GOOD DOCUMENTATION PRACTICES GDP PRESENTED BY SATYAVAN SINGH

GOOD DOCUMENTATION PRACTICES (GDP) PRESENTED BY: SATYAVAN SINGH GENERAL MANAGER QUALITY INTAS PHARMACEUTICALS LIMITED SIKKIM

General Principle of Documentations • • Design (correct, complete, current & traceable) Prepared Reviewed Proper Distribution

Why GDP ? • Good documentation practices (GDP) is required in order to ensure an auditable account of work performed. • GDP is required for all documentations included in a fully developed quality system.

Different types of documents • Batch records • Specifications • Validation & Qualification Records • Calibration Records • Standard procedure • Room Temperature/ %RH Records • Stability Study Summary Report • Machine Logs • Product and sample labels • Analytical Methods • Manufacturing Procedures

Characteristics of Good Documents – – – – ACCURATE AUTHENTIC CLEAR COMPLETE CURRENT VERSION LEGIBLE PERMANENT

Some Guidance Documents • Schedule M of Drug And Cosmetics Act 1940 • ISO - 9001 : 2008 • US FDA CFR sections – 21 CFR 11, 210 & 211

Schedule M 7

ISO – 9001 : 2008 Section 4: Quality Management System 4. 2. 2 Control of Documents 4. 2. 3 Control of Records 8

Document • An approved instruction either in paper or electronic form which guides about how an activity shall be executed. 9

Record • A record is often considered as document. • Records provide evidence that activities have been performed or results have been achieved. • They always document the past. 10

Document Vs Record • A document can be a procedure, specification, drawing, work instruction and the record is also a document with the difference that it provides evidence of compliance with established requirements and effectiveness of the operation of QMS. 11

How are mistakes corrected? • Draw a single line through the error • Make the correction next to the error • Write an explanation for the error • Sign and date the correction.

Some tips on GDP • Records should be filled online. • Superseded documents should be retained for a specific period of time. • Records should be retained for at least one year after the expiry date of the finished product. • Concise, legible, accurate and traceable.
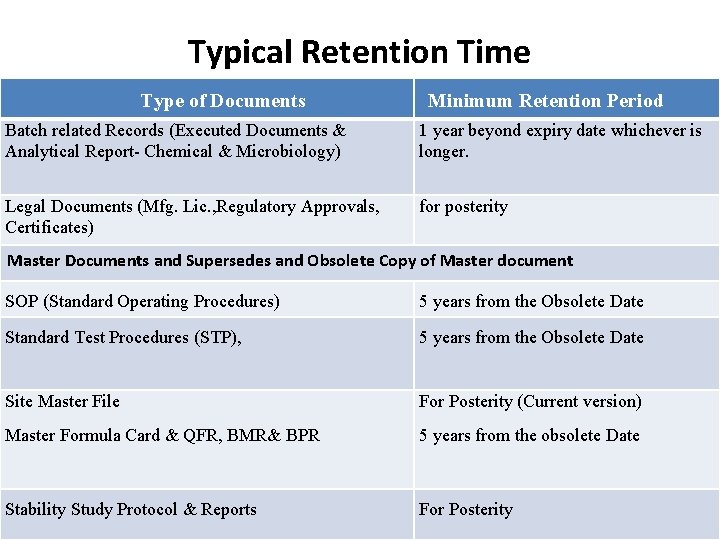
Typical Retention Time Type of Documents Minimum Retention Period Batch related Records (Executed Documents & Analytical Report- Chemical & Microbiology) 1 year beyond expiry date whichever is longer. Legal Documents (Mfg. Lic. , Regulatory Approvals, Certificates) for posterity Master Documents and Supersedes and Obsolete Copy of Master document SOP (Standard Operating Procedures) 5 years from the Obsolete Date Standard Test Procedures (STP), 5 years from the Obsolete Date Site Master File For Posterity (Current version) Master Formula Card & QFR, BMR& BPR 5 years from the obsolete Date Stability Study Protocol & Reports For Posterity

A Famous Proverb says: “IF YOU HAVE NOT DOCUMENTED AN ACTIVITY, YOU HAVE NOT DONE

U O Y K N A TH
- Slides: 16