 OECD PRINCIPLES OF GOOD LABORATORY PRACTICE Section 1 Introduction Preface 1. Scope 2. Definition of terms Section II GLP Principles 1. Test Facilities Orgnisation and Personnel 2. Quality Assurance Programme 3. Facilities 4. Apparatus, Materials, and Reagents 5. Test System 6. Test and Reference Items 7. Standard Operating Procedures 8. Performance of Studies 9. Reporting of Study Results 10. Storage and Retention of Records and Materials

2. 2 Terms Concerning the Organisation of a Test Facility 3. 2 시험연구기관의 조직과 관련된 용어 1. 2. 3. 4. 5. 6. 7. 8. 9. 10. Test facility Test site Test facility management Test site management Sponsor Study Director Principle Investigator Quality Assurance Programme Standard Operating Procedure Master schedule 1. 2. 3. 4. 5. 6. 7. 8. 9. 시험장소 운영책임자 다지점시험장소 운영책임자 시험의뢰자 시험책임자 주임시험자 신뢰성보증업무 표준작업지침서 시험일정 총괄표 「농약등의 시험연구기관 지정 및 관리기준」”별표 2 “GLP기관 지정기준”에는 Test facility 정의 없음

2. 3 Terms Concerning the Non-Clinical Health and Environmental Safety Study 3. 3 인간건강과 환경영향에 대한 시험과 관계된 용어 1. Non-clinical health and environmental safety study 2. Short-term study 3. Study plan 4. Study plan amendment 5. Study plan deviation 6. Test system 7. Raw data 8. Specimen 9. Experimental starting date 10. Experimental completion date 11. Study initiation date 12. Study completion date 1. 시험계획서 2. 시험계획서의 변경 3. 시험계획서의 이탈 4. 시험계 5. 시험기초자료 6. 검체 7. 실험개시일 8. 실험종료일 9. 시험개시일 10. 시험종료일 농약등의 시험연구기관 지정 및 관리기준”별표 2의 GLP기관 지정기준에는 Non-clinical health and environmental safety study와 Short-term study 사항 없음

2. 4 Terms Concerning the Test Item 3. 4 시험물질 관련 용어 1. 2. 3. 4. Test item Reference item Batch Vehicle any agent which serves as a carrier used to mix, disperse, or solubilise the test item or reference item to facilitate the administration/application to the test system 1. 2. 3. 4. 시험물질 대조물질 제조단위 부형제 시험물질 또는 대조물질이 시험계 에 용이하게 적용되도록 시험물질 또는 대조물질을 혼합, 분산, 용 해시키는데 이용되는 물질

II. 우수실험실 운영규정 1. 조직과 인원 1. 2. 3. 4. 운영책임자 시험책임자 주임시험자 시험담당자 (Test Facility Management) (Study Director) (Principal Investigator) (Study personnel) 2. 신뢰성보증업무 1. 신뢰성보증업무담당자 (Quality Assurance Programme)


- Slides: 27